Clear Sky Science · en
Histidine-rich coiled-coils promote zinc-dependent self-assembly and curing of porous mussel glues
How Mussels Make Super Glue Underwater
Mussels cling to wave-battered rocks with a natural glue that scientists have tried to imitate for decades. This study reveals that a long-overlooked ingredient—rather than the usual chemical star—quietly does much of the hard work. By uncovering how a special protein and the metal zinc help mussels build a strong, sponge-like glue, the research opens new routes to gentler, longer-lasting adhesives for medicine, engineering, and everyday wet surfaces.
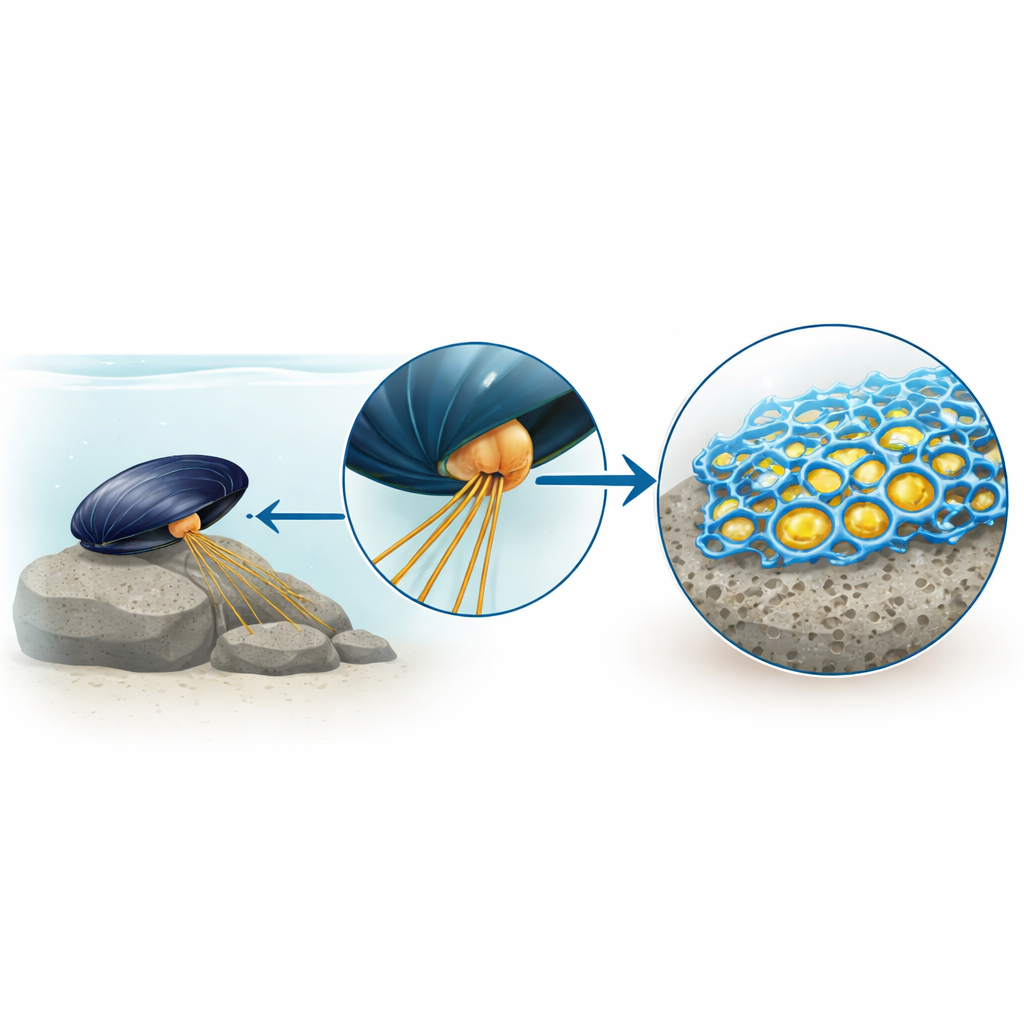
From Sticky Mystery to New Glue Hero
For years, the mussel glue story centered on a modified building block called DOPA, which sticks strongly to surfaces and metals. Synthetic materials inspired by DOPA have been impressive, but still fall short of real mussel glue. The authors suspected that other, unstudied proteins inside the mussel’s glue-making glands might be missing pieces of the puzzle. By isolating tiny glue-filled sacs from the mussel foot and watching them burst and solidify, they found that a subset of proteins formed a solid scaffold with a porous, foam-like structure, while the well-known DOPA-rich proteins stayed mostly as a fluid within the pores.
