Clear Sky Science · en
Lactate-mediated macrophage polarization promotes splenomegaly in acute erythroleukemia
Why this matters for patients and families
When blood cancers like leukemia spread beyond the bone marrow, they can cause the spleen to swell dramatically, leading to pain, anemia, and life‑threatening complications. This study asks a deceptively simple question: what, exactly, makes the spleen grow so large in a rare but aggressive leukemia called acute erythroleukemia? The researchers uncover an unexpected culprit—a common metabolic by‑product called lactate—and show that dialing down lactate production in mice can shrink the spleen and extend survival.
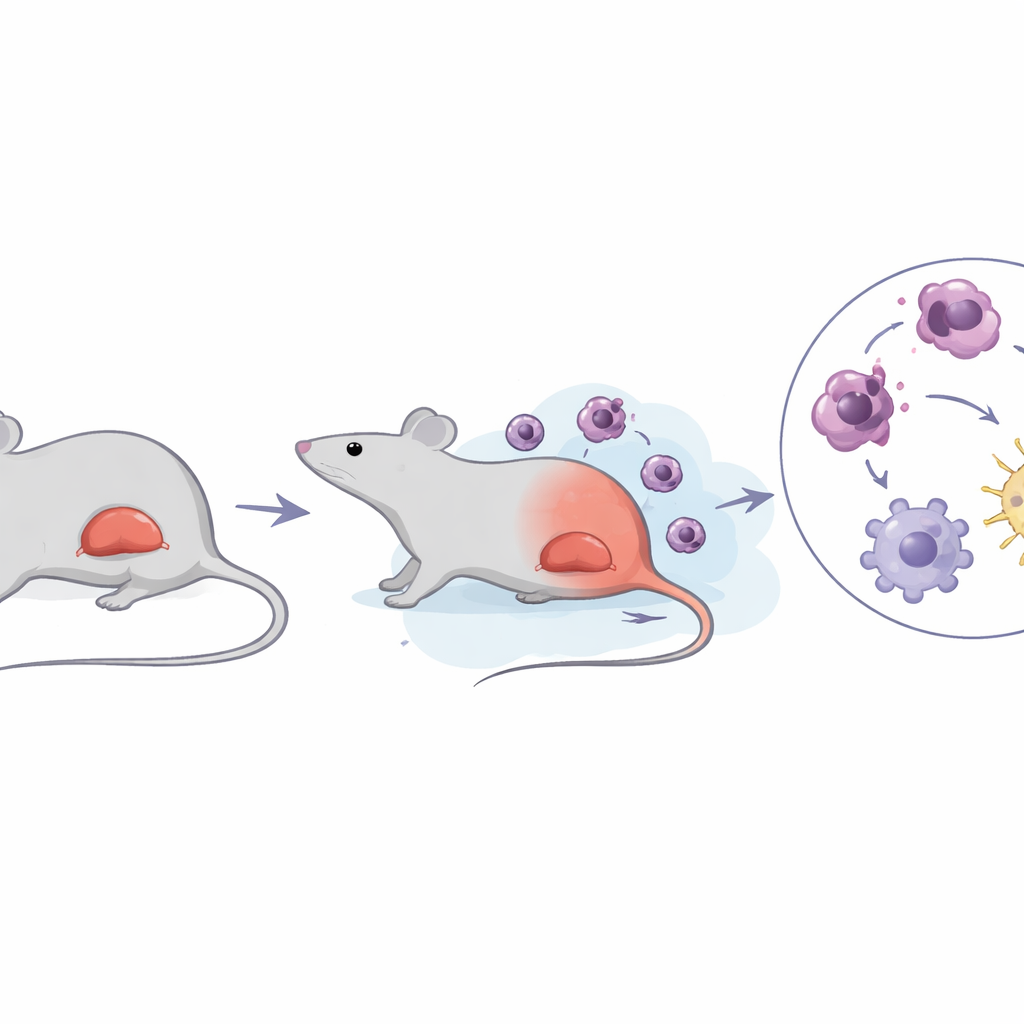
A dangerous swelling of the spleen
Acute erythroleukemia is an uncommon subtype of acute myeloid leukemia marked by explosive growth of immature red blood cell precursors and a very poor outlook, with average survival measured in months. Many patients develop splenomegaly, where the spleen balloons in size and becomes packed with leukemic cells. Using a well‑established mouse model infected with Friend murine leukemia virus, the authors carefully tracked disease progression. They confirmed that mice developed anemia, sky‑high white blood cell counts, and severely enlarged spleens, closely mirroring the human condition. Importantly, they found that spleen enlargement occurred first; only later did levels of lactate—a breakdown product of sugar metabolism—rise sharply within the spleen.
