Clear Sky Science · en
DNA framework-based nanomedicine platform: a triple-function strategy for treating periodontitis via antibacterial, anti-inflammatory, and osteogenesis-promoting activities
Why Your Gums Matter to Your Whole Body
Bleeding gums and loose teeth are often seen as minor annoyances, but advanced gum disease, or periodontitis, is closely linked to heart disease, diabetes, and even brain disorders. This study explores a new kind of “smart medicine” built from DNA that can slip into damaged gum tissue, wipe out harmful bacteria, calm runaway inflammation, and help the jawbone rebuild itself. By tackling all three problems at once—germs, swelling, and bone loss—the work points toward shorter, more effective treatments for a disease that currently affects hundreds of millions of people worldwide.
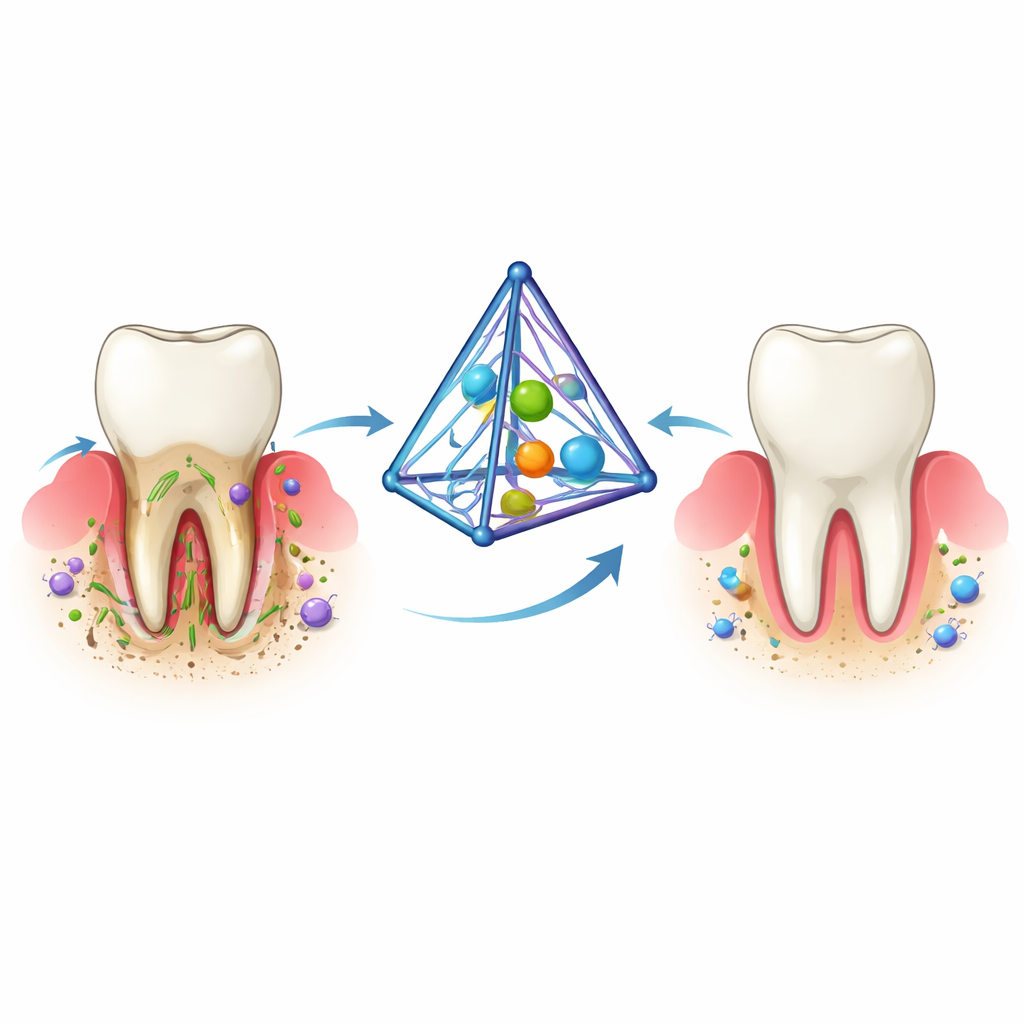
The Problem With Stubborn Gum Disease
Periodontitis starts when sticky bacterial plaque collects under the gumline. The body responds with inflammation meant to defend against infection, but over time this defense backfires. Chemical signals and reactive oxygen molecules build up, collagen fibers that hold the tooth in place break down, and the bone around the teeth slowly dissolves. Today’s treatments—deep cleaning under the gums and repeated local drug applications—can reduce bacteria and inflammation but rarely fix all three issues in one go: infection, ongoing inflammation, and lost bone. As a result, many patients need long-term care and may still lose teeth.
Building a Tiny DNA Delivery Vehicle
The researchers turned to a special pyramid-shaped DNA structure called a tetrahedral framework nucleic acid. This tiny framework, only a few billionths of a meter across, can be precisely built from strands of DNA and then decorated with useful cargo. In this work, the team attached an antimicrobial peptide called defensin to the DNA corners and then tucked curcumin—an anti-inflammatory and bone-supporting compound from turmeric—into the grooves of the DNA. The resulting complex, named Cur-de-tFNA, is small, stable in body-like fluids, easy for cells and even bacteria to take up, and breaks down into natural DNA components without leaving harmful residues.
Fighting Germs and Calming the Fire
In lab tests against several key gum-disease bacteria, the new complex proved far more powerful than defensin or curcumin alone. Under the microscope, bacteria exposed to Cur-de-tFNA showed ruptured walls and damaged interiors, and their growth was almost completely halted. In human periodontal ligament stem cells exposed to bacterial toxins, the complex sharply reduced levels of reactive oxygen species and lowered the production of aggressive inflammatory signals. It also dialed down a central alarm pathway inside cells that normally amplifies inflammation. At the same time, it boosted protective signals that are known to help maintain bone and guide stem cells toward repair.
