Clear Sky Science · en
Electrochemical Ferrier rearrangement of glycals in flow
Turning Simple Sugars into Powerful Building Blocks
Many life-saving medicines, from antibiotics to cancer drugs, rely on carefully tailored sugar structures. Making these sugar-based pieces in the lab has traditionally required harsh chemicals and energy-intensive conditions, which create waste and limit large-scale manufacturing. This paper describes a new way to reshape simple sugar molecules using electricity and a tiny flow reactor, offering a faster, cleaner, and more scalable route to important ingredients for pharmaceuticals and other advanced materials.
A Classic Sugar Trick, with Modern Limits
Chemists have long used a transformation called the Ferrier rearrangement to turn “glycals” – ring-shaped sugar derivatives – into 2,3-unsaturated glycosides. These products are key building blocks in complex natural molecules such as the anticancer drug paclitaxel and certain antibiotics. Traditionally, the rearrangement relies on strong acids or powerful oxidizing agents to activate the sugar, generating a highly reactive intermediate that can be attacked by another molecule, the nucleophile. While effective, these methods require corrosive reagents, can be hazardous to handle, generate substantial waste, and are not ideal for environmentally responsible or industrial-scale synthesis.
Using Electricity Instead of Harsh Reagents
In recent years, organic electrochemistry has emerged as a greener strategy, using electric current to drive chemical reactions instead of chemical oxidants and reductants. The authors previously showed that the Ferrier rearrangement could be carried out in an electrochemical batch reactor, but that approach still suffered from long reaction times, large amounts of supporting salt, and high electrical input. In this work, they move the process into a continuous-flow electrochemical microreactor equipped with inexpensive graphite electrodes. The tiny gap between electrodes and the serpentine flow channel greatly improves mixing and charge transfer, so the reaction can be completed in under 20 seconds of residence time while using minimal supporting electrolyte and a fraction of the charge previously required.

A Tiny Flow Reactor with Big Flexibility
To test the new setup, the team started with a common glycal (tri-O-acetyl-D-glucal) and benzyl alcohol as a partner molecule. Under optimized conditions in acetonitrile solvent, they obtained the desired 2,3-unsaturated glycoside in up to 94% yield with only 0.05 units of electrical charge per mole of starting material, far less than in earlier methods. They then explored how general the process is. Different glycals, including those derived from D-galactose or bearing alternative protecting groups, reacted smoothly. The reactor also tolerated a broad range of nucleophiles: simple alcohols, a sugar-based alcohol that formed a disaccharide, carbon-based partners that forged new carbon–carbon bonds, azides, nitrogen-containing sulfonamides, and sulfur-based nucleophiles. In many cases, the products were obtained in high yields and with favorable control over the three-dimensional arrangement of atoms at the new bond.
Fast, Scalable, and Greener
The continuous-flow design naturally lends itself to scale-up. The authors demonstrated a multi-gram preparation by pumping reactants through the microreactor at a higher flow rate, shrinking the residence time to just 18 seconds while maintaining high conversion and yield. This translated to a productivity of over 10 millimoles per hour and an impressive space-time yield, meaning a large amount of material can be produced per unit reactor volume and time. Using a standard green-chemistry assessment toolkit, they compared their method to earlier electrochemical Ferrier protocols. The new process avoids cryogenic temperatures and particularly hazardous solvents, improves yields, and dramatically reduces process mass intensity – a measure of how much material, including waste, is required per unit of product.
