Clear Sky Science · en
Baboon endogenous retrovirus (ERV) envelope pseudotyped lentiviral vectors outperform human ERV lentivectors for transduction of T, B, NK and HSPCs
Turning Viruses into Helpful Delivery Trucks
Modern gene therapies often depend on hollowed-out viruses that act like microscopic delivery trucks, carrying corrective genes into patients’ blood and immune cells. But not all viral “trucks” are equally good at reaching the right destinations. This study compares two such vehicles—one built from a baboon virus and one from a human endogenous virus—to see which can more efficiently deliver genes into key immune cells and blood stem cells that are central to future cancer, immune, and genetic therapies. 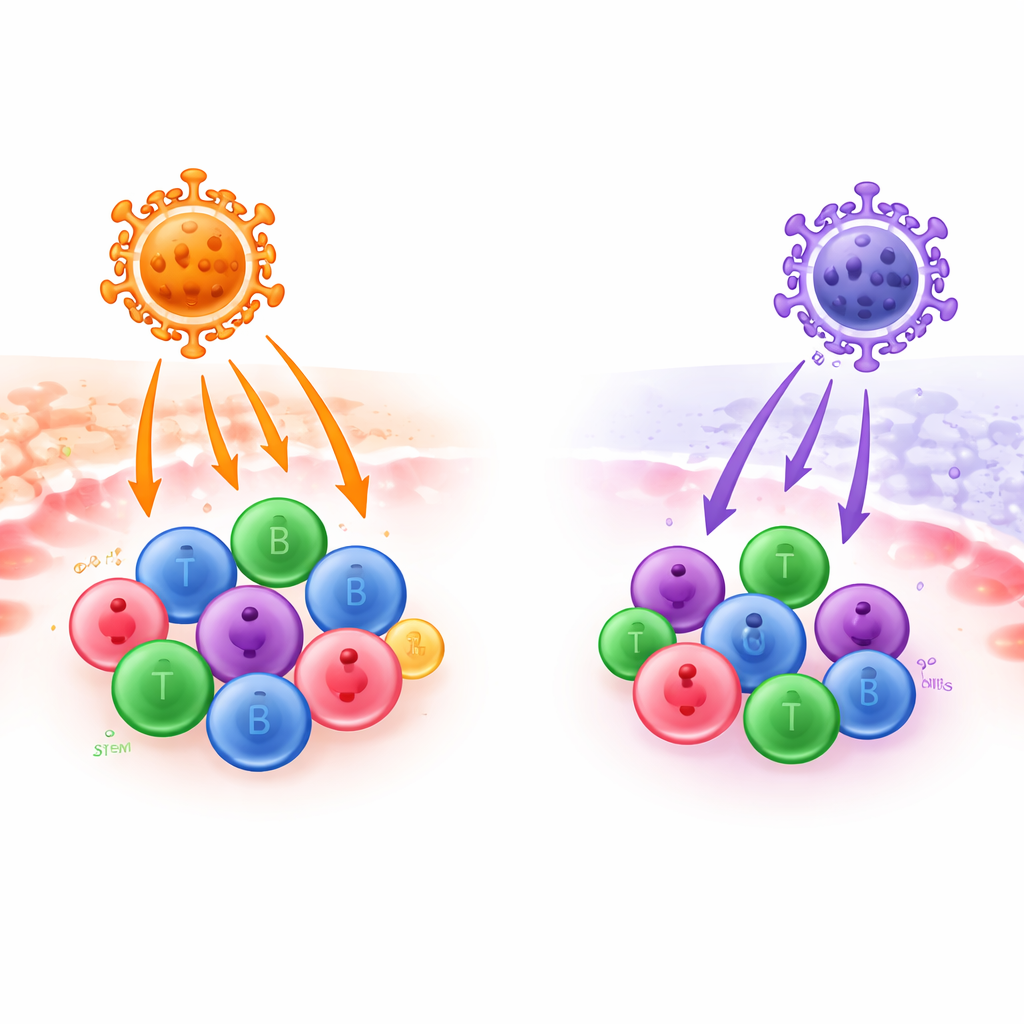
Why the Choice of Viral Shell Matters
To change the behavior of immune cells such as T cells, B cells, natural killer (NK) cells, and blood-forming stem cells, gene therapy researchers often use lentiviral vectors: disabled viruses that can slip new DNA into a cell’s genome. The outer coat, or envelope, of these vectors determines which cells they can enter by binding to specific “doorway” proteins on the cell surface. A widely used envelope, called VSV-G, struggles with resting immune cells because its doorway protein is scarce there. That means doctors must strongly stimulate cells with cocktails of growth signals before gene transfer, a step that can unintentionally alter cell identity or reduce the regenerative potential of stem cells.
