Clear Sky Science · en
Repurposing caspofungin as a small-molecule inhibitor of Clostridium perfringens α-toxin for treatment of gas gangrene
A Dangerous Infection in Need of Better Treatments
Gas gangrene is a fast-moving, often deadly infection that can follow serious injuries or sometimes appear without warning. It is caused by the bacterium Clostridium perfringens, which releases powerful toxins that shred muscle, cut off blood flow, and overwhelm the body in hours. Current treatments—urgent surgery, antibiotics, and sometimes oxygen therapy—can save lives, but they do not always stop the damage in time. This study explores whether an existing antifungal drug, caspofungin, could be repurposed to directly disarm the bacterium’s main toxin and offer a new, faster way to protect patients.
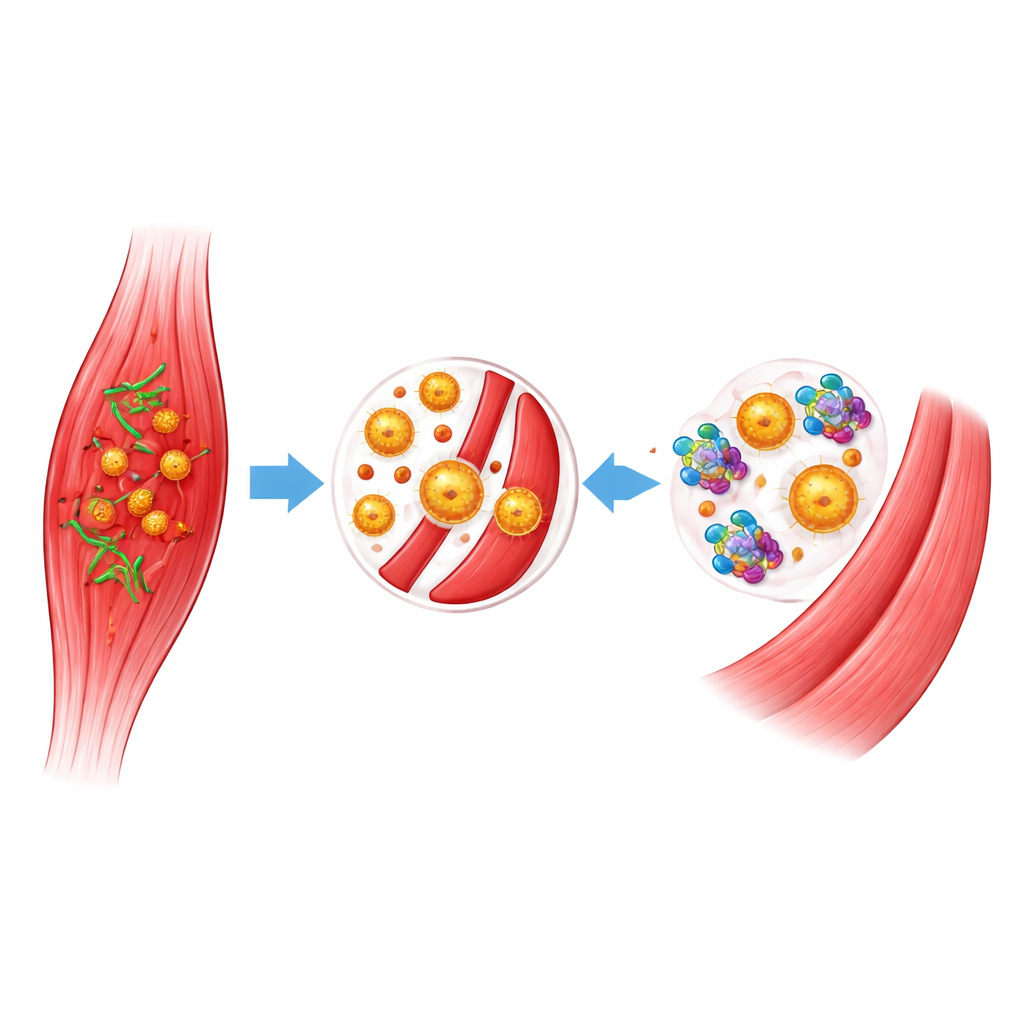
The Bacterial Toxin That Wrecks Muscle and Blood Flow
C. perfringens produces several toxins, but one called alpha-toxin plays a central role in gas gangrene. Alpha-toxin attacks key fats in cell membranes, killing muscle and blood vessel cells and helping the bacteria spread. It also disrupts the immune system by making white blood cells and platelets clump together, blocking tiny blood vessels and preventing infection-fighting cells from reaching the site. Over time, this chokehold on circulation causes severe muscle death, shock, and organ failure. Because alpha-toxin drives so much of this devastation, blocking its activity is an attractive strategy to slow or halt the disease.
Searching Old Medicines for a New Use
Instead of starting from scratch to invent a new drug, the researchers screened 764 medicines that are already approved for human use. They tested each one for its ability to block the enzyme activity of alpha-toxin, focusing on how well it could stop the toxin from cutting apart a key membrane fat. From this large library, 21 compounds substantially reduced the toxin’s activity. The team then ran a second, more demanding test using human blood vessel cells in culture. Only one drug, micafungin—an antifungal agent used in hospitals—protected these cells from toxin-induced death. A related antifungal, caspofungin, was then brought into the experiments because it has a similar structure and clinical profile.
