Clear Sky Science · en
Oncogenic GPRIN1 sustains proliferation and mitochondrial homeostasis via dual‑layer CDK1-PI3K/Akt signalling in gallbladder cancer
Why this research matters
Gallbladder cancer is rare but extremely deadly, often discovered too late for surgery or effective treatment. This study uncovers a previously unknown “master switch” protein, called GPRIN1, that helps gallbladder cancer cells multiply rapidly while keeping their tiny power plants—mitochondria—running smoothly. Understanding this switch could open the door to more precise, less toxic therapies for a cancer that currently has very few options.
A hidden driver in a lethal cancer
The researchers began by examining tumor samples from 100 patients with gallbladder cancer. They found that GPRIN1 levels were much higher in tumors than in nearby healthy tissue. Patients whose tumors showed the most GPRIN1 had more advanced disease and shorter survival, suggesting that this protein is closely linked to how aggressive the cancer becomes. In mouse models, tumors engineered to produce extra GPRIN1 grew faster, reinforcing the idea that this molecule is not a bystander but an active driver of tumor growth.
Connecting cell division to cellular power
Fast-growing cancer cells must simultaneously push through the cell cycle and secure enough energy to support their expansion. The team showed that GPRIN1 sits at the center of this coordination. When they reduced GPRIN1 in gallbladder cancer cell lines, the cells lost energy, produced less ATP, and accumulated harmful reactive oxygen molecules. Under the microscope, their mitochondria appeared swollen and damaged. At the same time, the normal cleanup system—mitophagy, which removes defective mitochondria—stalled. Key proteins that launch this cleanup, PINK1 and PARKIN, dropped, and damaged mitochondria piled up. In both cultured cells and mouse tumors, GPRIN1 proved essential for keeping mitochondria functional enough to fuel relentless growth.
The double control switch on cell‑cycle machinery
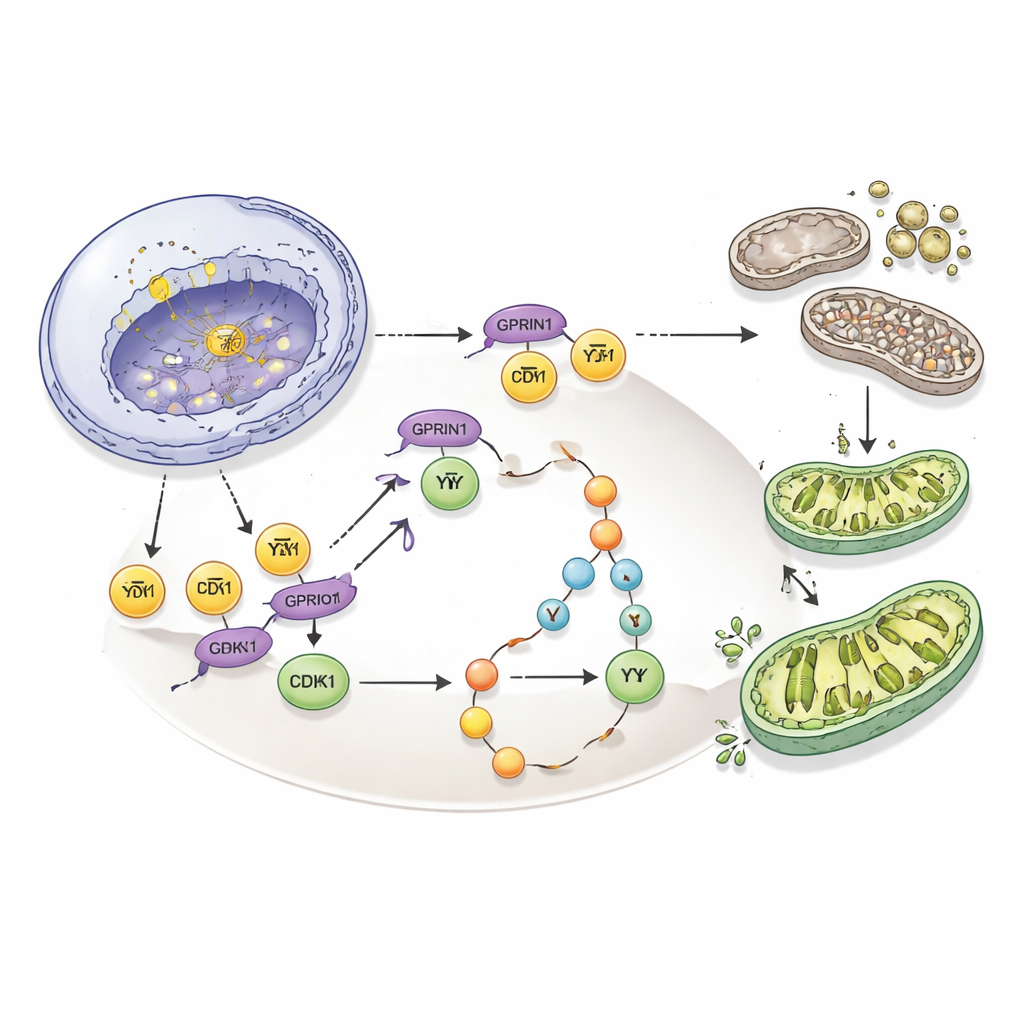