Baboon Versus Human Viral Envelopes
The authors focused on two envelopes that both use the same pair of cellular doorways, transport proteins known as ASCT-1 and ASCT-2, which are naturally abundant on many blood and immune cells. One envelope comes from a baboon endogenous retrovirus (BaEV), and the other from a human endogenous retrovirus of the W family (HERV-W), better known for helping form the placenta. The team engineered lentiviral vectors with each envelope and carefully optimized their production. While they tested several modified versions of the human envelope, the unmodified HERV-W version actually produced the highest usable vector yields, though still at lower titers than baboon-based vectors. 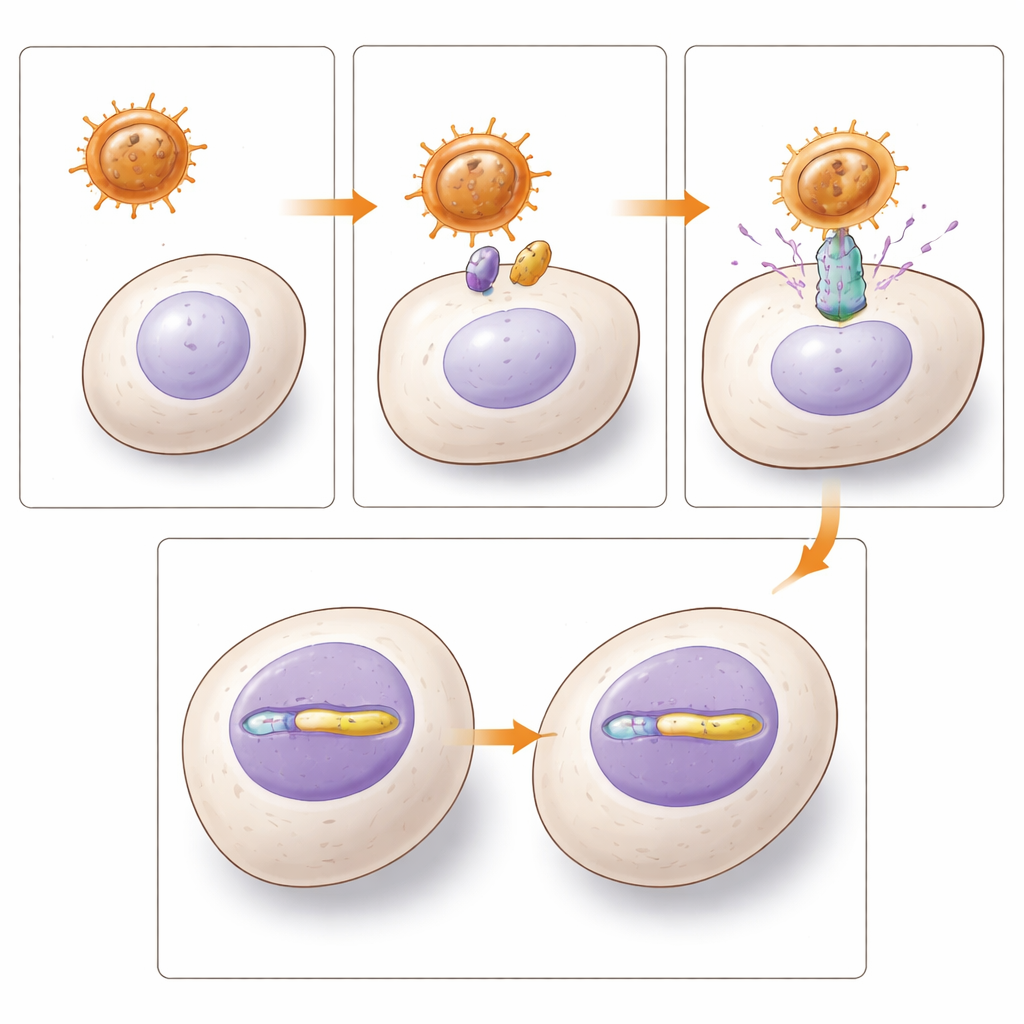
How Well Each Vector Reaches Immune Cells
The researchers then asked how efficiently each type of vector could introduce a reporter gene into human immune cells harvested from donors. In T cells stimulated through their antigen receptor—a situation similar to how T cells are prepared for CAR-T therapies—both vectors worked, but the baboon vector routinely produced around twice as many modified cells as the human vector at the same dose. Under milder stimulation with survival signals IL-7 and IL-15, the gap widened dramatically: baboon vectors reached about 70–80% of T cells, while HERV-W vectors reached only around 10%. When applied to B cells and NK cells, the baboon vector again came out clearly ahead. It could drive high gene transfer at relatively modest doses, whereas the human envelope needed far higher doses to achieve lower levels of modification.
Reaching the Source: Human Blood Stem Cells
Because blood-forming stem and progenitor cells (CD34+ HSPCs) sit at the top of the blood hierarchy, they are prime targets for correcting inherited blood diseases. The team showed that both vectors could modify these stem cells after a brief exposure to growth factors. However, the baboon-based vector achieved strong gene transfer at medium doses, while the HERV-W vector required much higher dosing for similar results. To see whether the modified stem cells could still rebuild a human-like blood system, the researchers transplanted them into a specialized immunodeficient mouse strain (NBSGW) that supports human blood development. In all six mice receiving BaEV-treated cells, over 80% of human blood cells across bone marrow, spleen, thymus, and blood carried the introduced gene. In contrast, only one of five mice receiving HERV-W–treated cells reached that level; several showed much lower percentages, revealing more variable and less reliable performance.
What This Means for Future Gene Therapies
For a layperson, the key message is that not all viral delivery trucks are created equal, even if they seem to use the same doors to enter cells. In this head-to-head comparison, the baboon-derived envelope allowed more consistent, high-level gene delivery into a wide range of human immune cells and blood stem cells than the human-derived HERV-W envelope. That makes BaEV-based lentiviral vectors especially attractive for therapies that depend on efficiently and gently reprogramming T cells, B cells, NK cells, and stem cells without excessive pre-stimulation. The human envelope still performs better than some traditional options and may be useful in specific situations, but the baboon envelope currently looks like the more powerful and reliable tool for many next-generation gene and cell therapies.
Citation: Périan, S., Castellano, E., Costa, C. et al. Baboon endogenous retrovirus (ERV) envelope pseudotyped lentiviral vectors outperform human ERV lentivectors for transduction of T, B, NK and HSPCs. Gene Ther 33, 144–155 (2026). https://doi.org/10.1038/s41434-025-00587-w
Keywords: lentiviral vectors, gene therapy, immune cells, hematopoietic stem cells, viral envelopes