Clear Sky Science · en
β1,4-galactosyltransferase III drives retinoblastoma invasion via activation of integrin-FAK axis
Why This Childhood Eye Cancer Study Matters
Retinoblastoma is a rare eye cancer that strikes very young children, and while many cases can now be cured with modern therapies, the disease can still turn deadly when tumor cells break out of the eye and spread. This study reveals how a single sugar-adding enzyme on tumor cells helps them grow faster, grip their surroundings more tightly, and invade nearby tissues—and how a natural compound might block this process. Understanding this molecular chain reaction could lead to safer, eye-preserving treatments for children at highest risk.
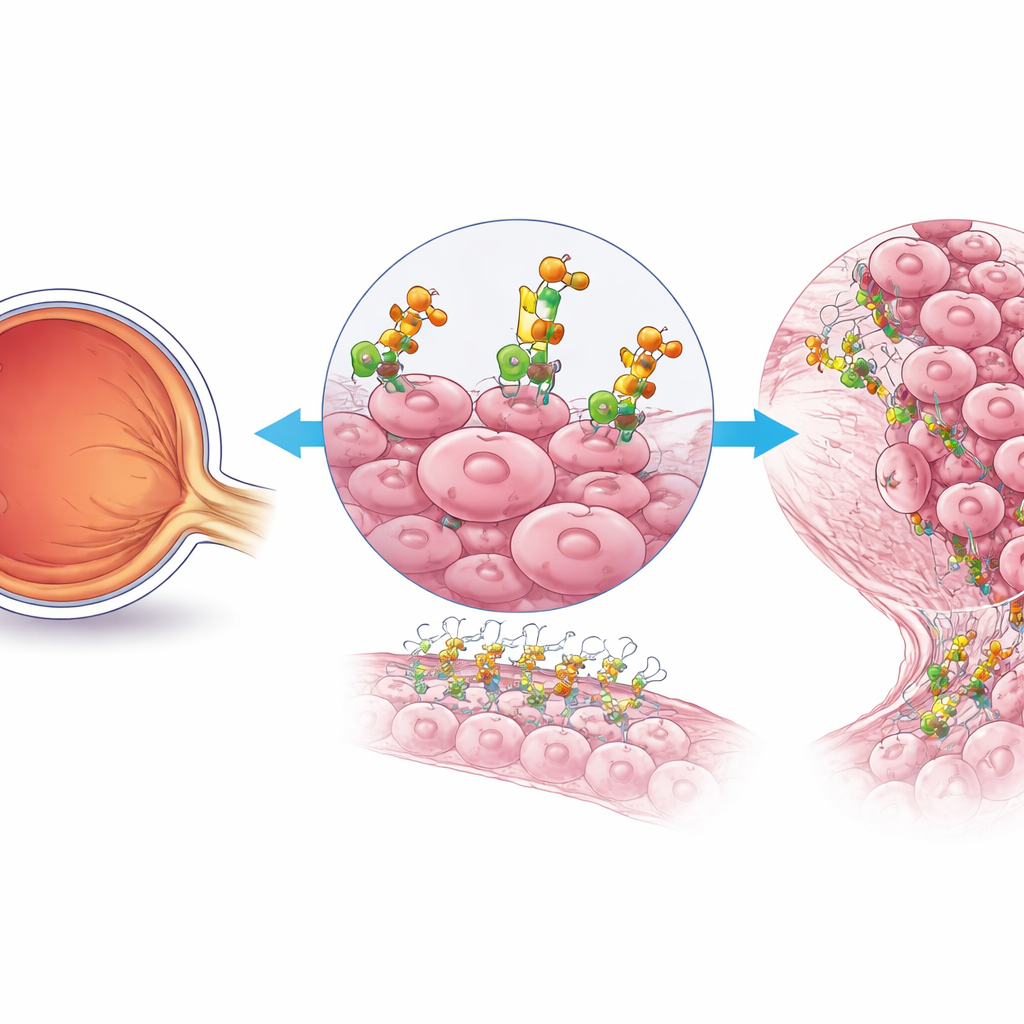
A Hidden Helper of Tumor Growth
The researchers focused on retinoblastoma cells that are especially aggressive and fast-dividing. Using single-cell genetic analysis of patient samples, they found that these cells ramp up activity in a specific sugar-building pathway on their surface. One enzyme stood out: B4GALT3, which decorates proteins and fats with chains of sugar molecules. Compared with normal retina, retinoblastoma tissue—especially in advanced, outward-spreading tumors—showed much higher levels of B4GALT3. In both human samples and mouse models, B4GALT3 was concentrated in regions packed with highly proliferating tumor cells, suggesting it plays a central role in the most dangerous part of the cancer.
How Tumor Cells Grip and Break Through Barriers
Cancer cells must latch onto and remodel the supportive mesh around them to grow and spread. In this study, the team discovered that B4GALT3 modifies a key “grappling hook” protein on the tumor cell surface called β1-integrin by adding sugar chains to it. This chemical decoration strengthens the receptor’s ability to bind to fibronectin, a major component of the eye’s supporting tissue. When the scientists reduced B4GALT3 in retinoblastoma cell lines, the cells divided more slowly, stuck less well to fibronectin, and showed weaker activity in an internal signaling protein called FAK and its downstream survival pathway. Boosting B4GALT3 had the opposite effect: cells grew faster, clung more firmly, and turned on this growth-and-survival circuitry.
