Clear Sky Science · en
In silico high-resolution whole lung model to predict the locally delivered dose of inhaled drugs
Why this matters for people with lung disease
Millions of people with asthma, COPD, or lung scarring rely on inhalers, yet doctors still cannot see exactly where the medicine lands inside each person’s lungs. Does the drug truly reach the diseased spots, or does it mostly stick in the throat and large airways? Because this is hard to measure inside living people, inhaled drugs are slow and expensive to develop, and doses are often set using rough averages rather than the needs of an individual patient. This study introduces a detailed computer model of the human lung that can predict, patient by patient, where every tiny inhaled particle goes and settles, potentially transforming how inhaled medicines are designed and prescribed.
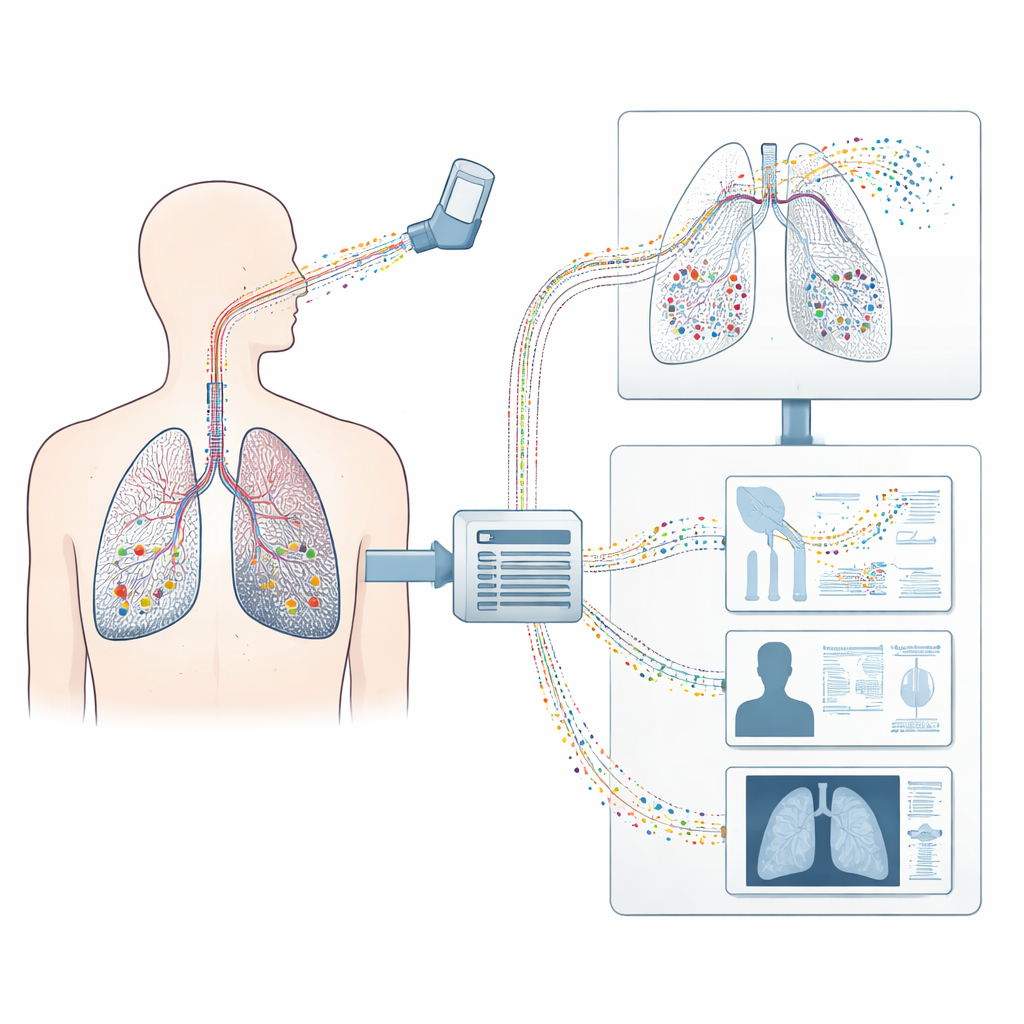
Turning a lung scan into a virtual twin
The researchers start from a standard medical CT scan and build a three-dimensional “digital twin” of a person’s lungs. They digitally extract the shape of the lung surfaces, the different lobes, and the visible airways. Because CT can only see the larger branches, a special growth algorithm fills in the missing smaller airways and the half billion grape-like air sacs where gas exchange occurs. The result is a complete branching tree of conducting airways linked to countless simplified alveolar units, plus surrounding structures like the chest wall and diaphragm that drive breathing motion. This full model captures both the geometry and the mechanical behavior of healthy and diseased lung tissue.
Following every particle with physics, not guesswork
On top of this digital lung, the team runs a physics-based simulation of air flow and tissue motion as a person breathes in and out along a real recorded breathing pattern. They then release virtual particles that represent the inhaled drug and compute their paths one by one as they are carried by the moving air, bounce, slow down, and eventually stick to airway walls or alveolar surfaces, or leave again during exhalation. Unlike older simplified models that treated the lung as a set of tubes or one-dimensional “trumpets,” this approach resolves the full three-dimensional paths from the windpipe all the way to the deepest regions. It tracks each particle at each moment of the breathing cycle, yielding a high‑resolution “map” of exactly where the medicine ends up.
