Clear Sky Science · en
Targeting erythroid progenitor cell metabolism to enhance cancer immunotherapy
Why Immune-Based Cancer Treatments Sometimes Fall Short
Immune-based drugs have changed how many cancers are treated, yet most patients still do not get lasting benefit. This article explains an unexpected culprit: a population of immature blood cells known as erythroid progenitor cells, or EPCs. Normally on their way to becoming red blood cells, these cells can be rewired by tumors into powerful blockers of the body’s defenses. Understanding and disarming this hidden network offers a new way to make current cancer immunotherapies work better and for more people. 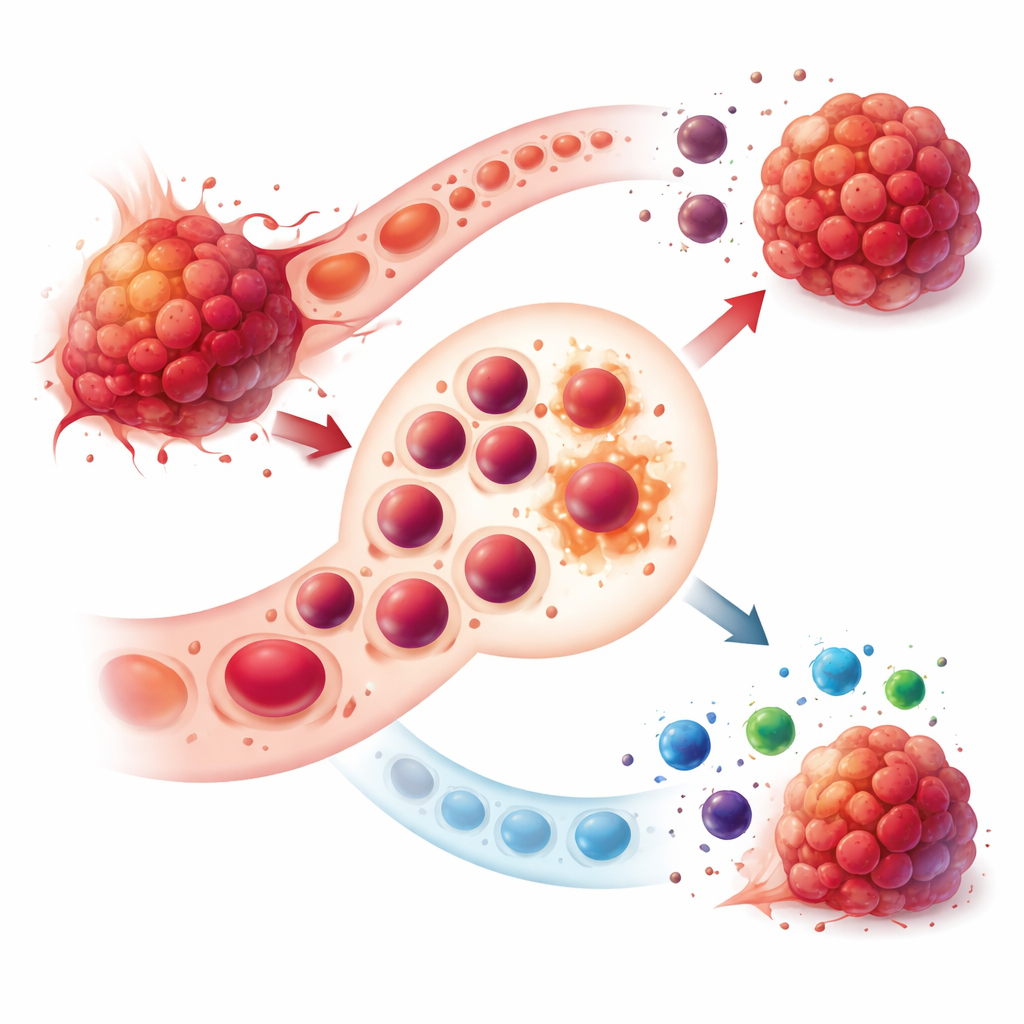
A Hidden Cell Group That Helps Tumors
Under healthy conditions, EPCs live mainly in the bone marrow and quietly mature into red blood cells that carry oxygen. In people with advanced cancers, this calm process is disrupted. Tumors trigger blood formation outside the bone marrow, especially in the spleen, in a process called extramedullary hematopoiesis. This leads to a surge of EPCs that no longer act like simple precursors. Instead, they split into two main groups: CD45-positive EPCs that can travel into tumors, and CD45-negative EPCs that stay in the spleen but influence immunity from a distance. Both groups gain strong immune-dampening abilities that help cancer escape attack.