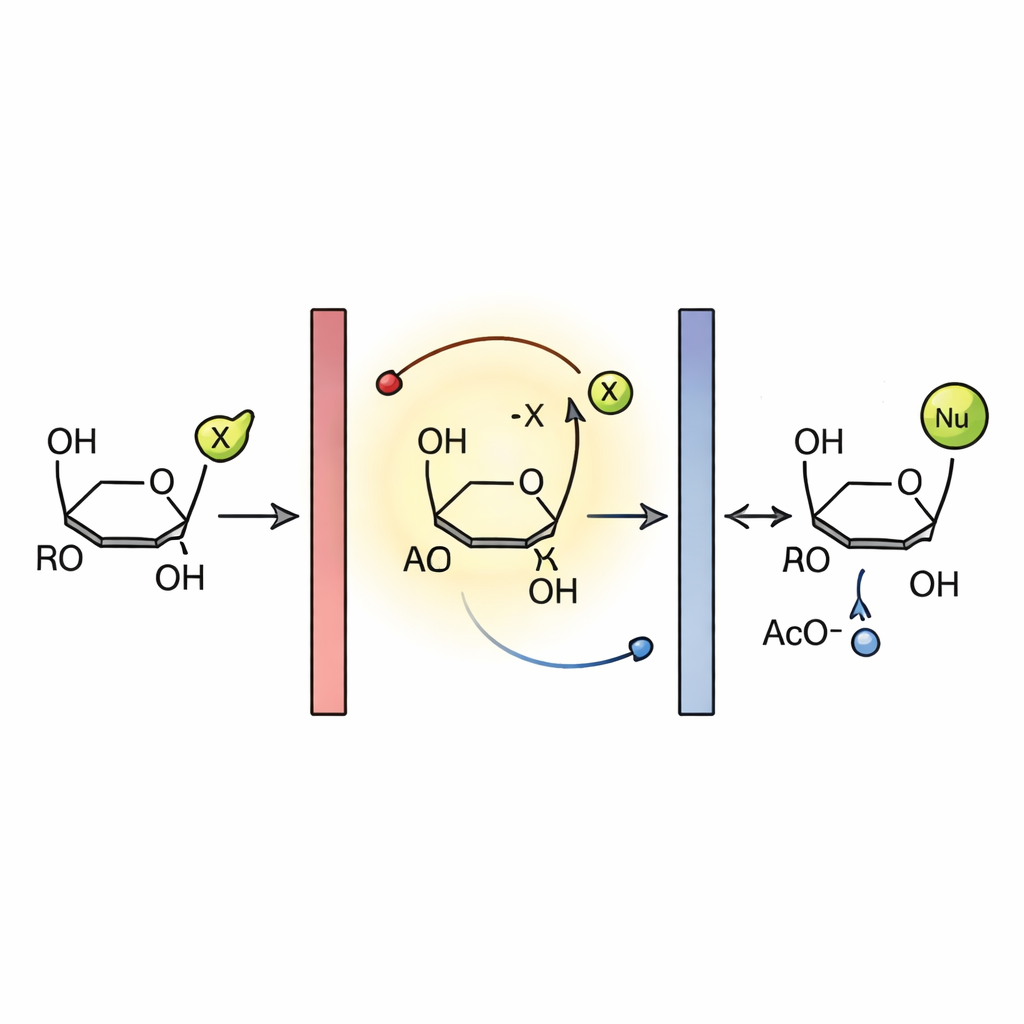
How Electricity Drives the Sugar Switch
Electrochemical measurements and prior studies point to a radical chain mechanism. In the reactor, the glycal is first oxidized at the anode, forming a short-lived radical cation. This species ejects an acetoxy radical, giving a positively charged intermediate that is quickly attacked by the nucleophile to form the new glycoside after loss of a proton. The expelled acetoxy radical helps propagate the chain by oxidizing another glycal molecule, while excess radicals are ultimately reduced at the cathode. Impervious graphite electrodes were crucial: other materials showed poor performance, likely because reactive species stuck to their surfaces and blocked efficient electron transfer. The combination of this robust electrode, rapid flow, and short diffusion paths underpins the high efficiency and selectivity of the process.
A Cleaner Future for Complex Sugars
Overall, the study shows that a classic sugar-rearranging reaction can be reimagined for modern needs by running it in an electrochemical flow microreactor. The method converts a variety of simple glycals and partner molecules into valuable 2,3-unsaturated glycosides rapidly, in high yield, and with far less waste and energy input than traditional routes. For non-specialists, the key message is that electricity, when carefully applied in miniature flow devices, can replace harsh reagents and enable more sustainable production of the sophisticated sugar fragments that underpin many advanced medicines and materials.
Citation: Suman, P., Fokin, M., Hunt, K.E. et al. Electrochemical Ferrier rearrangement of glycals in flow. Commun Chem 9, 145 (2026). https://doi.org/10.1038/s42004-026-01948-1
Keywords: electrochemical synthesis, flow chemistry, carbohydrate chemistry, glycosylation, green chemistry