Matching real-world scans in healthy volunteers
To test whether the computer predictions reflect reality, the authors compared their results with nuclear imaging data from an earlier clinical study in six healthy volunteers who inhaled a radioactive aerosol. That study used SPECT/CT scanners to visualize where the particles deposited in the lungs. For ten different inhalation experiments with two particle sizes and two breathing patterns, the model predicted how much drug mass reached each lung lobe and whether it landed more centrally or in the peripheral regions. These predictions closely matched the scan-based measurements, with typical discrepancies of only a few percentage points. The model also reproduced how smaller particles tend to penetrate deeper into the tissue, an effect seen in the imaging but never before captured so quantitatively by a whole-lung simulation.
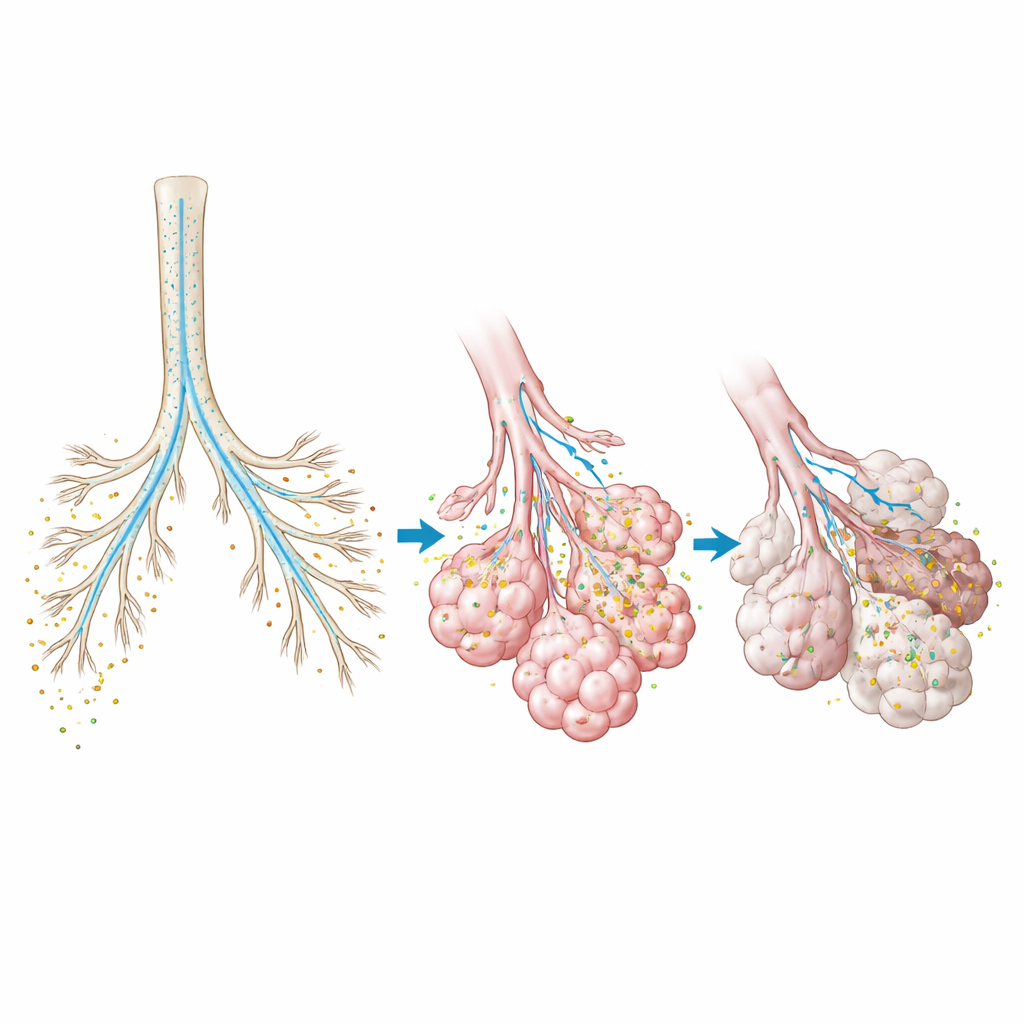
Zooming in on deep tissue and diseased areas
Because the virtual lung includes every generation of airways and the entire alveolar region, it can reveal details that imaging cannot easily provide. The researchers analyzed how much of the inhaled dose deposits in large airways versus the fine, gas-exchanging tissue and how this varies with breathing style and particle size. They also built a model of a lung with idiopathic pulmonary fibrosis, a disease that stiffens and scars patches of tissue. By assigning higher stiffness to the fibrotic regions seen on CT, they showed that these diseased zones received about 40% less drug per unit volume than healthier areas. This suggests that standard dosing may under-treat exactly the regions that most need medication, and that drug and device designs might need to be adapted for diseased lungs.
From better inhalers to fewer radioactive scans
In plain terms, this work shows that a computer model built from a patient’s scan can accurately forecast where inhaled medicine will land inside their lungs, without exposing them to extra radiation or invasive tests. Such a tool could help engineers design inhalers that deliver more drug to the right regions, help doctors choose particle size and breathing instructions tailored to each disease pattern, and offer regulators a way to test whether a generic inhaler truly matches a brand-name product. With fast, mostly automated model generation and high computing efficiency, the authors argue that virtual lung trials could eventually complement or even replace many nuclear imaging studies, making the development and personalization of inhaled treatments safer, faster, and less costly.
Citation: Grill, M.J., Biehler, J., Wichmann, KR. et al. In silico high-resolution whole lung model to predict the locally delivered dose of inhaled drugs. Commun Med 6, 188 (2026). https://doi.org/10.1038/s43856-026-01459-z
Keywords: inhaled drug delivery, lung modeling, aerosol deposition, personalized medicine, computational simulation