Clear Sky Science · en
Integration of radiomics, deep learning, transcriptomics, and metabolomics reveals prognostic risk stratification and underlying biological mechanisms in colorectal cancer
Why this research matters to patients and families
Colorectal cancer is one of the most common and deadly cancers worldwide, yet doctors still struggle to predict which patients will do well after surgery and which are at higher risk of dying from the disease. This study shows that ordinary medical scans, when combined with advanced computer analysis and molecular tests, can sort patients into clear risk groups and uncover hidden biology inside their tumors. In the future, this kind of approach could help tailor treatment and follow-up so that high-risk patients get closer monitoring and stronger therapies, while low-risk patients avoid unnecessary side effects.
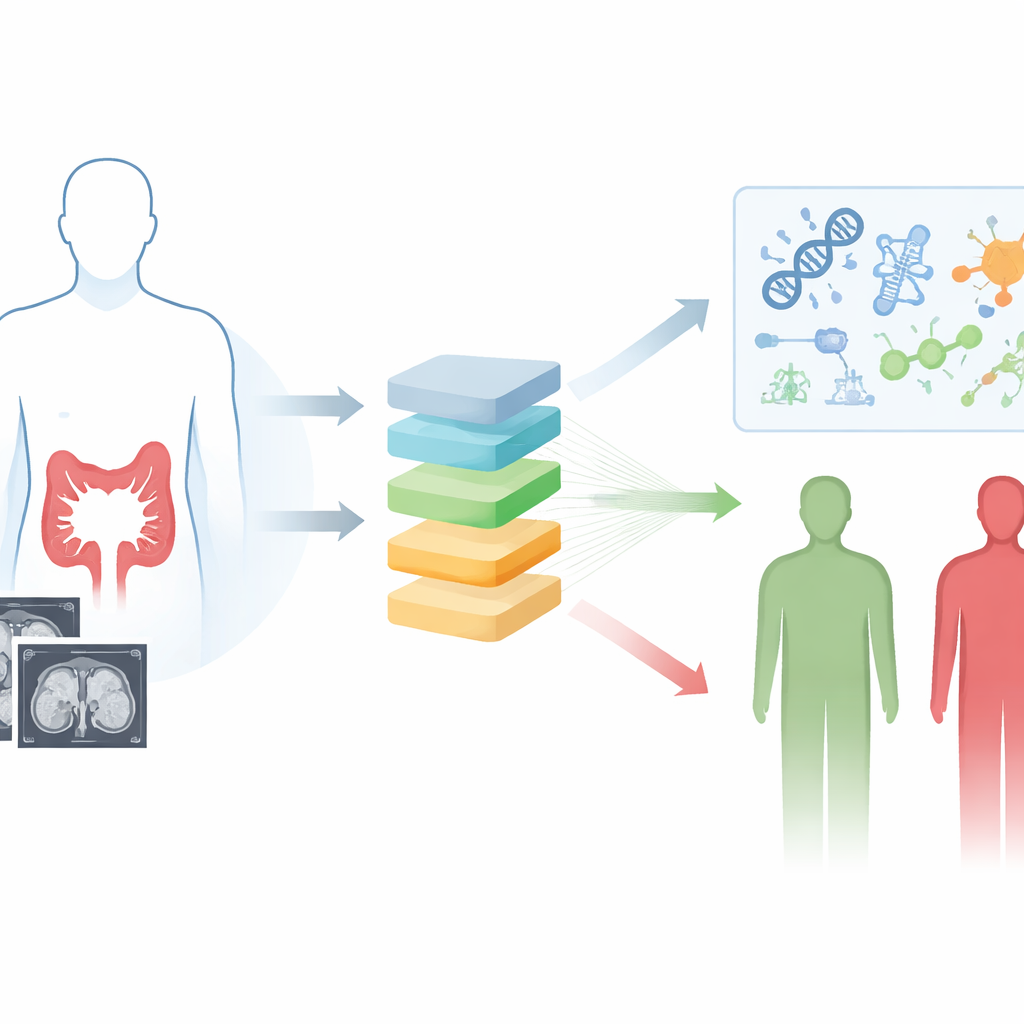
Looking inside tumors without a scalpel
The researchers focused on contrast-enhanced CT scans, a routine imaging test already used to evaluate colorectal cancer. Instead of relying on what the human eye can see, they used radiomics and deep learning to extract nearly 2,000 tiny quantitative details from the images, capturing subtle patterns of shape and texture that reflect tumor heterogeneity. They then systematically tested 117 combinations of ten machine-learning algorithms to find the most reliable way to link these image features to how long patients survived. The winning model, which blended two techniques called Lasso and gradient boosting, produced a single risk score for each person based purely on their preoperative CT scan.
Separating low-risk and high-risk patients
Using CT scans from 1,183 patients treated at four hospitals, the team trained and rigorously tested their deep learning radiomics model. They identified a cut-off in the image-based score that split patients into low-risk and high-risk groups. Across the training set, an internal validation set, and two independent hospitals, high-risk patients consistently lived for a shorter time than those in the low-risk group. The model’s ability to distinguish outcomes held up across many subgroups, including younger and older patients, men and women, different tumor stages, and whether or not patients received chemotherapy. When the researchers combined the image-based score with two standard clinical indicators—blood levels of CEA and lymph node stage—into a simple graphic tool called a nomogram, predictions of three- and five-year survival became even more accurate and clinically useful.
What makes one tumor more dangerous than another?
