Clear Sky Science · en
Redox cycling nitroxide limits cellular iron availability and selectively inhibits iron-sulfur cluster metabolism
Why This Matters for Everyday Health
Our cells depend on iron not only to carry oxygen in the blood, but also to power energy production and repair DNA. Yet too much of the wrong kind of iron can fuel harmful chemical reactions and even trigger a form of cell death. This study explores how a small molecule called Tempol, already tested in several disease models, subtly changes how cells handle iron. By nudging iron into a less usable state and opposing the action of vitamin C, Tempol exposes a previously underappreciated layer of control over iron use that may be relevant to conditions ranging from neurodegeneration to cancer.
Iron’s Delicate Balancing Act
Inside cells, iron is used either as free ions or built into special structures called iron–sulfur clusters that help many enzymes do their jobs in energy production, DNA copying, and other vital processes. Because iron can easily switch between chemical states, it is both useful and potentially damaging. Cells therefore rely on a network of sensors and regulators to keep a small, carefully managed pool of available iron. Two proteins, IRP1 and IRP2, sense iron status and adjust how much iron is imported, stored, or exported. At the same time, vitamin C helps keep iron in a chemically “reduced” form that many enzymes require to function properly.
A Different Way to Limit Iron
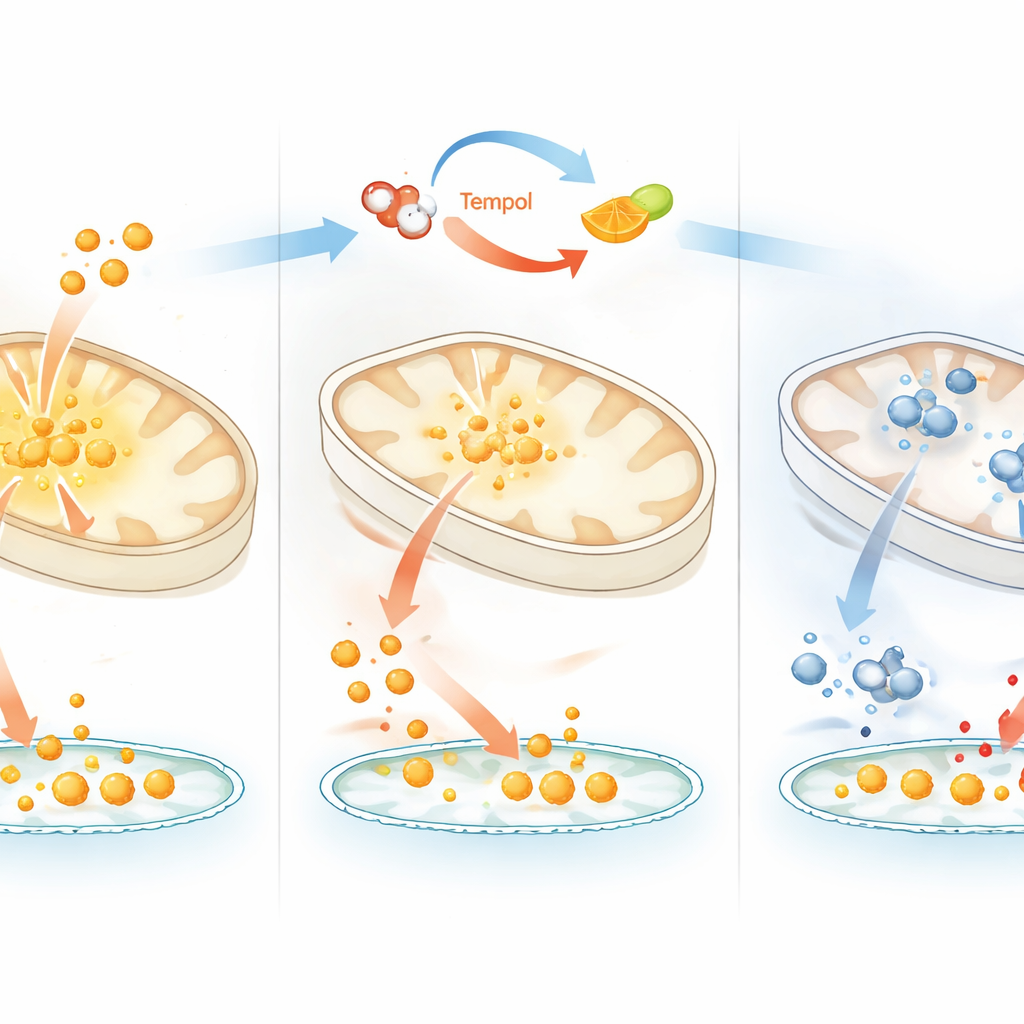
The researchers focused on Tempol, a redox-active compound previously thought mainly to act as an antioxidant and to directly damage iron–sulfur clusters. They found that Tempol instead behaves more like an “anti–vitamin C.” In cell cultures, Tempol lowered the pool of usable iron and stabilized IRP2, a hallmark of iron shortage. However, unlike classic iron-binding drugs, the effects of Tempol could not be fully reversed simply by adding extra iron, and Tempol did not strongly strip iron from mitochondria in the same way. Instead, its impact was almost completely undone by adding vitamin C, which restored cell growth, iron use, and iron–sulfur cluster–dependent enzymes. This points to a mechanism in which Tempol interferes with vitamin C–driven recycling of iron to its active state, shifting iron toward a less reactive form.
Selective Hit on Key Cell Processes
