Clear Sky Science · en
Iron-based single-atom catalysts for selective ammoxidation of C(sp3)–H bonds and oxidative C–C cleavage reactions
Turning Everyday Chemicals into Valuable Building Blocks
Chemists have long relied on toxic reagents and energy-hungry factories to make nitriles, a family of small but powerful chemical groups that appear in many medicines and materials. This study introduces a safer and more efficient way to make these compounds using a catalyst built from single iron atoms anchored in a porous carbon framework. By using ordinary air and ammonia under comparatively mild conditions, the new system can transform simple hydrocarbon feedstocks and even plastic waste into high‑value nitriles, pointing toward cleaner chemical manufacturing and more sustainable recycling. 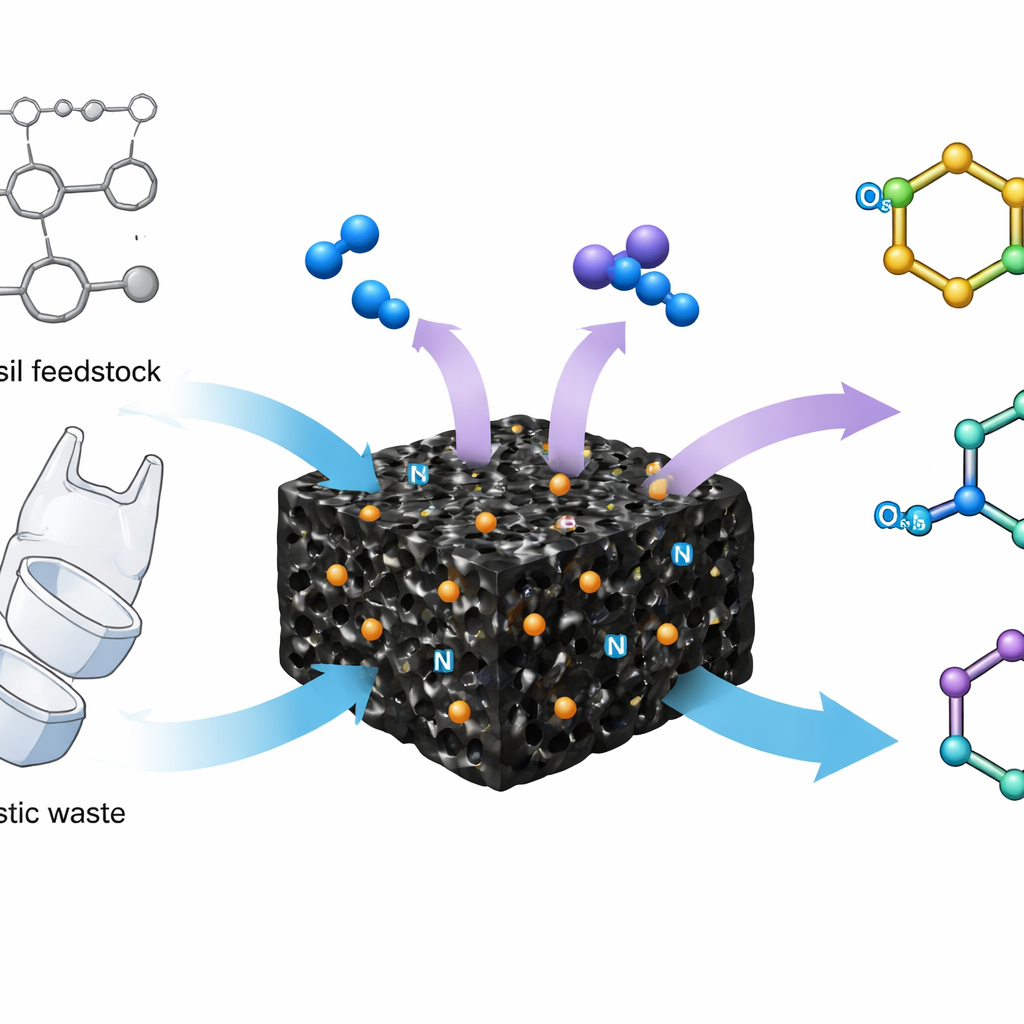
Why Nitriles Matter in Daily Life
Nitriles are quiet workhorses of modern life. The cyano group they contain appears in more than 60 approved drugs, including treatments for depression, breast cancer and leukemia, as well as in many agrochemicals and specialty materials. Traditionally, industry makes nitriles by reacting aromatic building blocks with cyanide salts or hydrogen cyanide gas. These routes are efficient but hazardous: they use highly poisonous reagents and generate large amounts of toxic waste. A more attractive approach is to “upgrade” simple hydrocarbons by selectively oxidizing their carbon–hydrogen bonds with oxygen, turning cheap methylarenes and related molecules directly into nitriles without cyanide. Until now, however, this strategy has required rare metals, harsh conditions, or has worked only for a narrow set of starting materials.
