Clear Sky Science · en
Microfluidic co-culture system for synaptically segregated neural networks to explore astrocyte-driven neural pathology
How brain support cells can quietly spread trouble
When brain cells fall sick, the damage rarely stays in one place. This study reveals how astrocytes, the star-shaped support cells of the brain, can silently carry harmful signals from one group of nerve cells to another. Using a tiny lab-on-a-chip, the researchers recreated a simplified brain circuit to watch how stress in one corner can ripple across to nearby regions that never see the original insult.
A tiny maze that mimics brain neighborhoods
The team built a palm-sized microfluidic device that acts like a miniature city for brain cells. Two separate chambers house distinct neuron populations, while a central lane is reserved for astrocytes. Between these areas lies a maze of narrow channels that lets astrocytes slip through but blocks the long nerve fibers of neurons. By carefully controlling fluid levels in connected reservoirs, the researchers could keep the chemical environment in each compartment isolated for set periods, ensuring that any communication between neuron groups had to travel through the astrocyte layer rather than by direct synaptic contact or shared liquid.
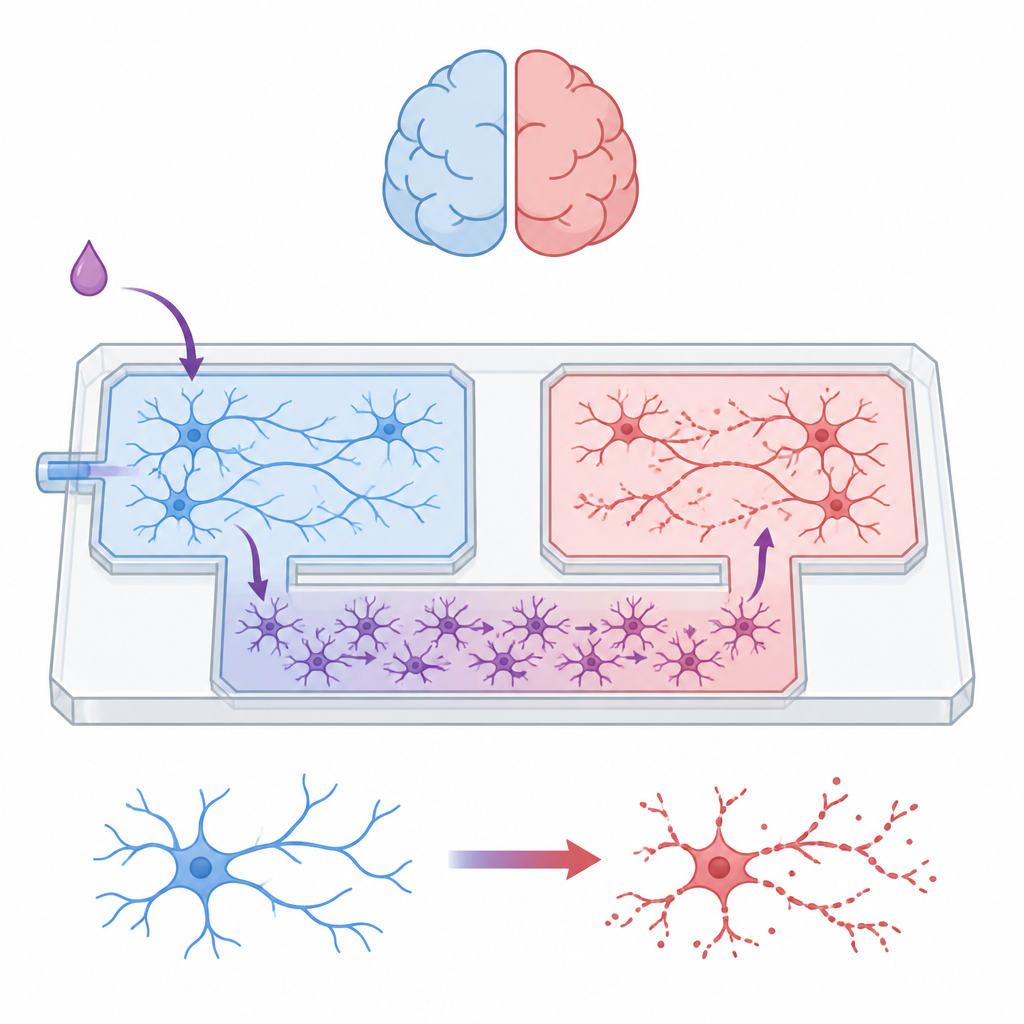
Letting helpers roam while fencing in neurons
Astrocytes grew happily throughout the device, forming a continuous carpet that stretched across the maze and into all chambers. Neurons, by contrast, remained confined. Tests showed that as their branches tried to cross the maze, their numbers dropped sharply at each barrier, and none reached the astrocyte-only lane. Protein profiling of the different regions confirmed this physical separation: the mixed neuron–astrocyte chambers were enriched in proteins linked to nerve growth, synapses, and electrical signaling, while the astrocyte-only area showed signatures of metabolism and immune-like functions typical of glial cells. Together these results showed that the device could host complex, mixed networks while keeping cell types and signals cleanly separated.
