Clear Sky Science · en
NAT10-mediated ac4C RNA acetylation stabilizes CXCL5/DEK mRNA to drive proliferation and metastasis in lung adenocarcinoma
Why this research matters for people with lung cancer
Lung adenocarcinoma is the most common form of lung cancer, and even with modern drugs many patients still face early spread of their disease. This study uncovers a hidden layer of control inside cancer cells: tiny chemical tags on RNA, the molecules that carry genetic messages. By showing how one enzyme helps lung tumors grow and travel through the body, the work points to a new kind of drug target that could one day slow or stop metastasis.
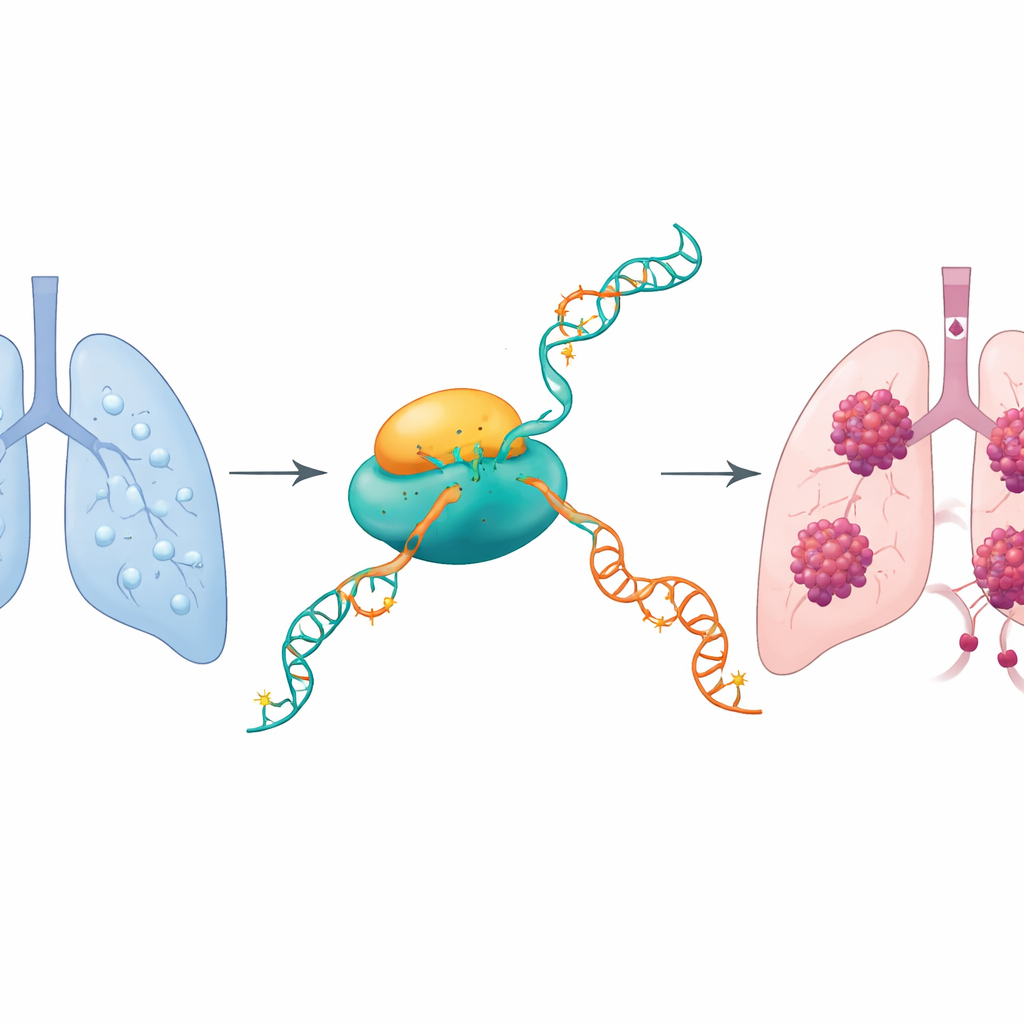
A chemical switch on the cell’s messages
Our cells constantly read DNA and copy it into RNA messages that tell the cell which proteins to make. The authors focused on a subtle chemical mark on RNA called N4-acetylcytidine (ac4C). Only one known enzyme, NAT10, can add this mark. Earlier studies in other cancers hinted that NAT10 makes tumor cells more aggressive. Here, the researchers asked whether NAT10 plays a similar role in lung adenocarcinoma and, if so, which specific RNA messages it alters to drive tumor growth and spread.
NAT10 is turned up in lung tumors
Using large public cancer databases and samples from patients, the team found that NAT10 levels are much higher in lung adenocarcinoma tissue than in nearby normal lung. Its activity rises as tumors progress to more advanced stages, and patients whose tumors have more NAT10 tend to have shorter survival. In both patient samples and lung cancer cell lines, overall ac4C marks on RNA were elevated alongside NAT10, suggesting that this enzyme broadly rewires how cancer cells handle their genetic messages.
Finding the key messages that NAT10 protects
To see what happens when NAT10 is removed, the scientists used CRISPR–Cas9 gene editing to knock out NAT10 in A549 lung cancer cells. They then combined two global approaches: RNA sequencing to measure which genes changed in activity, and acRIP-seq to map where ac4C marks appeared on RNA. Hundreds of genes lost both expression and ac4C marks after NAT10 was deleted, and many of these were linked to how cells stick to their surroundings and migrate—processes central to metastasis. Two standouts were CXCL5, a signaling factor that attracts and shapes immune cells, and DEK, a protein that helps organize DNA and influences tumor behavior in several cancers.
