Clear Sky Science · en
Multiscale biomechanics-driven design pipelines for reconstructive bone scaffolds
Helping Broken Bones Heal Better
When a bone is badly damaged by an accident, disease, or surgery, the body sometimes cannot repair the gap on its own. Surgeons can now implant three-dimensional "scaffolds"—porous, sponge-like supports that fill the defect and encourage new bone to grow. This article explains how a new design approach uses principles of mechanics at multiple scales to create smarter scaffolds that not only hold the bone together, but also deliver nutrients and send the right physical signals to the cells that rebuild bone.
What Bone Scaffolds Are Meant to Do
Traditional bone grafts rely on transplanting pieces of a patient’s own bone, which can cause pain and is limited in supply. Engineered scaffolds offer an alternative: they are custom-shaped implants placed into the defect, where they temporarily play the role of the bone’s natural supporting framework. Their job is demanding. They must carry loads so the limb or jaw can function, allow blood and nutrients to reach cells deep inside, and provide subtle mechanical cues that tell stem cells to turn into bone-forming cells rather than scar tissue. Modern 3D printing and advanced materials make it possible to control pore size, shape, and orientation with great precision, opening the door to highly tailored implants.
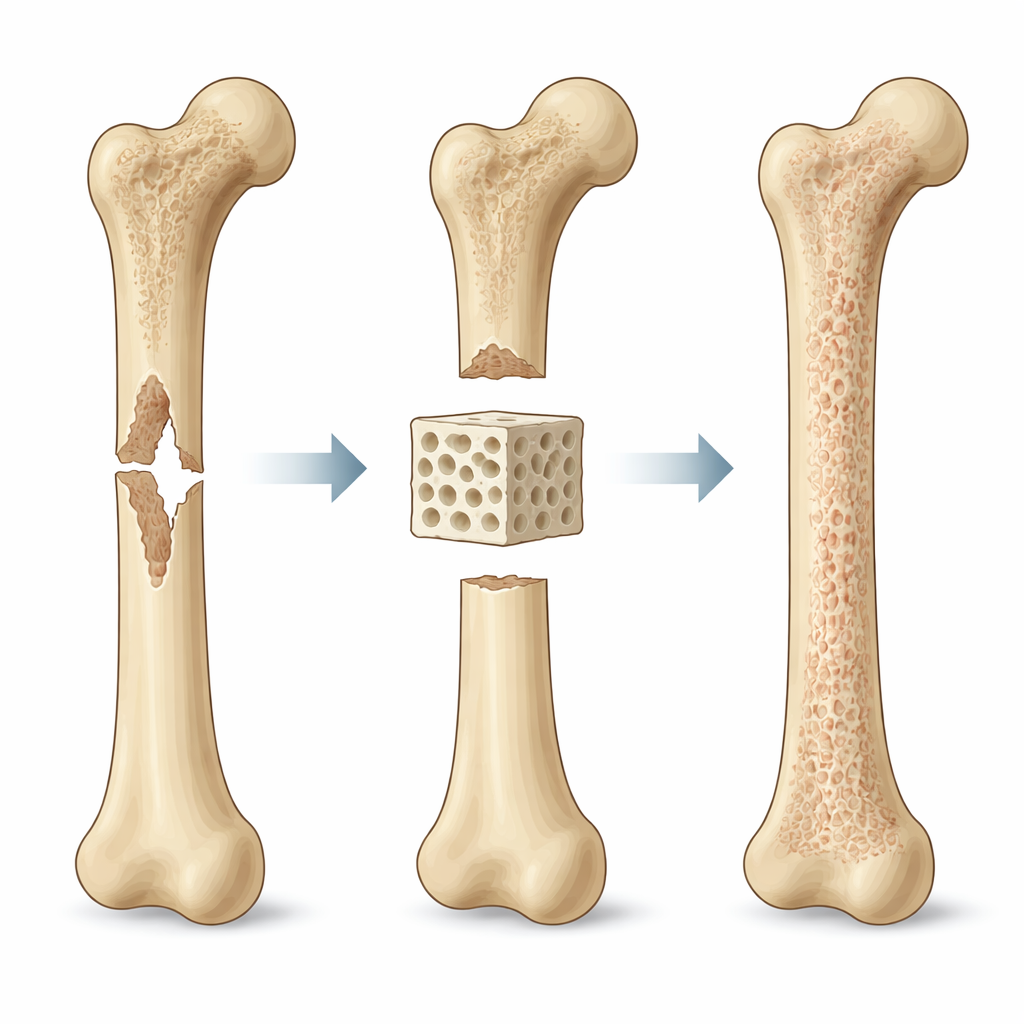
Looking at the Problem from Big to Small
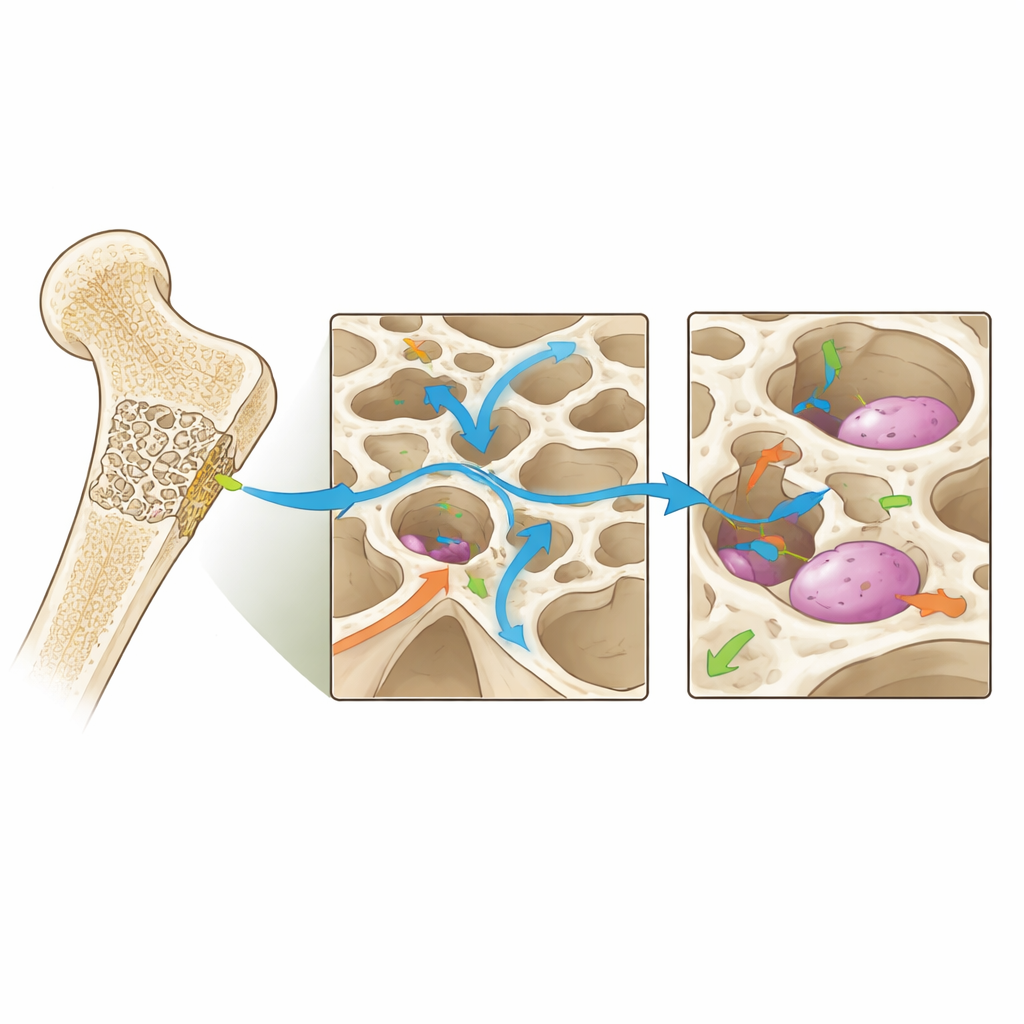
The authors propose that scaffold design must be guided by biomechanics at three linked scales. At the "macro" scale—the size of the whole bone segment—the scaffold has to bridge the defect and share forces with the surrounding bone. If it is too soft, tiny movements can loosen the implant and encourage soft tissue instead of bone. If it is too stiff, it can shield the natural bone from stress, slowing healing. At the "meso" scale—the level of pores and channels—its architecture governs how easily fluid, oxygen, and cells can move through the structure. Larger and more connected pores improve transport and blood vessel growth, but they also weaken the scaffold if overdone. At the "micro" scale—where individual cells feel their surroundings—local stretching, surface stiffness, and tiny flows influence how cells attach, change shape, and decide which tissue type to form.
Finding the Sweet Spot for Bone Growth