Diving deeper, the scientists used large-scale protein and phosphorylation screens to search for molecules that change when GPRIN1 is dialed down. One stood out: CDK1, a central engine that pushes cells through the final stages of division. GPRIN1 boosted both the amount and the activity of CDK1. In the cell nucleus, GPRIN1 physically bound to another protein, E2F1, and stabilized it, allowing E2F1 to more strongly turn on the CDK1 gene. Outside the nucleus, GPRIN1 reshaped the protein partners around CDK1, steering it away from its natural brakes and toward its activators. This “dual-layer” control—on the gene and on the protein—creates a hyperactive CDK1 engine that drives cells rapidly into division.
From molecular signals to mitochondrial balance
The story did not end with CDK1. The team discovered that this overactive CDK1 in turn switched on a well-known survival pathway, PI3K/Akt, which helps cancer cells resist stress and death. When CDK1 was blocked, GPRIN1 could no longer activate this pathway. And when the researchers used a PI3K inhibitor drug called Buparlisib, it wiped out GPRIN1’s advantages: cell division slowed, ATP production dropped back to normal, oxidative stress rose, and the mitophagy machinery was disrupted. In essence, GPRIN1 uses CDK1 to flip on PI3K/Akt, which then coordinates both rapid cell growth and careful maintenance of mitochondria, giving gallbladder cancer cells a powerful survival edge.
Turning a weakness into a treatment opportunity
Taken together, the findings reveal GPRIN1 as a central conductor that links how quickly gallbladder cancer cells divide with how well their mitochondria are maintained. By stabilizing E2F1, amplifying CDK1, and engaging PI3K/Akt, GPRIN1 builds a self-reinforcing loop that fuels aggressive tumor growth. Importantly, breaking this loop—either by reducing GPRIN1, silencing CDK1, or blocking PI3K—greatly slowed or even stopped tumor formation in cells and in mice. For patients, this suggests that measuring GPRIN1 levels could help predict prognosis, and that drugs aimed at this GPRIN1–CDK1–PI3K/Akt axis might provide a more targeted way to treat one of the most challenging cancers of the digestive system.
Citation: Xu, C., Gong, Z., Ni, X. et al. Oncogenic GPRIN1 sustains proliferation and mitochondrial homeostasis via dual‑layer CDK1-PI3K/Akt signalling in gallbladder cancer. Cell Death Dis 17, 333 (2026). https://doi.org/10.1038/s41419-026-08550-2
Keywords: gallbladder cancer, GPRIN1, CDK1, mitochondrial homeostasis, PI3K Akt signaling