Clear Sky Science · en
TAK1 is a key regulator of oncogenic signaling and differentiation blockade in rhabdomyosarcoma
Why this childhood cancer study matters
Rhabdomyosarcoma is an aggressive childhood cancer that looks, under the microscope, a lot like immature skeletal muscle. These tumor cells carry the genetic tools needed to become normal muscle, but they stubbornly refuse to grow up. Instead, they keep dividing and spreading. This study uncovers a central molecular switch, a protein called TAK1, that helps keep these cancer cells in their dangerous, never‑ending growth mode. By turning TAK1 off, the researchers were able to slow tumor growth and nudge the cells toward becoming more normal muscle‑like cells.
The problem: frozen muscle cells in children
Rhabdomyosarcoma accounts for about half of all pediatric soft‑tissue sarcomas and a notable fraction of childhood cancers overall. It comes in two main forms, embryonal and alveolar, driven by different genetic changes. Despite these differences, both share a key feature: their cells resemble muscle precursors that are stuck in an early state. They express muscle‑building regulators but do not complete the journey to form mature muscle fibers. At the same time, they tap into multiple growth and survival pathways, making them difficult to treat with a single targeted drug. Clinicians and scientists have long suspected that there must be upstream control points that coordinate these growth signals and block differentiation, but those master switches have been hard to pinpoint.
A master switch for cancer signals
The team focused on TAK1, a signaling protein known to relay messages from growth factors and inflammatory molecules into several major pathways inside cells. By mining publicly available gene expression data from patient tumors, they found that TAK1 and several of its partner proteins are consistently elevated in rhabdomyosarcoma compared with normal muscle. They confirmed this in multiple human tumor‑derived cell lines, where both the amount of TAK1 and its active, phosphorylated form were higher than in normal human muscle cells. This suggested that TAK1 is not just present but switched on in these cancers, potentially acting as a hub that ties together several cancer‑promoting routes inside the cell.
Turning TAK1 down slows growth and spread
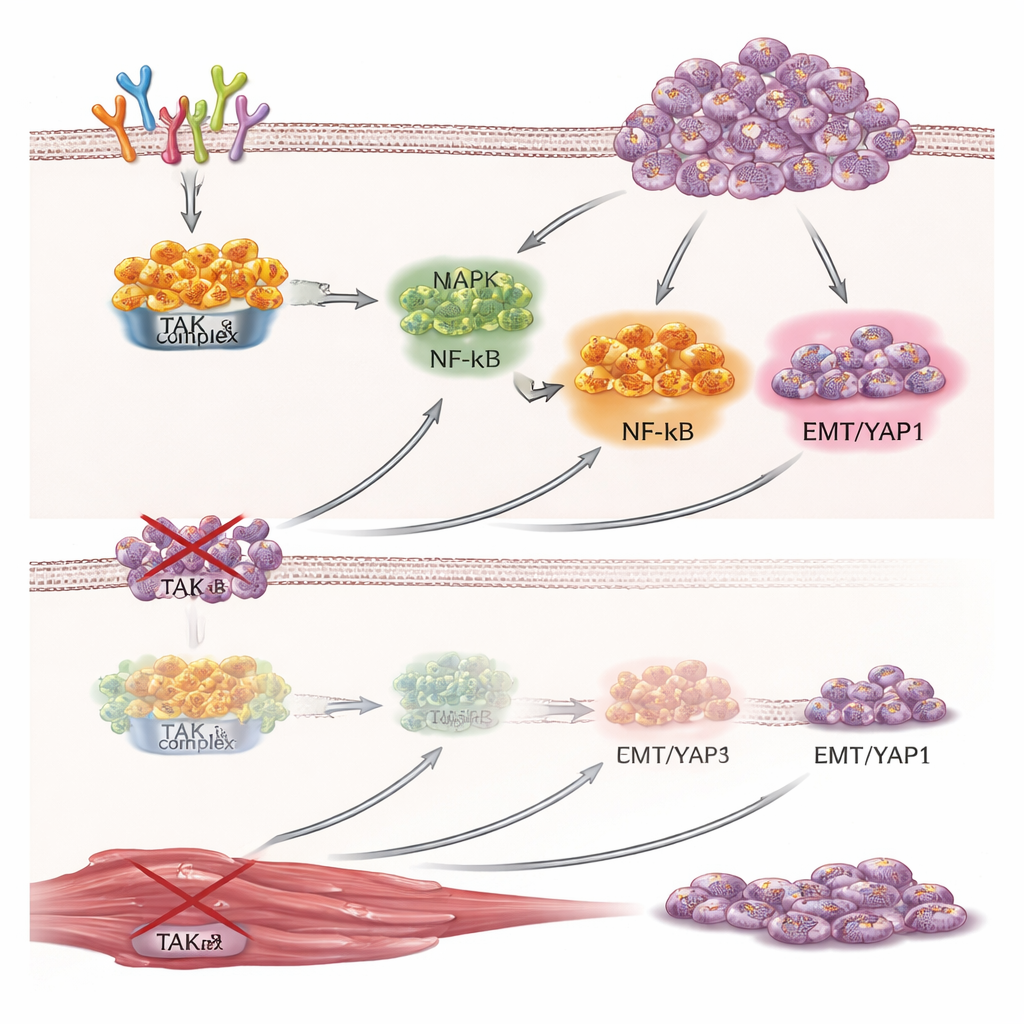
To test TAK1’s role, the researchers used two strategies to reduce its activity in rhabdomyosarcoma cells grown in the lab: genetic knockdown (using short hairpin RNA or small interfering RNA molecules) and a small‑molecule inhibitor called 5Z‑7‑oxozeaenol. Both approaches sharply reduced how fast the cancer cells divided, as measured by DNA synthesis, metabolic activity, and their ability to form colonies over time. The cells also became less able to migrate through pores and invade through a gel that mimics tissue, indicating that TAK1 helps drive behaviors associated with metastasis. Large‑scale RNA sequencing and protein array analyses showed that when TAK1 was silenced, many genes and proteins tied to the cell cycle, growth factor signaling, and epithelial–mesenchymal transition were dialed down, while programs involved in building muscle structure and differentiation were turned up.
