Clear Sky Science · en
Immune modulatory vaccines targeting tumor microenvironment antigens: recent advances in oncology and beyond
Turning the Body’s Brakes into Cancer Targets
Cancer often survives not because our immune system is weak, but because tumors learn to hide behind the body’s own “brakes” that normally prevent harmful inflammation. This article explores a new type of treatment, immune modulatory vaccines (IMVs), that teaches the immune system to recognize and attack those brakes themselves. For readers, it offers a glimpse of how vaccines may soon be used not only to prevent infections, but also to rewire the body’s internal environment so it can better fight cancer and possibly stubborn infections.
How Tumors Build a Protective Neighborhood
Rather than growing as isolated lumps, tumors live inside a complex neighborhood of supporting cells, blood vessels, and immune cells called the tumor microenvironment. Many of these surrounding cells are “regulators” whose normal job is to calm the immune system after an infection and prevent autoimmunity. In cancer, however, they are hijacked. Specialized immune cells, scar‑forming tissue cells, and blood vessel cells begin to release molecules that starve immune cells of nutrients, signal them to stand down, or block their entry into the tumor. Together they form a multilayered shield that makes many cancers resistant to even modern drugs that release immune brakes, such as PD‑1 checkpoint inhibitors. 
Anti‑Regulatory T Cells: The Immune System’s Internal Watchdogs
The review highlights an unexpected discovery: our bodies naturally carry small numbers of T cells that specifically recognize these suppressive molecules and the cells that make them. These “anti‑regulatory T cells,” or anti‑Tregs, are not a special cell type but ordinary helper and killer T cells whose targets happen to be proteins like IDO, PD‑L1, arginase‑1, and TGF‑β—key players in immune dampening and tissue scarring. When activated, anti‑Tregs can directly kill suppressive cells or reprogram them by releasing inflammatory messengers. In effect, they police the regulators, preventing the immune system from being locked permanently in a “brakes‑on” state. Importantly, such cells are found even in healthy people, suggesting they are part of normal immune balance rather than a sign of disease.
Vaccines that Aim at the Cancer’s Shelter, Not Just the Cancer Cells