When sugar burning goes off balance
Cancer cells often favor a fast but inefficient way of burning sugar known as glycolysis, which churns out lactate even when oxygen is plentiful. Proteomic (large‑scale protein) analysis of diseased mouse spleens revealed that key enzymes controlling sugar use and production were out of balance. Enzymes that convert pyruvate into lactate were turned up, while those that recycle lactate back into glucose were dialed down. Transporters that pull lactate into cells were also more active. Together, these shifts created a metabolic trap: sugar flowed into the spleen, was rapidly funneled into lactate, and that lactate was then retained rather than cleared, leading to a highly lactate‑rich environment inside the swollen organ.
Immune cells pushed into an inflammatory gear
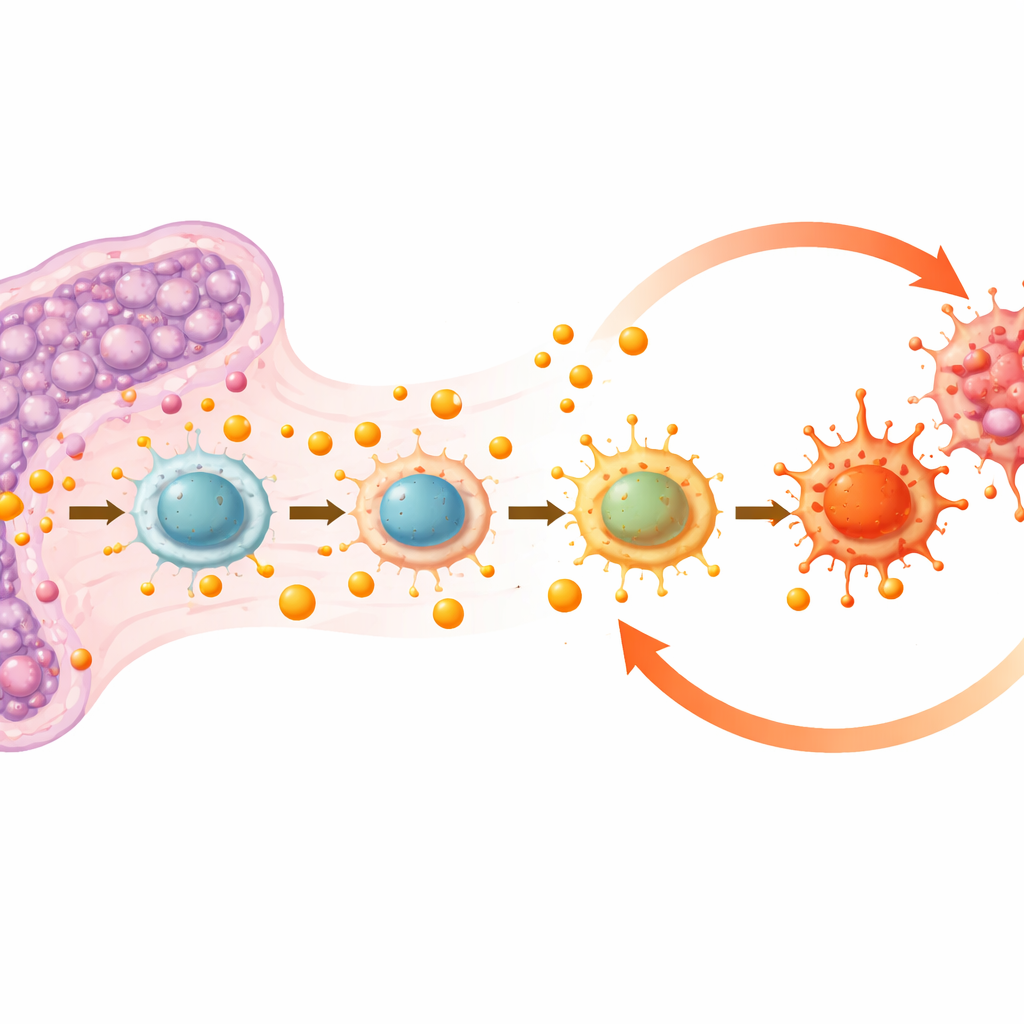
The spleen is not only a filter for blood cells; it is also a busy immune hub. The team discovered that high lactate levels were changing the behavior of immune cells, especially macrophages—versatile cells that can either fan the flames of inflammation (often called an M1‑like state) or help resolve damage and restore balance (an M2‑like state). In the leukemic spleens, overall macrophage numbers fell, but the remaining cells were strongly skewed toward the inflammatory M1‑like state. Proteins inside these macrophages showed increased “lactylation,” a chemical tag derived from lactate, suggesting that excess lactate was directly rewiring how these cells worked. At the same time, other immune cell types were thrown off balance, with fewer T cells and more myeloid cells, signaling a broader remodeling of the spleen’s immune landscape.
A self‑reinforcing loop driven by lactate
To probe cause and effect, the researchers turned to cell culture experiments. When they exposed mouse macrophages to the leukemia‑causing virus, the cells cranked up sugar use, pumped out lactate, and adopted an M1‑like inflammatory profile. Blocking two signaling switches inside the cells, called STAT1 and mTOR, prevented both the rise in lactate and the shift toward the inflammatory state. Strikingly, bathing