Clear Sky Science · en
Local delivery of OSK factors enables partial cellular reprogramming to mitigate osteoarthritis and cartilage fibrosis
Why this matters for aching joints
Osteoarthritis of the knee is a leading cause of pain and disability, yet today’s treatments mostly mask symptoms rather than restore damaged cartilage. This study explores a bold idea from regenerative medicine: gently “resetting” the aging joint’s cells with three biological switches called OSK factors, delivered directly into the knee. The work suggests it may be possible not only to slow joint damage, but also to reverse some of the scarring and stiffness that make everyday movement difficult.
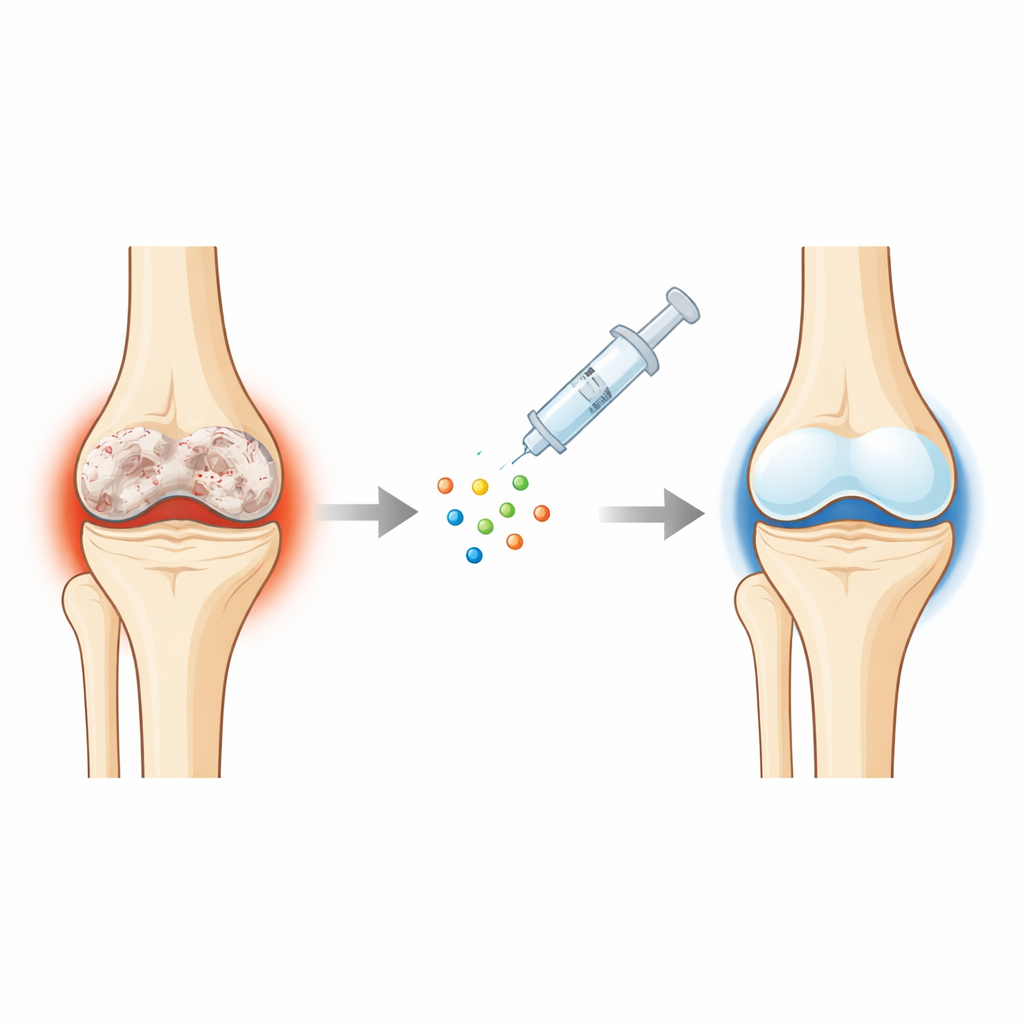
A closer look at worn-out cartilage
In osteoarthritis, the smooth, white cartilage that cushions the ends of bones gradually breaks down. The underlying bone thickens, the surface becomes rough, and fibrous scar-like tissue replaces the original springy layer. The cells that maintain cartilage, chondrocytes, are driven by age, stress, and inflammation into a harmful state: they inflame their surroundings, die off more easily, and start behaving more like bone-forming cells. Because cartilage has little blood supply and few repair cells, once this process starts it is very hard to reverse, leaving patients with pain, swelling, and loss of mobility.