Experiments and computer models suggest that bone cells respond best when mechanical signals fall within certain "windows," rather than at a single perfect value. Too little strain or fluid movement tends to produce fibrous, scar-like tissue; too much can damage cells or disrupt early healing. In between lies an osteogenic window where bone formation is favored. The exact ranges depend on the stage of healing, the location in the body, and the patient’s condition, but the concept is robust: scaffolds should be designed so that, under everyday loads, most of their interior exposes cells to these favorable mechanical conditions while still ensuring adequate strength and fluid transport.
A Step-by-Step Design Pipeline
To turn these ideas into practice, the article outlines a four-stage design pipeline. First, clinicians and engineers translate patient scans, defect shape, and expected loads into clear goals and limits for the scaffold—such as target stiffness, allowable porosity, and desired bone and vessel growth. Second, computers generate many virtual scaffold designs by varying material, pore size, and internal patterns, then simulate how each one carries forces, moves fluid, and shapes the cell-scale environment. Third, the most promising candidates are built and tested in the lab and in animal models to check that real behavior matches predictions and that cells indeed form bone. Finally, multi-objective optimization techniques help choose designs that best balance conflicting needs, such as strength versus permeability, for specific clinical situations.
Looking Ahead to Smarter Implants
The review also explores future directions, including using artificial intelligence to search vast design spaces, "smart" materials that change stiffness or release factors in response to use, and digital twin models that track how an implanted scaffold and surrounding bone co-evolve over time in a given patient. Together, these advances point toward scaffolds that are not mere fillers but active partners in healing—structures tuned to keep the mechanical environment inside a bone defect within the sweet spot that encourages robust, long-lasting regeneration.
Citation: Hou, B., Yang, X., Li, Y. et al. Multiscale biomechanics-driven design pipelines for reconstructive bone scaffolds. npj Biol. Phys. Mech. 3, 5 (2026). https://doi.org/10.1038/s44341-026-00035-9
Keywords: bone scaffold design, biomechanics, tissue regeneration, 3D printed implants, mechanobiology