macrophages in lactate alone was enough to push them into the M1‑like state and to make them generate even more lactate, creating a vicious cycle. Fluid taken from these lactate‑conditioned macrophages made human erythroleukemia cells grow faster, showing that the altered immune cells were actively helping the cancer.
Breaking the cycle to protect the spleen
Finally, the team asked whether interrupting lactate production in living animals could change the course of disease. They treated leukemic mice with oxamate, a drug that inhibits the enzyme that makes lactate. Compared with untreated animals, oxamate‑treated mice had smaller spleens and other organs, improved blood counts, less tissue damage, and lived longer. Their splenic macrophages shifted back toward a more balancing, M2‑like profile, and overall lactate levels in the spleen dropped. Giving extra lactate had the opposite effect, further enlarging the spleen. To a lay observer, the message is clear: in this leukemia model, lactate is not just a metabolic waste product—it is a powerful signal that locks immune cells into an inflammatory, tumor‑supporting mode and helps drive organ enlargement.
What this could mean for future care
This work reframes splenomegaly in acute erythroleukemia as an active process fueled by a feedback loop between cancer metabolism and the immune system. By showing that blocking lactate production can reverse harmful immune changes and ease spleen swelling in mice, the study points to a new class of “metabolic‑immune” therapies. While much more research is needed to test safety, timing, and combinations with standard leukemia drugs, targeting this lactate‑driven circuit may one day help protect patients from painful, dangerous spleen enlargement and slow the advance of this aggressive blood cancer.
Citation: Yang, M., Xie, D., Zhang, Y. et al. Lactate-mediated macrophage polarization promotes splenomegaly in acute erythroleukemia. Cell Death Dis 17, 373 (2026). https://doi.org/10.1038/s41419-026-08612-5
Keywords: acute erythroleukemia, lactate metabolism, macrophage polarization, splenomegaly, tumor microenvironment