Clear Sky Science · en
ENO2 drives tumor cell-induced M2 macrophage polarization to promote colorectal cancer liver metastasis
Why this research matters
When colorectal cancer spreads to the liver, it becomes far harder to treat and is the main reason many patients die of the disease. This study digs into a basic question with big clinical stakes: how do tumor cells persuade the body’s own immune cells to switch sides and help cancer grow in the liver? By pinpointing a single tumor protein that drives this harmful alliance, the work opens the door to a new kind of targeted treatment.
A closer look at liver-spreading colon tumors
The researchers began by building a detailed “cell-by-cell map” of colorectal cancers that had spread to the liver, comparing them with tumors that had not metastasized. Using single-cell RNA sequencing, they analyzed thousands of individual cells from primary colon tumors, nearby normal tissue, and liver metastases. They found that cancer cells in liver deposits were more unstable genetically, divided more rapidly, and were better adapted to low-oxygen conditions than those in the original colon tumors. This confirmed that liver metastases are not just copies of the primary tumor, but contain a more aggressive, survival-hardened cancer cell population.
Finding a dangerous tumor cell subset
To understand which cancer cells are primed to spread, the team grouped tumor cells based on shared gene activity patterns. One particular gene module, linked to poor patient survival, stood out. Within it, a subset of tumor cells showed a strong “shape-shifting” program called epithelial-mesenchymal transition, which helps cells detach and travel. Among ten top candidate genes in this module, one enzyme, ENO2, was uniquely tied to worse outcomes. Patients whose tumors had higher ENO2 levels lived significantly shorter lives, and independent patient groups confirmed that ENO2 was elevated both in primary tumors and in liver metastases compared with normal tissue.
How tumors coach immune cells to help them
The next question was how ENO2-positive cancer cells influence their surroundings. Using computational tools that infer signaling between cell types, the researchers found that ENO2-rich tumor cells acted as major “broadcast hubs,” especially through a pathway involving a protein called MIF. They discovered that ENO2 physically binds to MIF inside cancer cells and shields it from being tagged for destruction, allowing MIF to accumulate. This, in turn, activates growth and survival pathways in tumor cells and sends strong signals to nearby macrophages—immune cells that can either attack tumors (M1-like) or, in their M2-like form, support tumor growth and dampen other immune responses. In co-culture experiments and animal models, ENO2-high cancer cells consistently pushed macrophages toward this tumor-helping M2 state.
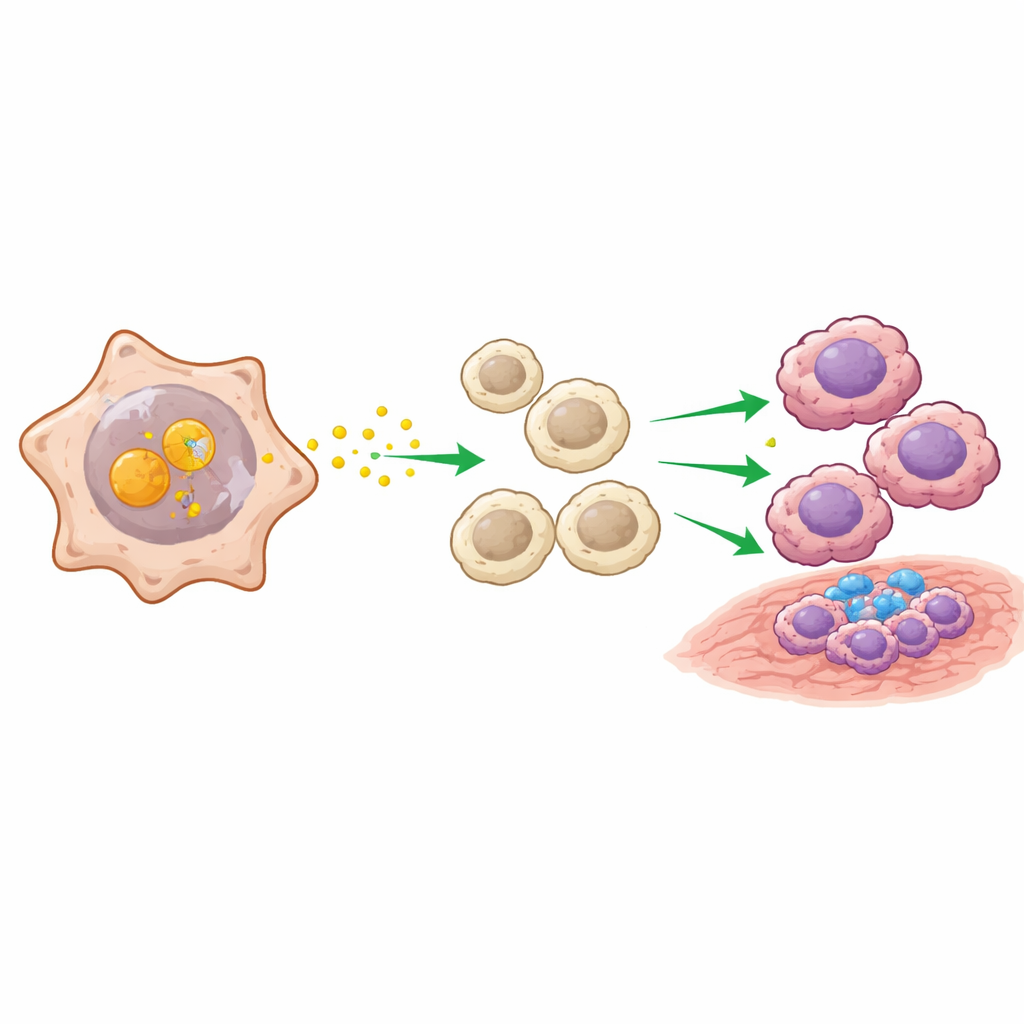
From mechanism to potential medicine
Armed with this mechanistic insight, the team searched for a drug that could break the ENO2–MIF partnership. Through computer-based screening of more than 6,000 small molecules, followed by laboratory tests, they identified pyrithioxin as a compound that wedges into the contact site between ENO2 and MIF. In cancer cells, pyrithioxin disrupted their interaction, restored MIF’s breakdown, and weakened downstream growth signals. In mouse models where colon cancer cells were injected to seed liver metastases, oral pyrithioxin treatment sharply reduced the number and size of liver tumors and lowered markers of M2-like macrophages in the metastatic sites.
What this means for patients
In simple terms, this study reveals that a single enzyme, ENO2, helps colon cancer cells “reprogram” nearby immune cells into a nurturing, tumor-promoting role in the liver by protecting the signal protein MIF from being destroyed. This creates a self-reinforcing loop of immune suppression and metastatic growth. By showing that blocking the ENO2–MIF interaction can shrink liver metastases in mice, the work highlights a promising new treatment strategy: instead of just attacking cancer cells directly, disrupt the hidden conversations that let them turn the immune system into an accomplice.
Citation: Tang, J., Chen, Z., Zhang, D. et al. ENO2 drives tumor cell-induced M2 macrophage polarization to promote colorectal cancer liver metastasis. Sig Transduct Target Ther 11, 166 (2026). https://doi.org/10.1038/s41392-026-02732-2
Keywords: colorectal cancer liver metastasis, tumor microenvironment, macrophage polarization, ENO2 MIF axis, cancer immunotherapy