Designing a Catalyst One Atom at a Time
The authors tackled this challenge by designing a heterogeneous catalyst in which individual iron atoms are locked into a sponge‑like carbon network that also contains nitrogen and boron. Instead of forming larger iron particles, which can be less selective and harder to control, the preparation method disperses the iron as isolated sites surrounded by nitrogen atoms, with nearby boron fine‑tuning the local environment. Advanced microscopy and spectroscopy show that these iron centers sit in a micro‑ and mesoporous carbon matrix, offering a very high surface area and channels that allow oxygen, ammonia and organic molecules to diffuse in and react. Measurements of how the catalysts store and activate oxygen, and how acidic their surfaces are, reveal that the boron–nitrogen co‑doping makes the iron sites particularly good at triggering the first, difficult bond‑breaking steps.
From Simple Aromatics to Medicines and Beyond
When tested on a model compound, 4‑methylanisole, the iron single‑atom catalyst outperformed a wide range of commercial noble‑metal catalysts and industrial materials, delivering the corresponding nitrile in high yield and selectivity using only aqueous ammonia, oxygen and water as solvent. The team then explored the scope of the reaction and found it could convert more than 60 different starting materials, including toluenes, multi‑substituted aromatics and nitrogen‑containing ring systems, into their nitriles. These products are valuable intermediates for pharmaceuticals and crop‑protection agents. The catalyst also promotes a more demanding transformation: the oxidative cutting of carbon–carbon bonds in alkylarenes. In these cases, side chains are cleaved to give simple benzonitriles, even from complex starting materials such as lignin model compounds derived from biomass. 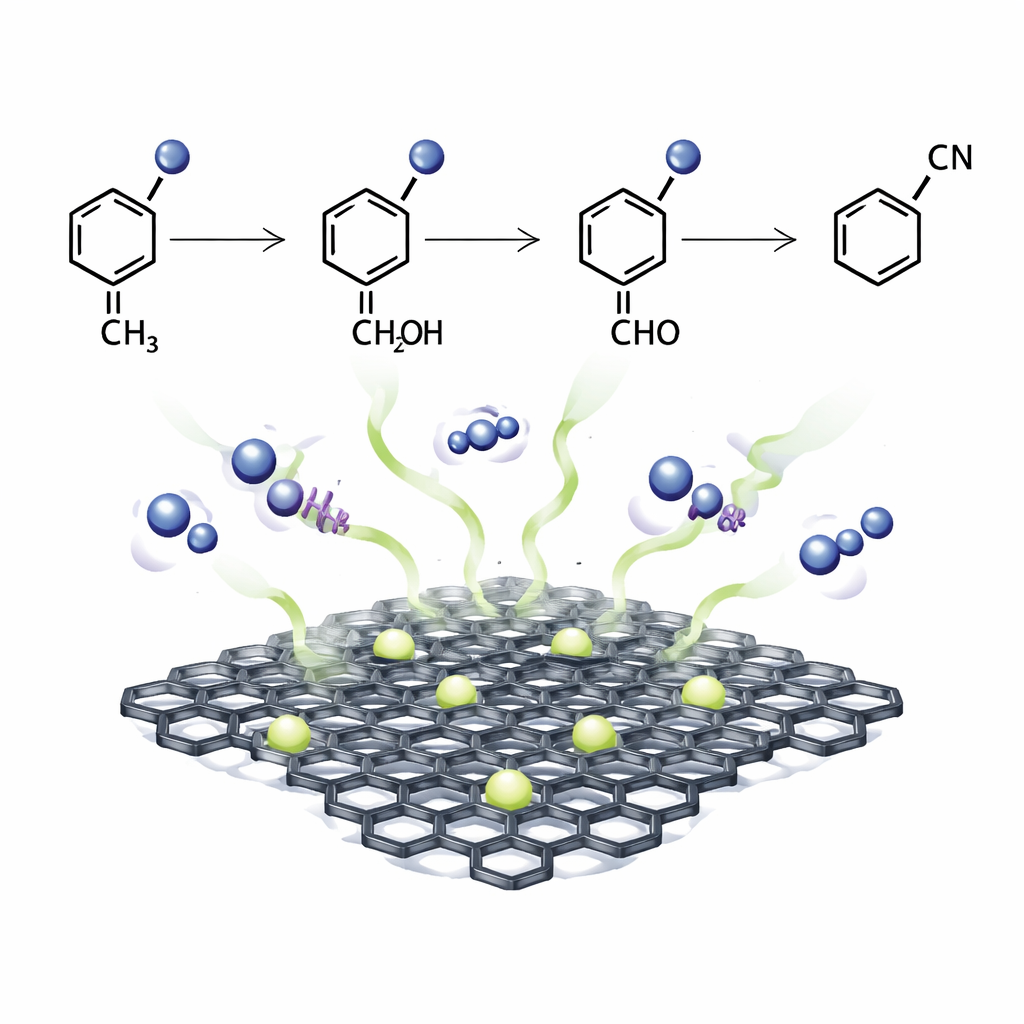
Upcycling Plastic Waste into Useful Molecules
