Clear Sky Science · en
Molecular variants, clonal evolution and clinical relevance in pediatric and adult T-cell lymphoblastic neoplasia
Why these blood cancers matter to families
T-cell leukemias and lymphomas are aggressive cancers that mostly strike children, teenagers, and young adults. Despite modern treatments, many patients who relapse still face poor odds of survival. This study asks a deceptively simple question with big practical consequences: are the T-cell leukemia found in the blood and bone marrow and the closely related lymphoma found in lymph nodes really the same disease, or do they behave differently at the genetic level—and can that knowledge help doctors better predict who will relapse?
Two related cancers under the microscope
The researchers examined tumor samples from 211 children and adults with either T-cell acute lymphoblastic leukemia (T-ALL) or T-cell lymphoblastic lymphoma (T-LBL). Both cancers arise from very early T cells in the thymus but are usually classified by where the cancer cells show up: mainly in the bone marrow and blood for leukemia, and mainly in lymph nodes or other tissues for lymphoma. Using modern DNA sequencing and genome-wide scans, the team cataloged different types of genetic damage—small DNA changes and larger gains or losses of chromosome pieces—and compared patterns between age groups and between the two diseases.
Shared roots with age-related twists
Overall, T-ALL and T-LBL turned out to share a very similar set of “driver” genes—genes that, when damaged, help launch or sustain cancer. One gene in particular, NOTCH1, was altered in the majority of patients in all groups and tended to be more common in younger patients. Another gene, PHF6, became more frequent with increasing age. While the total number of mutations per patient was similar across groups, larger chromosome changes accumulated with age in T-ALL but not in T-LBL. Certain chromosome gains and losses were linked to relapse in children with lymphoma, suggesting that even closely related cancers can age and evolve in different ways.
Early hits that shape cancer’s family tree
Looking more deeply, the team reconstructed how each tumor grew over time, building “family trees” of cancer cell clones. They found that large-scale changes on chromosome 9, especially deletions or loss of one parental copy, were often among the very first genetic hits in both leukemia and lymphoma. These early events frequently affected a stretch of chromosome 9 that includes important cell cycle control genes, essentially removing key brakes on cell growth. In many children, cells carrying this chromosome 9 damage later acquired NOTCH1 mutations, forming new branches of the cancer that could either expand or die out during treatment.
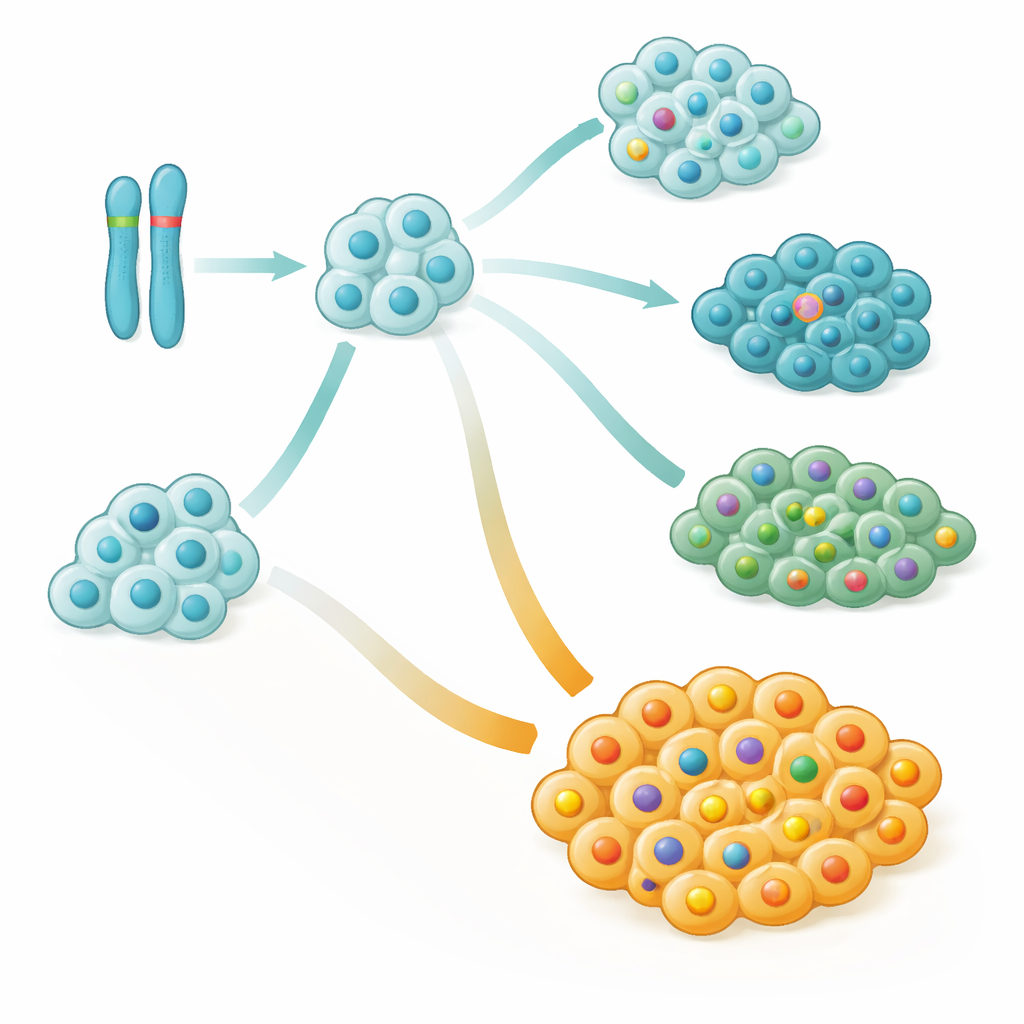
Good and bad branches inside the same tumor
One surprising insight was that NOTCH1 changes, long viewed simply as cancer-promoting, were actually linked to better outcomes when they appeared early and dominated the tumor. Patients tended to fare worse when a large fraction of their cancer cells lacked NOTCH1 mutations, even if some cells did carry them. In statistical terms, every small increase in the proportion of NOTCH1-normal cancer cells translated into a meaningful jump in relapse risk. Detailed case studies showed that aggressive relapses often arose from minor side branches of the cancer family tree that never acquired NOTCH1 changes but instead collected other damaging alterations.
What this means for patients and doctors
The study suggests that in these T-cell cancers, when and where a mutation appears within the tumor’s family tree matters as much as which gene is mutated. Early chromosome 9 damage seems to set the stage, while different branches—with or without NOTCH1 changes—compete and respond differently to therapy. For patients, this raises the possibility of more precise risk estimates: not just asking “is NOTCH1 mutated?” but “how much of the tumor is driven by NOTCH1-mutant cells versus NOTCH1-normal cells?” The authors argue that such clonal information, already promising in lymphoma, could be used to fine-tune risk groups and treatment intensity in T-cell leukemia as well, helping to better match therapies to each patient’s true relapse risk.
Citation: Sandmann, S., te Vrugt, M., Randau, G. et al. Molecular variants, clonal evolution and clinical relevance in pediatric and adult T-cell lymphoblastic neoplasia. Blood Cancer J. 16, 57 (2026). https://doi.org/10.1038/s41408-026-01488-w
Keywords: T-cell leukemia, T-cell lymphoma, clonal evolution, chromosome 9 alterations, NOTCH1 mutation