How Tumors Rewire EPCs
Tumors release a cocktail of growth factors and inflammatory signals that reshape EPC behavior. Substances such as erythropoietin, G-CSF, GM-CSF, TGF-beta, and others expand EPC numbers and push them away from becoming healthy red blood cells. Some CD45-positive EPCs convert into erythroid-derived myeloid cells, a hybrid cell type that resembles other known suppressor cells. Inside these cells, core metabolic pathways are altered: they ramp up the production of reactive oxygen species, enzymes that break down the amino acid arginine, and other metabolites. These changes turn EPCs into factories of immune-inhibiting molecules rather than helpers of normal blood production. 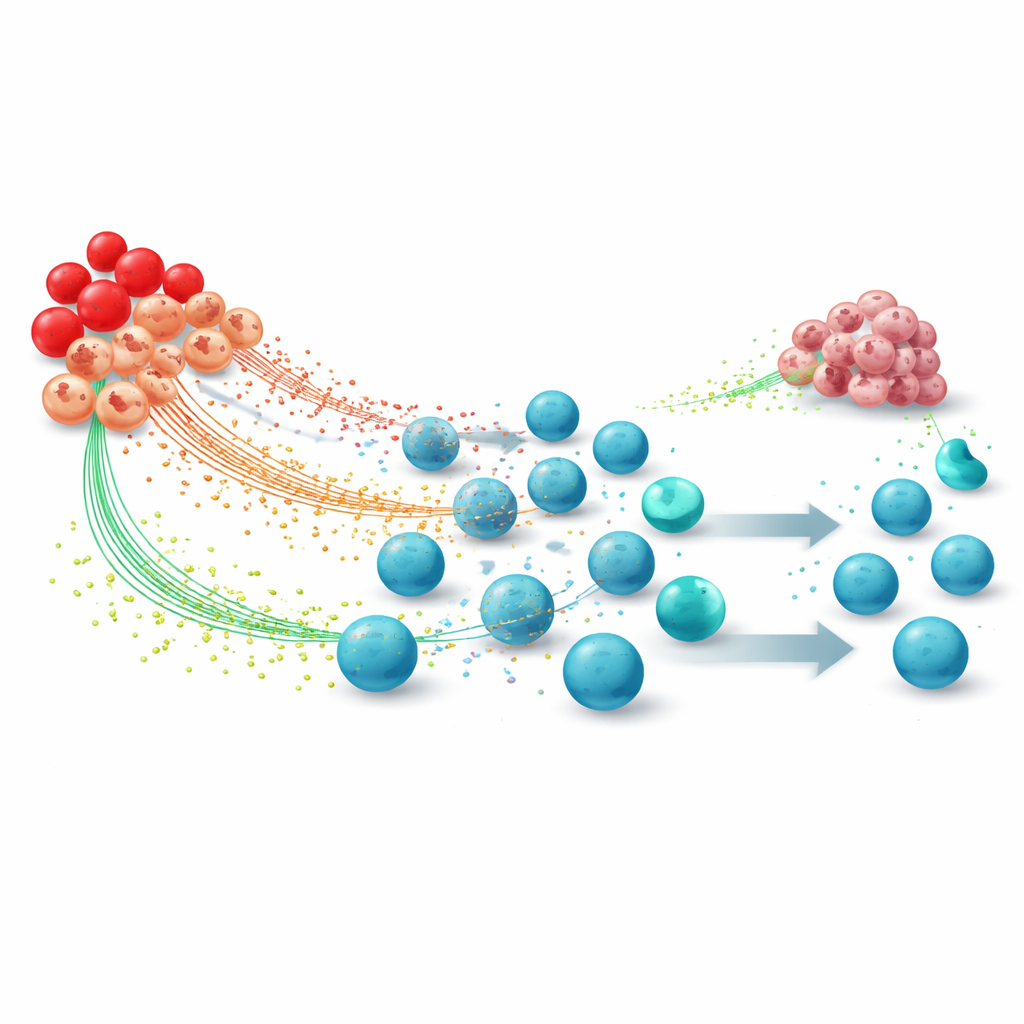
Shutting Down Immune Cells from Multiple Angles
Rewired EPCs undermine anti-cancer immunity in several coordinated ways. High levels of reactive oxygen species and related chemicals can damage key structures on T cells, the very cells that immunotherapies aim to activate, blunting their ability to recognize cancer. Arginase enzymes released by EPC-derived cells strip arginine from the tumor environment, starving T cells of a nutrient they need to grow, divide, and fuel their energy demands. At the same time, EPC-derived cells display molecules such as PD-L1 that send a “stop” signal to T cells, driving them into an exhausted state. The CD45-negative EPCs that remain in the spleen secrete TGF-beta and other factors that limit the activation of killer T cells and natural killer cells while fostering regulatory T cells that further quiet immune responses.
New Ways to Tip the Balance Back to the Patient
Because EPCs sit at a crossroads between blood formation, metabolism, and immunity, they offer many new drug targets. One strategy is to prevent the excess production and misdirection of EPCs by blocking the signals that draw blood stem cells to the spleen or push EPCs toward suppressive fates. Another is to keep EPCs from entering tumors by interfering with the chemical trails that guide them. More direct approaches aim to eliminate the most harmful EPC populations using targeted antibodies or to reset their metabolism by blocking arginase, tuning reactive oxygen species, or neutralizing hormones such as erythropoietin and TGF-beta. The authors also highlight future work using single-cell and spatial profiling to map EPC metabolism in detail and to develop blood-based biomarkers that could flag when these cells are driving treatment resistance.
What This Means for Cancer Patients
The central message of the article is that EPCs are not just bystanders in cancer; they can act as active allies of the tumor by reshaping both blood production and immune defenses. Tumors turn what should be a normal red blood cell factory into a source of cells that shield them from attack. By learning how this metabolic hijacking works, researchers hope to design drugs that either prevent EPCs from becoming suppressive or selectively remove them, while preserving healthy red blood cell formation. Combined with existing immune checkpoint drugs and other therapies, EPC-focused strategies could make it harder for tumors to hide, improving outcomes and survival for many patients.
Citation: Li, ZZ., Huang, HL., Li, YC. et al. Targeting erythroid progenitor cell metabolism to enhance cancer immunotherapy. npj Precis. Onc. 10, 163 (2026). https://doi.org/10.1038/s41698-026-01362-9
Keywords: cancer immunotherapy, erythroid progenitor cells, tumor microenvironment, immune suppression, cell metabolism