Watching toxic signals jump the gap
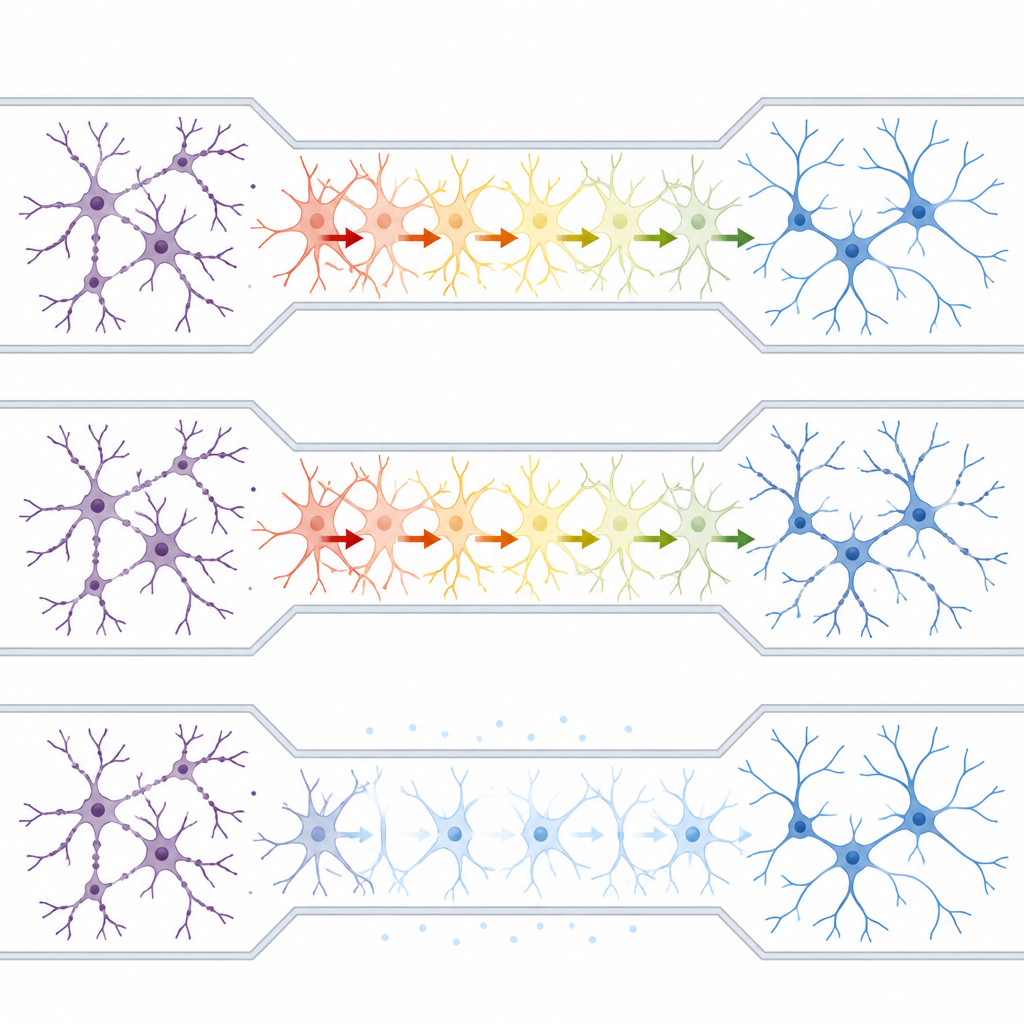
With this platform in place, the researchers asked whether astrocytes could carry “excitotoxic” stress between isolated neuron groups. They applied kainic acid, a seizure-inducing compound, to only one neuron–astrocyte chamber while maintaining fluidic isolation. Within 15 minutes, nerve branches in the treated side developed bead-like swellings, a hallmark of damage. Surprisingly, similar beading soon appeared in the untouched neuron group across the maze, even though no direct neuron-to-neuron connections or shared fluid existed between the chambers. When the same toxin was applied to neurons grown without astrocytes, the damage stayed local, indicating that astrocytes were essential for spreading the pathology.
Calcium waves in astrocytes as the hidden messenger
Astrocytes communicate using waves of calcium inside their cells. The team used a fluorescent calcium indicator to track these changes and found that toxin exposure on one side of the device triggered elevated calcium signals in astrocytes in the central lane. Blocking astrocyte calcium with a membrane-permeable chelator dampened these waves, reduced damage in the directly exposed neurons, and, importantly, prevented the spread of injury to the distant neuron group. Interestingly, completely silencing astrocyte calcium on its own harmed neurons, suggesting that normal calcium activity in these cells supports healthy network function, while excessive calcium helps drive disease-like processes.
Why this matters for brain disease and treatment testing
This work introduces a versatile brain-on-a-chip system that can separate, connect, and independently treat different brain cell populations while still allowing realistic contact between neurons and astrocytes. It shows that astrocytes can relay excitotoxic stress between neuron groups through a calcium-dependent mechanism, even when neurons themselves are cut off from one another. For non-specialists, the key message is that brain support cells are not passive bystanders: they can amplify and spread damage, but also offer a target for protective treatments. This platform provides a controlled way to probe those roles and to test future therapies aimed at calming harmful astrocyte signals in conditions such as epilepsy, stroke, and neurodegenerative disease.
Citation: Yap, Y.C., Musgrove, R.E., Breadmore, M.C. et al. Microfluidic co-culture system for synaptically segregated neural networks to explore astrocyte-driven neural pathology. Microsyst Nanoeng 12, 181 (2026). https://doi.org/10.1038/s41378-026-01187-3
Keywords: astrocytes, microfluidic brain chip, excitotoxicity, neuron glia interactions, calcium signaling