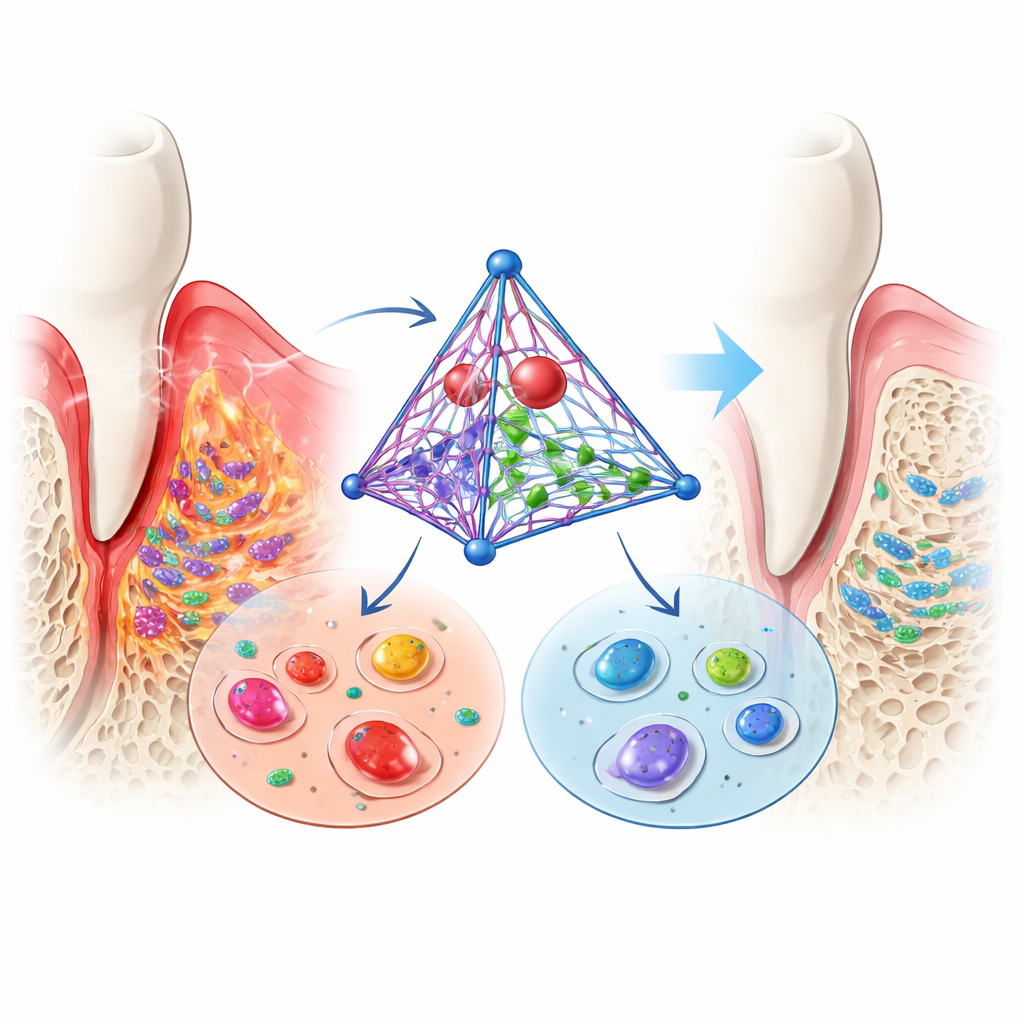
Helping Stem Cells Rebuild Lost Bone
The ligament cells that anchor teeth can, under the right conditions, mature into bone-forming cells. In an inflamed setting that mimics periodontitis, this ability is usually suppressed: bone-building genes quiet down, and mineral deposits are scarce. When the researchers added their DNA complex, the situation reversed. Markers of early and late bone formation rebounded or even exceeded levels seen in healthy control cells, and the cultures produced more mineralized nodules. These findings suggest that the platform not only shields cells from damage but also nudges them toward rebuilding the bone and connective tissue that support teeth.
Proof of Concept in Diseased Gums
To see if the strategy works in living organisms, the team tested the complex in rats with ligature-induced periodontitis, a standard model of severe gum disease. Animals that received local injections of Cur-de-tFNA around the affected teeth lost far less supporting bone—about half the resorption seen in untreated animals—and showed denser, thicker bone under 3D imaging. Tissue sections revealed fewer bone-resorbing cells, more organized collagen fibers, and reduced inflammatory cell infiltration. Blood markers of systemic inflammation also dropped, and the treated rats gained weight more like healthy controls, hinting at better chewing comfort and overall health.
What This Could Mean for Future Dental Care
Overall, the study introduces a DNA-based nanomedicine that can, in a single platform, kill disease-causing bacteria, quiet harmful inflammation, and encourage new bone to form around teeth. Although more work is needed to confirm long-term safety and to adapt the approach for human use, this triple-function strategy offers a glimpse of future gum treatments that are more targeted, longer lasting, and less reliant on broad-spectrum antibiotics—potentially protecting not just our smiles but our whole-body health.
Citation: Zhang, G., Cui, W., Wu, H. et al. DNA framework-based nanomedicine platform: a triple-function strategy for treating periodontitis via antibacterial, anti-inflammatory, and osteogenesis-promoting activities. Int J Oral Sci 18, 39 (2026). https://doi.org/10.1038/s41368-026-00439-2
Keywords: periodontitis, nanomedicine, DNA nanostructure, curcumin, bone regeneration