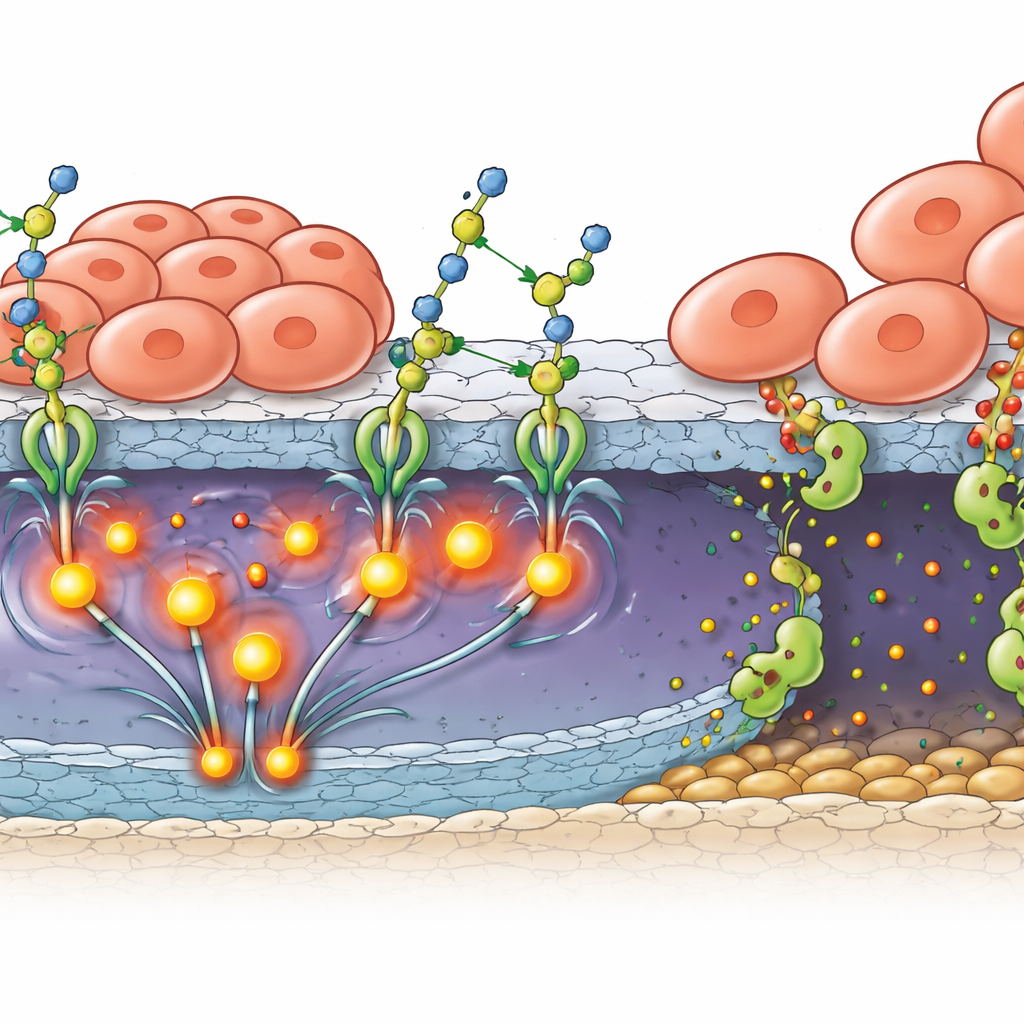
From Stronger Grip to Active Invasion
Attachment alone does not explain how tumors pierce the natural barriers that protect the back of the eye. The team showed that B4GALT3 also drives production and activation of MMP2, a protein that chews through the structural proteins in basement membranes. In patient samples and mouse tumors, B4GALT3 and MMP2 were found together in tumor cells near the retina’s outer border. In lab-grown models where retinoblastoma cells were layered above retinal pigment epithelial cells—the cells that help form the outer blood–retinal barrier—tumor cells with extra B4GALT3 caused the barrier layer to lose its tight sealing proteins and become discontinuous. Blocking either FAK or MMP2 could restore much of this barrier integrity, linking B4GALT3’s sugar-adding activity to a full invasion program: stronger grip, stronger signaling, and more powerful local tissue digestion.
Proof in Animal Models and a Natural Compound Blocker
In mice injected with retinoblastoma cells, tumors engineered to overproduce B4GALT3 grew larger, caused bulging of the eye, showed more abnormal surface sugars, and contained more dividing and fewer dying cancer cells. Tumors with reduced B4GALT3 did the opposite, remaining smaller and less invasive. To turn this insight into a potential therapy, the researchers sifted through a library of natural compounds using computer models of B4GALT3’s structure. They identified myricoside, a plant-derived molecule that fit snugly into the enzyme’s active pocket. In cell tests, myricoside made retinoblastoma cells less viable, reduced their grip on fibronectin, lowered sugar decoration on β1-integrin, and dampened the FAK survival pathway, mimicking genetic loss of B4GALT3.
What This Could Mean for Future Treatments
When delivered directly into the eyes of tumor-bearing mice, myricoside shrank tumors, reduced invasive features, decreased B4GALT3 and MMP2 signals, restored barrier proteins, and increased cancer cell death—without obvious harm to normal retinal layers. Altogether, the work paints a clear picture: B4GALT3 sits near the top of a chain that lets retinoblastoma cells grip, signal, and carve their way through the eye’s defenses. For non-specialists, the takeaway is that a sugar-adding enzyme, once considered a background player, is in fact a key switch for making this childhood cancer more invasive. Targeting B4GALT3 with drugs like myricoside could help stop tumors from breaking out of the eye, improving the chances that children can keep both their sight and their lives.
Citation: Tang, J., Li, J., Wang, M. et al. β1,4-galactosyltransferase III drives retinoblastoma invasion via activation of integrin-FAK axis. Cell Death Dis 17, 336 (2026). https://doi.org/10.1038/s41419-026-08620-5
Keywords: retinoblastoma, cancer invasion, glycosylation, integrin signaling, targeted therapy