To move beyond prediction and understand why some tumors behave more aggressively, the team linked imaging-based risk groups to molecular changes in the cancer. In a subset of patients, they analyzed small molecules in tumor tissue using nuclear magnetic resonance spectroscopy and compared tumors to nearby normal tissue. They found that high-risk and low-risk tumors had distinct metabolic fingerprints, involving pathways that handle amino acids and short-chain fatty acids. In parallel, they examined gene activity in tumors from a public imaging database. High-risk tumors showed strong signals from pathways related to the extracellular matrix—the supportive scaffold around cells—which is known to help cancer cells spread and resist therapy. Low-risk tumors, in contrast, were enriched for immune-related pathways and had higher levels of cancer‑killing CD8 T cells, pointing to a more active anti-tumor immune environment.
Protective metabolic pathways linked to better survival
Strikingly, both the metabolite data and the gene activity data pointed to the same two metabolic routes—butanoate metabolism and nitrogen metabolism—as being more active in low-risk tumors. These pathways are connected to products of gut bacteria and to how cells process nitrogen, which influences energy balance and immune function. When the researchers checked a large, independent dataset of 417 colorectal cancer patients from The Cancer Genome Atlas, they found that lower activity in these two pathways was clearly associated with worse overall survival. This suggests that maintaining these metabolic processes may help restrain tumor growth or support a stronger immune response, and that they might one day be explored as biomarkers or therapeutic targets.
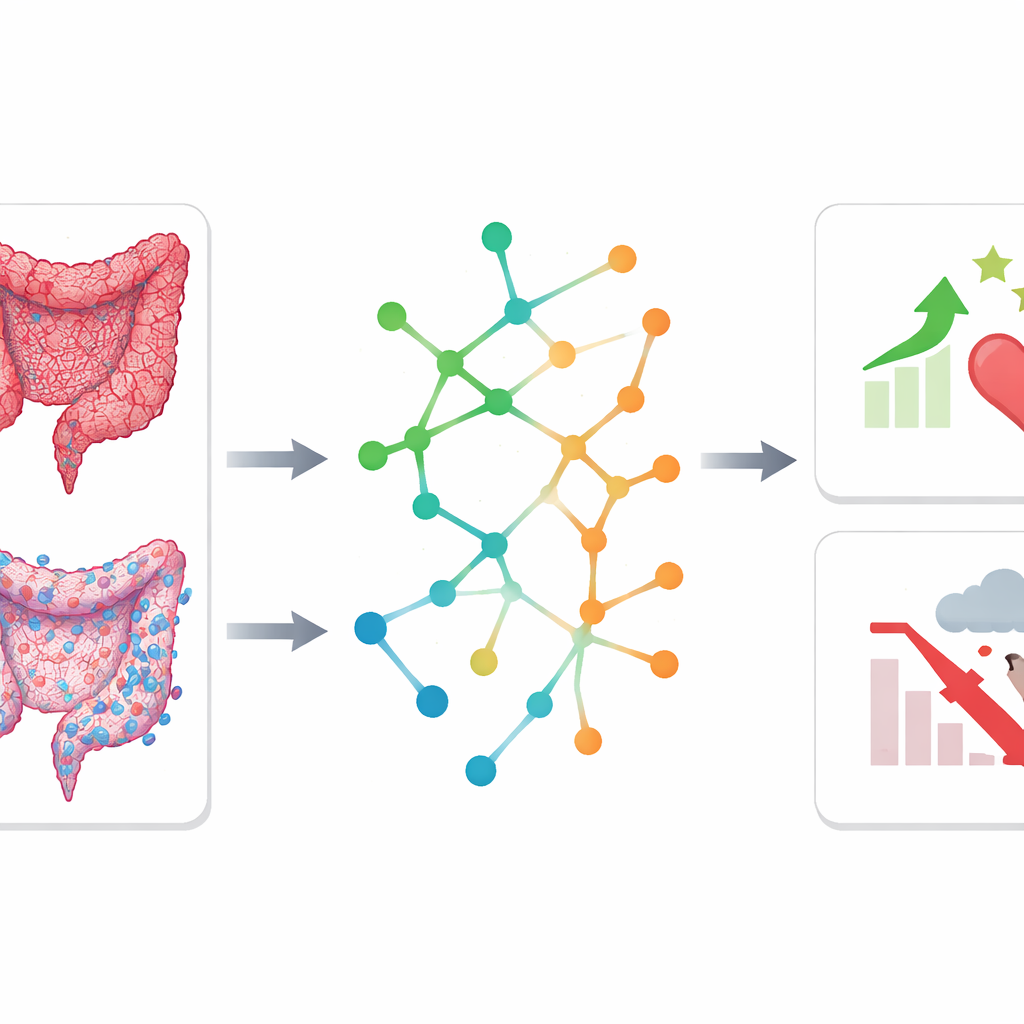
What this means for future cancer care
In simple terms, this study shows that combining smart image analysis with molecular profiling can both sharpen prognostic tools and reveal what is happening inside colorectal tumors. A risk score derived from standard CT scans, especially when paired with routine clinical measurements, could help doctors identify which patients need more intensive treatment or follow-up after surgery. At the same time, the discovery that certain immune patterns and metabolic pathways are linked to better outcomes hints at new ways to support the body’s defenses and design targeted therapies. While larger, prospective studies are needed to confirm these findings, this integrative approach marks a step toward more personalized, biology-informed care for people with colorectal cancer.
Citation: Li, Z., Cai, R., Qin, Y. et al. Integration of radiomics, deep learning, transcriptomics, and metabolomics reveals prognostic risk stratification and underlying biological mechanisms in colorectal cancer. npj Precis. Onc. 10, 155 (2026). https://doi.org/10.1038/s41698-026-01331-2
Keywords: colorectal cancer, radiomics, deep learning, tumor metabolism, immune microenvironment