Traditional cancer vaccines try to train T cells to recognize markers on tumor cells themselves. IMVs take a different route: they contain short pieces of proteins from the suppressive molecules and support cells in the tumor neighborhood—collectively called tumor microenvironment antigens. After vaccination, anti‑Tregs expand and home in on any cell displaying these antigens, whether it is a cancer cell, an immune cell, or a fibroblast that helps build a stiff, exclusionary scar. Killer T cells remove these cells outright, while helper T cells flood the area with signals that boost antigen presentation, attract more fighters, and soften rigid tissue barriers. This two‑pronged action can turn a “cold,” poorly infiltrated tumor into a “hot,” inflamed one that is far more accessible to other immune therapies. 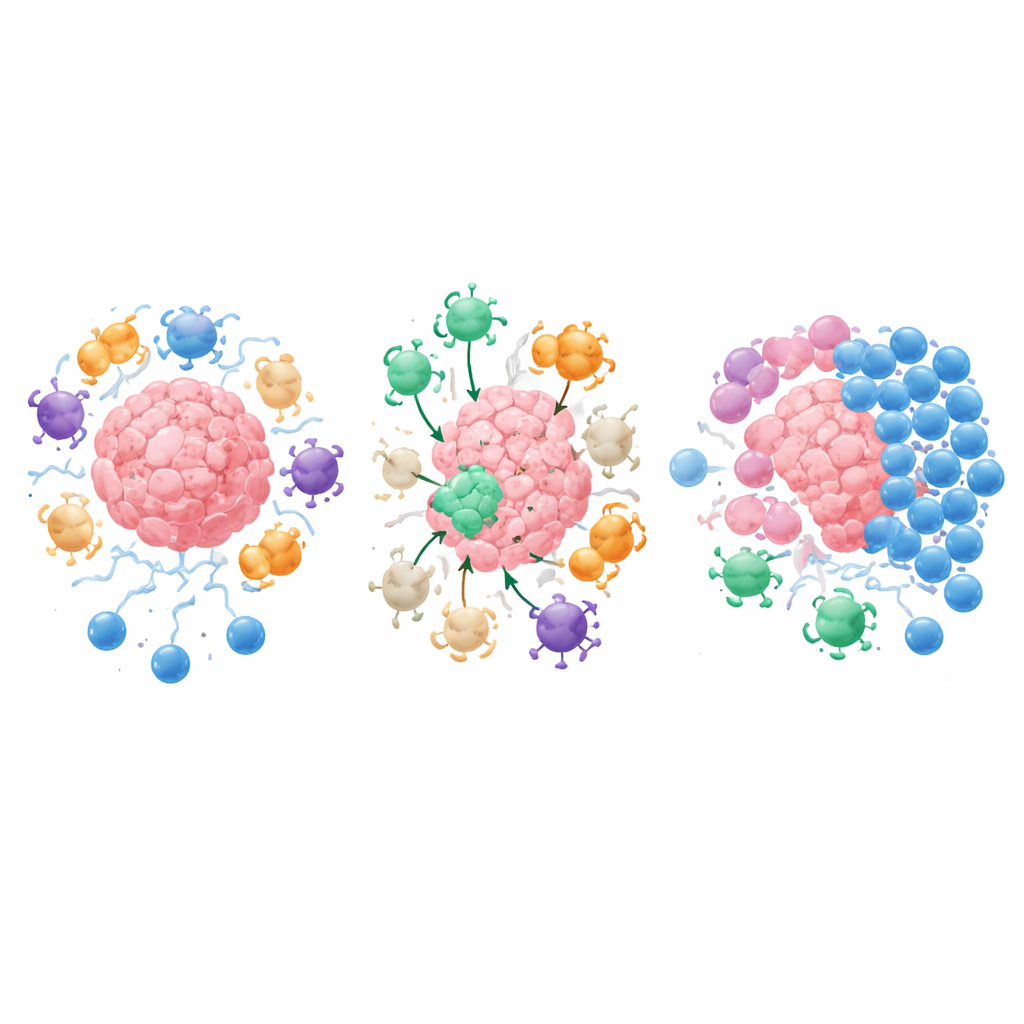
Evidence from Animal Studies and Early Clinical Trials
In multiple mouse models, vaccines targeting molecules such as IDO, PD‑L1, arginase, and TGF‑β slowed tumor growth, reduced immune‑suppressive cells, and increased cancer‑killing T cells inside tumors. These effects were even stronger when combined with PD‑1 checkpoint drugs. Early human trials largely echo these findings: peptide and mRNA‑based IMVs have proven safe and consistently trigger measurable T‑cell responses against their targets, often with reduced numbers or activity of suppressive cells in blood and tumor samples. A phase II melanoma study combining an IDO/PD‑L1 vaccine with anti‑PD‑1 therapy reported high response rates and long‑lasting control, and a larger phase III trial showed longer progression‑free survival overall, with the clearest benefit in patients receiving PD‑1 blockade for the first time and in tumors that were initially PD‑L1‑negative.
Looking Beyond Cancer to Infections and Future Uses
The same suppressive tricks used by tumors also appear in chronic infections and in severe acute infections in older or frail patients. The article argues that the IMV concept—vaccinating against the body’s own overactive brakes—could eventually help clear persistent viruses or boost responses to standard vaccines in the elderly by reshaping local immune environments. At the same time, it warns that care is needed in autoimmune diseases, where dialing down regulation too far might worsen self‑directed attacks. Overall, the conclusion is that IMVs add a new layer to immunotherapy: instead of only arming more soldiers, they dismantle the enemy’s fortress. Used in combination with existing drugs and moved earlier in the course of disease, they may help more patients’ own immune systems gain lasting control over cancer and other hard‑to‑treat illnesses.
Citation: Andersen, M.H. Immune modulatory vaccines targeting tumor microenvironment antigens: recent advances in oncology and beyond. Sig Transduct Target Ther 11, 130 (2026). https://doi.org/10.1038/s41392-026-02710-8
Keywords: tumor microenvironment, cancer vaccines, immune checkpoint, T cell therapy, immunotherapy