Releasing the brake on muscle differentiation
One of the most striking effects of blocking TAK1 was a surge in markers of muscle maturation. Across several rhabdomyosarcoma cell lines, reducing TAK1 led to many more cells expressing myosin heavy chain and myogenin, hallmark proteins of late‑stage muscle cells. A reporter sensitive to an important muscle‑specific promoter also became more active, indicating that the underlying gene‑expression machinery was shifting toward a muscle program. Further mechanistic work traced part of this effect to the Hippo–YAP1 pathway, a well‑known regulator of organ size and cancer. TAK1 knockdown reduced levels of YAP1 protein. When the scientists reintroduced a form of YAP1 that resists degradation, it largely reversed the boost in muscle differentiation caused by TAK1 loss, implying that TAK1 normally helps stabilize YAP1 and, through it, keeps tumor cells in an undifferentiated, proliferative state.
Putting TAK1 to the test in living tumors
Laboratory dishes can only tell part of the story, so the team engineered rhabdomyosarcoma cells to carry a TAK1‑targeting switch that could be flipped off by feeding mice doxycycline. When these cells were implanted under the skin of mice, tumors formed in both groups, but those in doxycycline‑treated animals grew much more slowly and weighed less at the end of the study. Microscopic examination revealed fewer dividing cells and more multinucleated, muscle‑like cells in the TAK1‑silenced tumors. Protein analysis mirrored the cell culture findings: TAK1 levels dropped, YAP1 decreased, and the muscle differentiation marker myogenin rose, reinforcing the idea that TAK1 sustains tumor growth while actively blocking the cells’ built‑in tendency to mature.
What this could mean for future treatments
Overall, the study positions TAK1 as a central coordinator of cancer‑promoting signals in rhabdomyosarcoma and as a key enforcer of the “no‑maturation” rule that keeps these cells locked in a harmful, stem‑like state. By dialing down TAK1, the researchers simultaneously weakened several growth pathways, reduced invasive behavior, and allowed the cells to progress toward normal muscle differentiation, both in cell culture and in mouse tumors. For patients, this raises the possibility of therapies that do more than just kill rapidly dividing cells: drugs targeting TAK1 could also coax tumor cells to exit the cell cycle and become less malignant. The work is still preclinical, and issues such as drug delivery, safety, and resistance must be addressed, but TAK1 now stands out as a promising therapeutic target in this challenging childhood cancer.
Citation: Vuong, A.T., Joshi, A.S., Roy, A. et al. TAK1 is a key regulator of oncogenic signaling and differentiation blockade in rhabdomyosarcoma. Oncogene 45, 1714–1728 (2026). https://doi.org/10.1038/s41388-026-03767-z
Keywords: rhabdomyosarcoma, TAK1 kinase, childhood cancer, muscle differentiation, YAP1 signaling