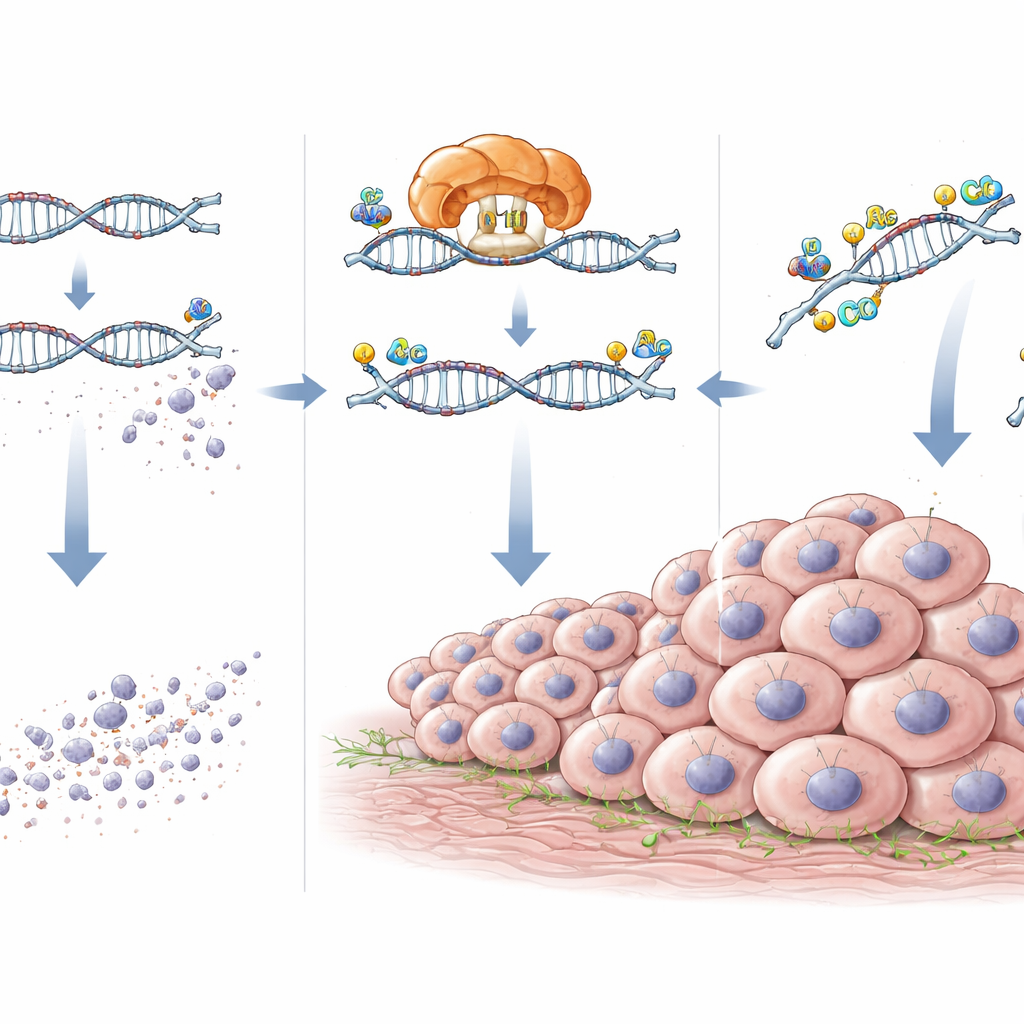
How NAT10 keeps tumor-promoting RNAs alive
Diving deeper, the team mapped specific ac4C sites on the RNA of CXCL5 and DEK and confirmed that they depended on NAT10. When NAT10 was knocked down, these chemical marks and the levels of CXCL5 and DEK RNA and protein all dropped. Reporter experiments, in which the ac4C sites were precisely mutated, showed that losing these sites made the RNAs less responsive to NAT10. Measurements of RNA decay revealed that, without NAT10, CXCL5 and DEK messages broke down more quickly. In other words, NAT10 acts like a protective coating, placing ac4C marks that stabilize these RNAs so they persist longer inside the cell.
From stronger grip to faster spread
What do these molecular tweaks mean for the behavior of cancer cells? In lab dishes, lung cancer cells lacking NAT10 grew more slowly, migrated and invaded less, and adhered less firmly to different components of the tissue scaffold, such as collagen and fibronectin. Their internal skeleton of actin fibers also became more compact, reflecting reduced motility. Re-introducing CXCL5 or DEK, and especially both together, partially restored the cells’ ability to proliferate, move, and stick, showing that these two NAT10-controlled genes are major drivers of the aggressive traits. In mice injected with lung cancer cells through the tail vein, NAT10-deficient cells formed far fewer metastatic spots in the lungs, and the animals gained more weight and carried a lower tumor burden than those injected with normal cancer cells.
What this means for future treatments
By piecing these results together, the authors propose a simple model: in lung adenocarcinoma, NAT10 adds ac4C marks to the RNA messages for CXCL5 and DEK, keeping them stable and highly expressed. Elevated CXCL5 and DEK then boost how tightly tumor cells adhere, how readily they grow, and how easily they spread to distant sites. Blocking NAT10 or its ability to place ac4C marks could therefore destabilize these pro-metastatic messages and weaken the tumor’s grip and growth. While more work is needed before this strategy can be tested in patients, the study highlights RNA acetylation—and NAT10 in particular—as a promising new handle for diagnosing, predicting, and ultimately treating lung adenocarcinoma.
Citation: Hu, X., Feng, M., Qi, C. et al. NAT10-mediated ac4C RNA acetylation stabilizes CXCL5/DEK mRNA to drive proliferation and metastasis in lung adenocarcinoma. Cell Death Dis 17, 326 (2026). https://doi.org/10.1038/s41419-026-08568-6
Keywords: lung adenocarcinoma, RNA acetylation, NAT10, metastasis, CXCL5 DEK