Clear Sky Science · en
Loss of PTH 1 receptor signaling in periodontal cells drives cementum dysfunction and molar ankylosis in mice
Why tooth support matters beyond cavities
Most of us think of dental health in terms of cavities and gum disease, but another hidden structure quietly keeps every tooth in place: a thin, living cushion that separates the tooth root from the surrounding jawbone. When this support system fails, teeth can literally fuse to the bone, becoming immobile and very difficult to treat. This study in mice uncovers how a single hormone receptor in periodontal cells acts as a safeguard against this kind of damaging root‑to‑bone fusion.
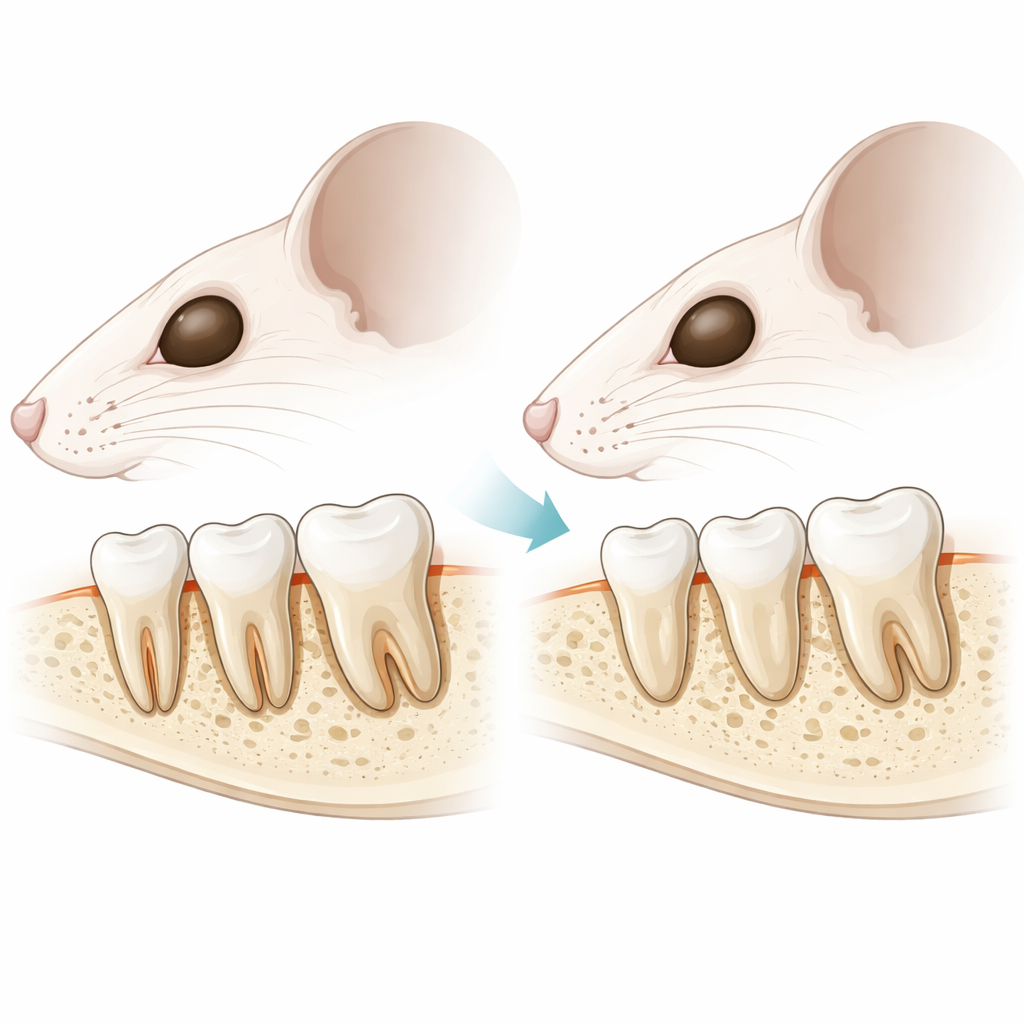
The quiet tissues that hold teeth in place
Each tooth is anchored in a socket by a small ecosystem of tissues called the periodontium. It includes the jawbone that surrounds the root, a thin mineral coating on the root called cementum, and a soft, fibrous layer in between known as the periodontal ligament. This ligament acts like a microscopic suspension bridge, allowing teeth to withstand chewing forces while remaining slightly flexible. The researchers focused on a hormone receptor, PTH1R, already known to be important for bone health and tooth eruption in childhood, and asked what it does later in life inside the mature cells that live in cementum and jawbone around the roots.