Discovery of a Hidden Structural Protein
When the team dissolved only the solid part of this miniature glue and analyzed its contents, a single dominant protein stood out. They named it mefp-12, and discovered that it is rich in the amino acid histidine and conserved across several mussel species, suggesting it is important for survival. Imaging of mussel foot tissue showed that cells in the glue-producing gland make this protein specifically for the plaque—the flattened pad that anchors the mussel to rock. Computer-based structure predictions indicated that mefp-12 has a long central segment that prefers to form rope-like bundles known as coiled coils, and several compact domains that resemble metal-binding “zinc finger” regions, all hinting that metal ions, especially zinc, might be crucial for its behavior.
How Zinc and pH Turn Liquid Droplets into Solid Foam
To test this idea in a simple way, the researchers made a 30–amino acid fragment of the central region of mefp-12. In salty water at slightly acidic conditions—similar to the storage environment inside the mussel foot—this short piece gathered into liquid droplets when zinc was present, but not with other metals such as copper or nickel. Raising the pH toward that of seawater triggered a striking transformation: the droplets coalesced, spread on surfaces, and then “cured” into a rigid, open-cell porous solid that closely resembled natural mussel glue under the microscope. Spectroscopic and nuclear magnetic resonance measurements showed that, during this transition, the protein pieces shift from a loose, disordered state into more ordered spiral shapes that pack together, while histidine side groups form sturdy bridges to zinc ions. These metal bridges lock the coiled structures into a stable, yet reversible, network.

A Tough, Reusable Network Built for Waves
The authors propose a new picture of how mussel glue forms. Inside the foot, mefp-12 is stored in acidic, zinc-containing droplets along with other glue proteins. As the secretion is released into the higher-pH seawater, histidine groups on mefp-12 begin to bind zinc, driving the protein to reorganize into coiled bundles that connect into a porous framework. DOPA-rich adhesive proteins remain as a more fluid phase inside the pores, ready to wet and re-wet surfaces. Later, other metals such as iron and vanadium strengthen the DOPA-containing components, producing an interlocking double network. The histidine–zinc framework likely acts as a sacrificial, yet recoverable, shock absorber, helping the plaque dissipate the energy of crashing waves while maintaining adhesion over long periods.
What This Means for Future Wet Glues
By shifting attention from DOPA alone to a partnership between histidine-rich proteins and zinc, this study rewrites the mussel glue playbook. It shows that the instructions for forming a self-assembling, porous solid are encoded directly in the sequence of mefp-12, activated simply by salt, metal ions, and a change in pH. These insights suggest new design rules for man-made adhesives and soft materials: use controlled phase separation, metal–protein bridges, and foam-like architectures to create glues and gels that cure in wet conditions, absorb damage, and potentially self-heal, without relying solely on one highly reactive chemical group.
Citation: Rivard, M.D., Poulhazan, A., Renner-Rao, M.J. et al. Histidine-rich coiled-coils promote zinc-dependent self-assembly and curing of porous mussel glues. Nat Commun 17, 2809 (2026). https://doi.org/10.1038/s41467-026-69504-y
Keywords: mussel adhesion, bioinspired glue, protein self-assembly, metal-coordinated materials, wet adhesives