Clear Sky Science · en
Deltex E3 ubiquitin ligase 2 potentiates STING-mediated type I interferon response by K63-linked ubiquitination
How Cells Sense Dangerous DNA
Our bodies are constantly on the lookout for viral invaders and rogue tumor cells. One of the key alarm systems for this early defense is a protein called STING, which detects stray DNA in the wrong place inside cells and triggers powerful antiviral and anticancer responses. This paper uncovers a previously overlooked helper protein, DTX2, that acts like a molecular booster for STING, making this alarm system louder and more effective against infections and tumors.
A Cellular Burglar Alarm for DNA
When viruses such as herpes simplex virus invade, or when damaged cells spill their DNA, fragments of double-stranded DNA appear where they do not belong inside our cells. A sensor enzyme called cGAS detects this stray DNA and produces a small messenger molecule that switches on STING, a protein sitting on an internal membrane compartment. Once activated, STING travels to another compartment known as the Golgi and kicks off a signaling chain that ends with the production of type I interferons and inflammatory molecules. These substances act as chemical sirens, alerting and activating immune cells to fight viruses and, in many cases, to recognise and attack tumors. 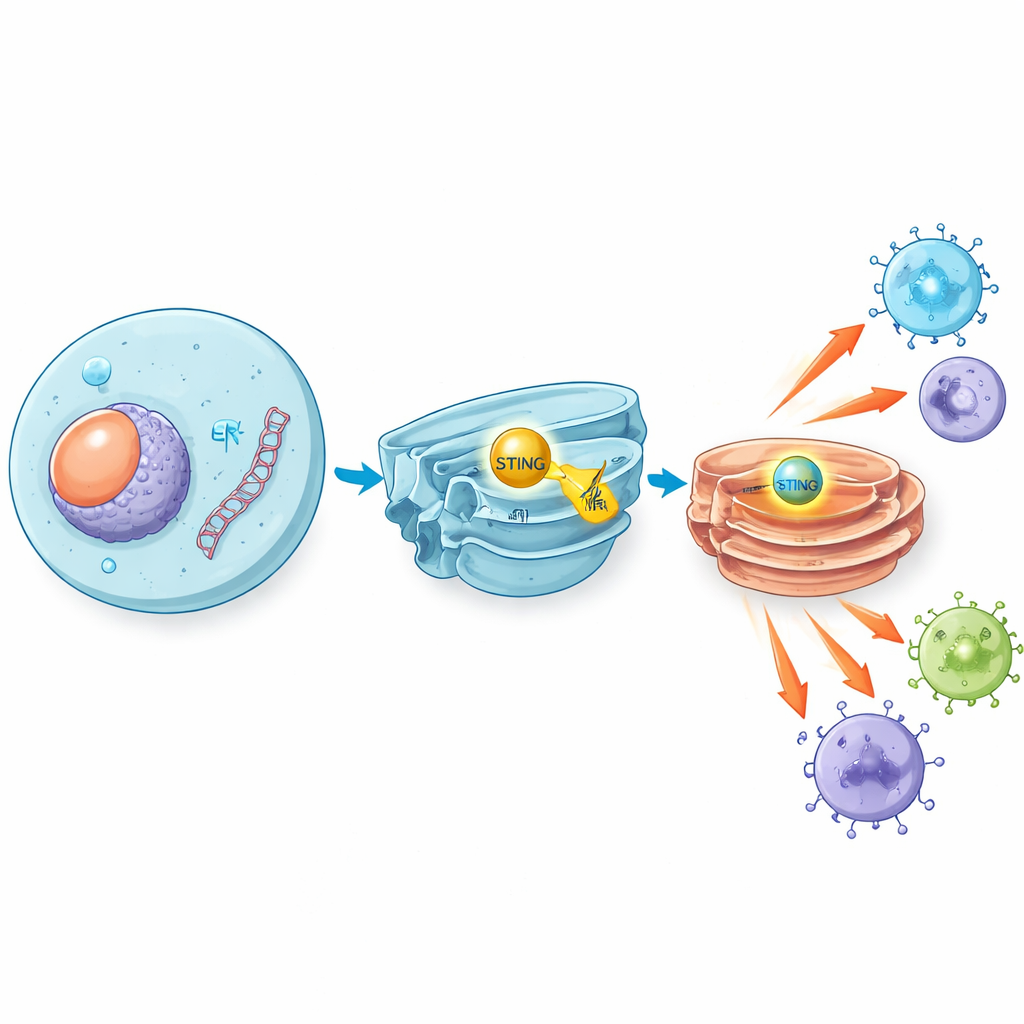
Discovering a New Helper for the STING Pathway
To find out which enzymes might fine-tune this DNA alarm system, the researchers infected mouse immune cells with herpes virus and measured which genes were turned up or down. Among hundreds of enzymes, one stood out: DTX2, an E3 ubiquitin ligase, meaning it can attach small ubiquitin chains to other proteins to alter their behavior. The team generated mice lacking the Dtx2 gene and found that their immune cells produced far less interferon and fewer antiviral signals when exposed to viral DNA, synthetic DNA, or a direct STING activator. This drop was seen not only in immune cells like macrophages but also in non-immune cells, and it was specific to DNA-triggered responses rather than RNA viruses, pointing to a focused role for DTX2 in the DNA-sensing pathway.