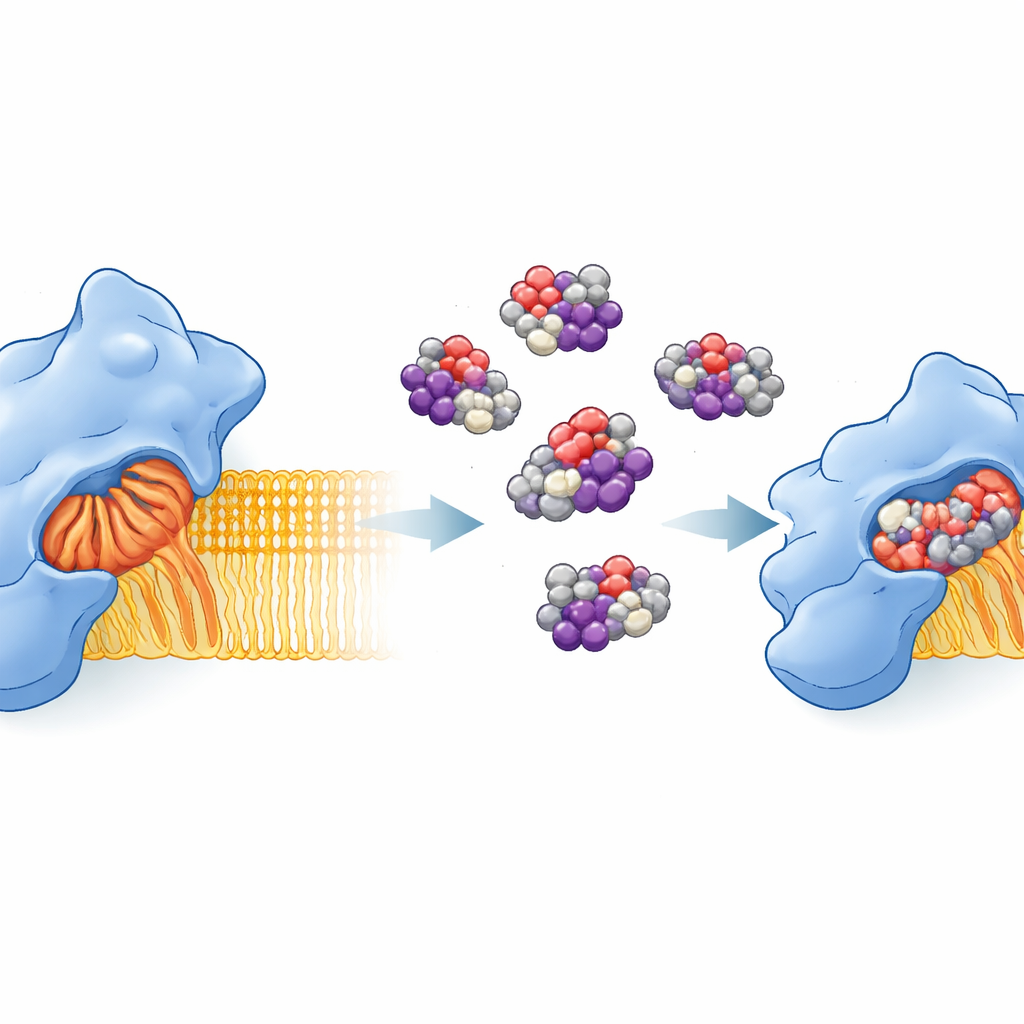
How Two Antifungal Drugs Grab Onto the Toxin
Both micafungin and caspofungin proved capable of blocking alpha-toxin’s ability to kill human blood vessel cells and to drive the release of inflammatory molecules. Computer modeling suggested why: the drugs appear to lodge in a cavity on the toxin where its natural fat-based target would normally bind. Micafungin’s rigid side chain fits deeply into this pocket, forming many tight contacts, while caspofungin’s more flexible side chain sits more shallowly and forms fewer strong bonds. These differences match laboratory measurements showing micafungin is the stronger blocker of one of the toxin’s activities. Yet caspofungin behaves differently on another arm of the enzyme’s function, hinting that subtle shifts in how each drug docks to the toxin can reshape its behavior.
From Cells to Mice: Testing Protection in Living Animals
Blocking a toxin in a dish is important, but the real test is whether this protection holds up in a living animal. When mice were given purified alpha-toxin, most untreated animals died within a day. Mice that received caspofungin along with the toxin, however, largely survived, showing that the drug can blunt the toxin’s lethal effects in the body. Surprisingly, micafungin, despite being a stronger blocker in test tubes, did not improve survival at the same dose. The authors suggest that small differences in how tightly these drugs bind to blood proteins—and thus how much free drug is available to neutralize the toxin—may explain this gap. In a second model, where mice were infected in the leg muscle with live C. perfringens, caspofungin significantly delayed death and reduced muscle damage. When combined with the standard antibiotic clindamycin, both survival and muscle preservation improved further.
What This Could Mean for Treating Gas Gangrene
The study concludes that caspofungin, a drug already in clinical use for serious fungal infections, has strong potential as a toxin-targeting therapy for gas gangrene. Rather than killing the bacteria directly, caspofungin disarms their chief weapon, giving standard antibiotics and surgery more time to work and helping protect muscle and blood vessels from catastrophic injury. Because caspofungin is already approved and widely available, it could, in principle, be moved into clinical testing for gas gangrene more quickly than a brand-new compound. More broadly, the work shows how scanning existing drug libraries can uncover small molecules that neutralize bacterial toxins, opening the door to a new class of treatments that focus on blocking the damage rather than just attacking the microbes.
Citation: Takehara, M., Homma, Y., Ishihara, T. et al. Repurposing caspofungin as a small-molecule inhibitor of Clostridium perfringens α-toxin for treatment of gas gangrene. Commun Med 6, 225 (2026). https://doi.org/10.1038/s43856-026-01503-y
Keywords: gas gangrene, Clostridium perfringens, bacterial toxins, drug repurposing, caspofungin