A targeted genetic change with surprising jaw effects
To explore this, the team engineered mice in which PTH1R was removed only from specific mineral‑embedded cells in the jawbone and cementum, using a genetic tool called DMP1‑Cre. Advanced 3D micro‑CT scans showed that these animals developed shorter upper jaws and thinner bone under the molars compared with normal littermates. Their molar roots were also shorter, and the bone that normally rises around them was reduced in height. Importantly, however, the teeth did erupt into the mouth and the front teeth (incisors) looked normal, allowing the scientists to study how this late loss of PTH1R affects an already formed periodontal system rather than early tooth development.
When the tooth cushion disappears
Detailed microscopic analysis revealed a dramatic breakdown of the ligament space in the molar region. In healthy mice, the tooth root and the jawbone were clearly separated by an organized band of fibers rich in structural proteins such as Periostin, Decorin, and collagen. In the mutants, that gap was often replaced by solid mineralized tissue directly connecting root and bone—a condition known as ankylosis. The fibrous matrix was fragmented or missing, its collagen fibers disorganized, and key ligament proteins sharply reduced. In many areas, the remaining ligament appeared pushed away from the root surface, suggesting that something growing out from the root itself was filling in the space where the ligament should be.
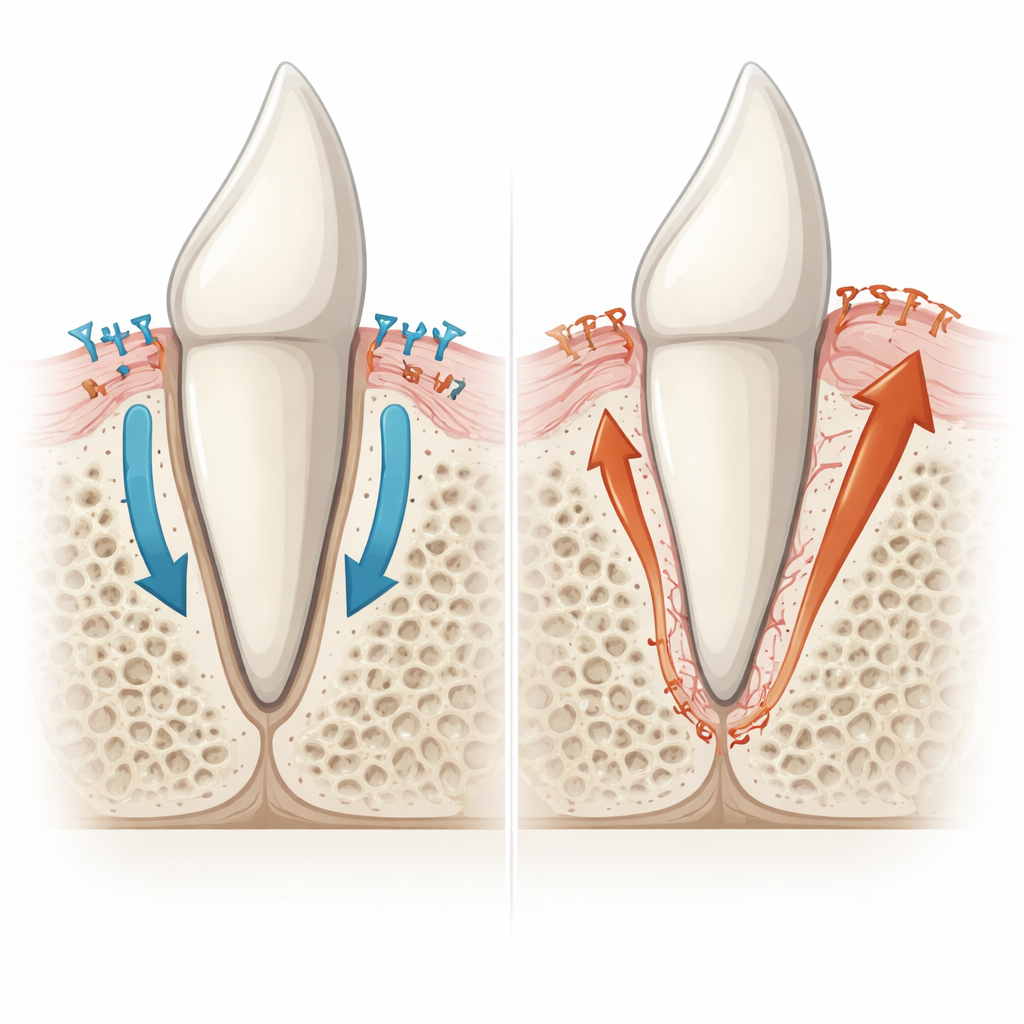
Root coating gone rogue
Closer inspection showed that the culprit was not overgrown jawbone, but rather an abnormal expansion of the root’s own mineral coating, cementum. Normally, a thin, acellular layer of cementum covers much of the root, providing a clean surface for ligament fibers to attach. Beneath the tip of the root, a thicker, cellular type of cementum forms, containing embedded living cells. In the mutant mice, regions that should have had only the thin acellular layer instead accumulated thicker, cellular‑like cementum that bridged straight to the bone. The tiny spaces housing cells in this tissue looked more like cementum than bone, and it failed to attract the bone‑eating cells that typically remodel jawbone. Molecular markers confirmed that these fused regions bore the signature of cementum, not bone, pointing to overactive cementum‑forming cells as the main driver of ankylosis.