Tempol did not affect all cell types equally. In a panel of dozens of human cancer cell lines, some tolerated Tempol well, while others showed sharply reduced growth. Sensitive cells lost activity of multiple iron–sulfur cluster enzymes, especially a mitochondrial enzyme called ACO2 that is central to energy metabolism. Genetic datasets revealed that cell lines more dependent on ACO2 were also more vulnerable to Tempol, suggesting that cells with a heavier reliance on iron–sulfur chemistry are less able to cope when iron redox balance is disturbed. Notably, lowering oxygen levels, closer to what many tissues experience in the body, protected iron–sulfur clusters and cell growth from Tempol without preventing the iron-sensing response, indicating that oxygen itself contributes to the damage seen under standard lab conditions.
Iron, Cell Death, and Disease Links
Beyond metabolism, Tempol’s redox-based iron limitation had two striking signaling effects. First, it quickly stabilized HIF1α, a protein that responds to low oxygen and depends on both iron and vitamin C–supported enzymes to be degraded. Second, it consistently suppressed ferroptosis, a type of cell death driven by iron-catalyzed damage to membrane fats. Both effects appeared at Tempol doses that reduced usable iron but before obvious loss of iron–sulfur clusters, indicating that modest shifts in iron’s chemical state can strongly influence stress responses and cell survival. The authors also showed that similar behavior was not reproduced by generic antioxidants or reducing agents, underlining the specific vitamin C–related chemistry of Tempol.
What This Means for Future Therapies
In plain terms, the study shows that Tempol acts as a targeted disruptor of how cells chemically “tune” iron, largely by opposing vitamin C. This reduces the form of iron that many enzymes need, dampens iron-driven cell death, and, in certain cells that depend heavily on iron–sulfur cluster enzymes like ACO2, slows growth. Because iron mismanagement is implicated in brain disorders, infections, and cancer, understanding and potentially harnessing this redox-based iron limitation opens the door to new strategies that do not simply remove iron, but instead change how cells can use it.
Citation: Terzi, E.M., Fujihara, K.M., Molenaars, M. et al. Redox cycling nitroxide limits cellular iron availability and selectively inhibits iron-sulfur cluster metabolism. Cell Death Discov. 12, 165 (2026). https://doi.org/10.1038/s41420-026-03042-w
Keywords: iron metabolism, Tempol, vitamin C, iron–sulfur clusters, ferroptosis