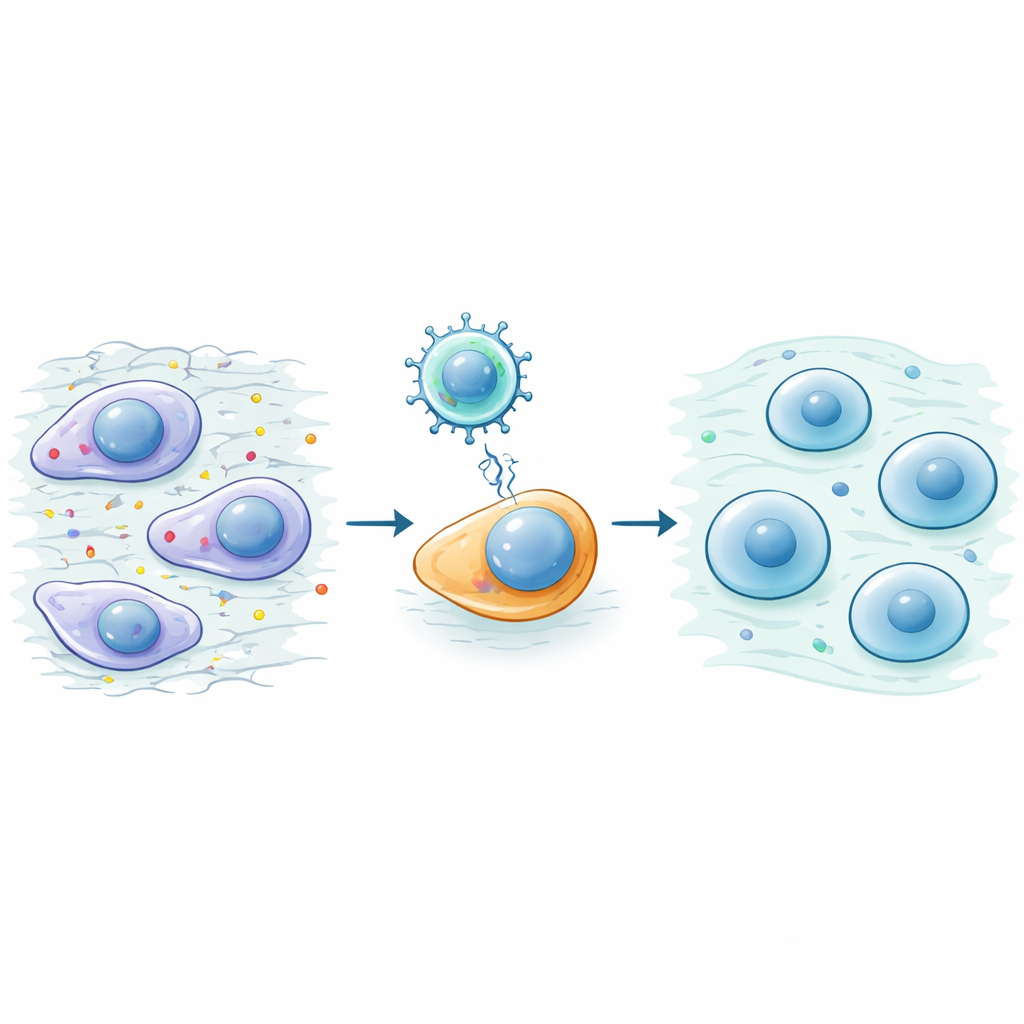
Gently turning back the cellular clock
Scientists have learned that certain gene switches can rewind a cell’s internal “age,” but pushing too hard can turn normal tissues into tumor-prone stem cells. To avoid this danger, the authors used only three factors—Oct4, Sox2 and Klf4, known together as OSK—omitting a fourth, riskier component. They packaged OSK into a harmless viral carrier and injected it directly into mouse knee joints. In lab dishes, cartilage cells exposed to OSK kept their normal identity while becoming more resilient: they produced fewer inflammatory molecules, resisted cell death, and were less likely to change into bone-like cells even in harsh, disease-like conditions.
Protecting and rebuilding damaged joints
The team then tested OSK in two standard mouse models of knee osteoarthritis created by surgical injury. Mice receiving the OSK treatment moved better, gripped more strongly, and showed less pain when their paws were touched. Scans of their knees revealed smoother joint surfaces and less abnormal bone thickening beneath the cartilage. Under the microscope, OSK-treated joints retained more of the original, glassy “hyaline” cartilage and fewer patches of stiff fibrocartilage. Helpful cartilage building blocks were preserved, while destructive and “overgrown” markers that signal breakdown and scarring were reduced. Notably, OSK also worked when given later in disease, nudging already formed fibrocartilage toward a more normal, hyaline-like state.
Resetting the epigenetic code of aging cartilage
Beyond structure and pain relief, the researchers asked whether OSK changes the deeper molecular signs of aging in joint tissue. They focused on DNA methylation, a chemical tagging system on DNA that acts like an epigenetic clock. In osteoarthritic cartilage, this clock runs fast: aging markers rise, and genes controlling inflammation and tissue quality are misregulated. By profiling methylation across the genome, the team found that OSK-treated cartilage looked “younger” than expected for its age and differed markedly from untreated diseased tissue. The therapy dampened expression of a DNA-methylating enzyme linked to cartilage aging and boosted another enzyme, TET2, which helps erase methylation marks. When TET2 was deliberately blocked, many of the protective effects of OSK vanished, highlighting this enzyme as a key mediator.
What this could mean for future care
Taken together, the findings suggest that carefully tuned delivery of OSK factors into the knee can partially reprogram aging, diseased cartilage cells without turning them into runaway stem cells. In mice, this approach eases pain, protects joint structure, and even converts scar-like fibrocartilage back toward its original, cushioning form—changes that align with a measurable “younger” epigenetic state. While much work remains to test safety, dosing, and long-term effects in larger animals and humans, this study offers an early glimpse of a future in which osteoarthritis treatment aims not just to soothe symptoms, but to rewrite the aging code of joint tissues themselves.
Citation: Liu, YW., Zou, JT., Gong, JS. et al. Local delivery of OSK factors enables partial cellular reprogramming to mitigate osteoarthritis and cartilage fibrosis. Exp Mol Med 58, 782–797 (2026). https://doi.org/10.1038/s12276-026-01662-x
Keywords: osteoarthritis, cartilage regeneration, cellular reprogramming, epigenetics, gene therapy