How DTX2 Tunes Up the STING Signal
Digging deeper, the scientists showed that DTX2 physically binds to STING and adds a particular form of ubiquitin chain, known as K63-linked chains, onto two specific spots on STING. Unlike other ubiquitin tags that mark proteins for destruction, these chains act more like functional add-ons that change where a protein goes and how it signals. When DTX2 was missing, STING levels stayed normal, but its ability to move from its starting membrane to the Golgi and recruit the next signaling enzyme, TBK1, was greatly reduced. The team pinpointed two key amino acids on STING, K236 and K370, as the attachment points for these chains. When they mutated these sites so the chains could not form, STING failed to travel properly, TBK1 was not efficiently recruited, and the downstream interferon response was blunted.
Protection Against Viruses and Support for Cancer Immunity
The consequences of this molecular tuning were clear in living animals. Mice lacking Dtx2 succumbed more readily to herpes virus infection, showed more severe lung damage, higher viral loads in multiple organs, and lower levels of protective interferons and inflammatory signals in their blood. In cancer models, the story flipped: higher DTX2 levels were associated with stronger type I interferon signatures and increased infiltration of key immune fighters such as dendritic cells, killer T cells, and natural killer cells in human tumor data sets. In mouse head and neck cancer models, tumors engineered to make more DTX2 responded better to a STING-activating drug and to immune checkpoint therapy, shrinking more and harboring more immune cells than control tumors. 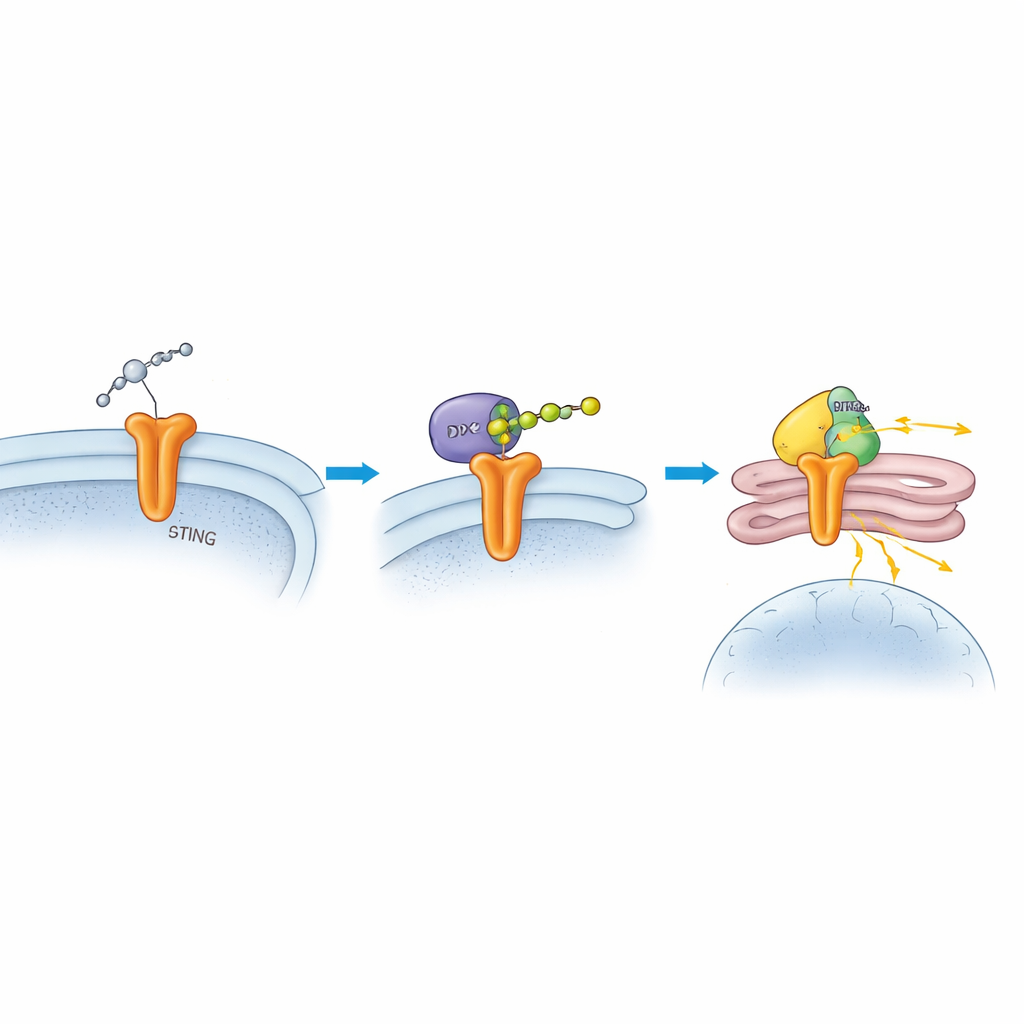
Why This Molecular Booster Matters
To a non-specialist, the main message is that DTX2 acts as a crucial amplifier for one of the immune system’s earliest warning pathways. By decorating STING with the right kind of molecular tags at the right spots, DTX2 helps STING move to where it needs to be inside the cell and fully switch on antiviral and antitumor defenses. When DTX2 is absent or its action is blocked, the alarm is muted: viruses spread more easily and tumors may find it easier to evade immune attack. This makes DTX2 a promising target for new therapies that could boost our natural defenses against infections and enhance the success of cancer immunotherapies.
Citation: Liu, Z., Li, R., Fan, C. et al. Deltex E3 ubiquitin ligase 2 potentiates STING-mediated type I interferon response by K63-linked ubiquitination. Cell Death Dis 17, 424 (2026). https://doi.org/10.1038/s41419-026-08659-4
Keywords: innate immunity, STING pathway, interferon signaling, cancer immunotherapy, viral infection