Beyond fine chemicals, the same chemistry can upgrade plastic waste. Polystyrene and related copolymers, common in packaging and foam products, consist of chains of aromatic rings linked by carbon–carbon bonds. Under the new catalytic conditions, these polymers break down to give benzonitrile and benzamide in useful yields, while size‑exclusion measurements confirm that the polymer chains are indeed being cut into smaller fragments. Remarkably, the catalyst tolerates real plastic waste containing additives and fillers, and can operate in continuous‑flow reactors for many hours before needing regeneration. A simple treatment with dilute hydrogen peroxide restores the single‑atom iron sites and revives activity, allowing multiple reuse cycles.
How the Catalyst Does the Job
Control experiments and real‑time spectroscopic studies suggest a stepwise mechanism. Oxygen is first activated on the iron sites to form reactive superoxide species. These species abstract hydrogen atoms from the benzylic positions of the organic substrate, creating radicals that are then converted into aldehydes or ketones. In the presence of ammonia, these intermediates form imines, which are further oxidized to nitriles. For alkylarenes and polymers, a similar sequence leads to cleavage of carbon–carbon bonds and release of aromatic nitriles. The key to this behavior is the unique arrangement of iron, nitrogen and boron within the porous carbon, which stabilizes iron as highly active single sites while avoiding inactive nanoparticles.
Toward Greener Chemistry and Circular Materials
In essence, this work shows that carefully engineered single‑atom iron catalysts can rival or surpass more expensive and hazardous systems for making nitriles, one of the chemical industry’s staple functional groups. By relying on abundant iron, air and ammonia and operating under liquid‑phase conditions, the process points toward safer and more sustainable production of pharmaceutical ingredients and specialty chemicals. At the same time, its ability to convert lignin‑like fragments and polystyrene waste into valuable nitriles illustrates how advanced catalysis could help close material loops and turn discarded carbon into useful resources rather than pollution.
Citation: Ma, Z., Rockstroh, N., Chen, Z. et al. Iron-based single-atom catalysts for selective ammoxidation of C(sp3)–H bonds and oxidative C–C cleavage reactions. Nat Catal 9, 389–403 (2026). https://doi.org/10.1038/s41929-026-01513-y
Keywords: single-atom catalysis, green nitrile synthesis, plastic upcycling, C–H bond activation, iron catalysts