Signals that tip the balance toward fusion
To understand what pushed cementum into overdrive, the researchers examined signaling pathways inside periodontal cells. Without PTH1R, they found more cells showing activated Smad3, a protein involved in transforming growth factor (TGF‑β) signaling, and higher levels of Osterix, a key controller of bone‑ and cementum‑forming cells. Markers of mineral production, including alkaline phosphatase and DMP1, were increased throughout the remaining ligament, indicating that cells there were behaving more like cementum builders than supportive ligament cells. At the same time, the team observed increased amounts of Dkk1, a natural brake on the Wnt pathway that normally helps keep mineralization in check, and a decline in S100A4 and other molecules that restrain hard‑tissue formation. Together, these changes created a strong push toward extra cementum and away from a soft, flexible ligament space.
What this means for future dental care
In simple terms, this work shows that the PTH1R receptor in mature periodontal cells acts like a safety valve, preventing the root’s mineral coating from growing unchecked and sealing the tooth to the bone. When that valve is removed in mice, signaling pathways that encourage hard‑tissue formation become overactive, and those that support a healthy ligament are dampened. The result is excessive cementum growth, loss of the tooth’s shock‑absorbing attachment, and eventual fusion of the tooth to the jaw. By clarifying how this balance is normally maintained—and how it can fail—these findings open the door to future strategies that might preserve or restore the ligament space, offering new ways to prevent or treat ankylosis and related craniofacial problems in humans.
Citation: Turkkahraman, H., Walton, C.J., Zhu, T. et al. Loss of PTH 1 receptor signaling in periodontal cells drives cementum dysfunction and molar ankylosis in mice. Bone Res 14, 46 (2026). https://doi.org/10.1038/s41413-026-00533-5
Keywords: tooth ankylosis, cementum, periodontal ligament, PTH1R signaling, craniofacial bone