Clear Sky Science · en
Experimental and theoretical insights into the inhibitory capabilities of polythiophene and polypyrrole molecules for protecting mild steel from corrosion in sulfuric acid
Why protecting everyday steel matters
From bridges and cars to factory pipes, much of modern life depends on mild steel. Yet this common metal slowly dissolves when it meets harsh acids used in cleaning and manufacturing. The study behind this article explores how two special plastic-like materials, polythiophene and polypyrrole, can wrap steel in a molecular raincoat that keeps it from wasting away in sulfuric acid, a widely used industrial chemical.
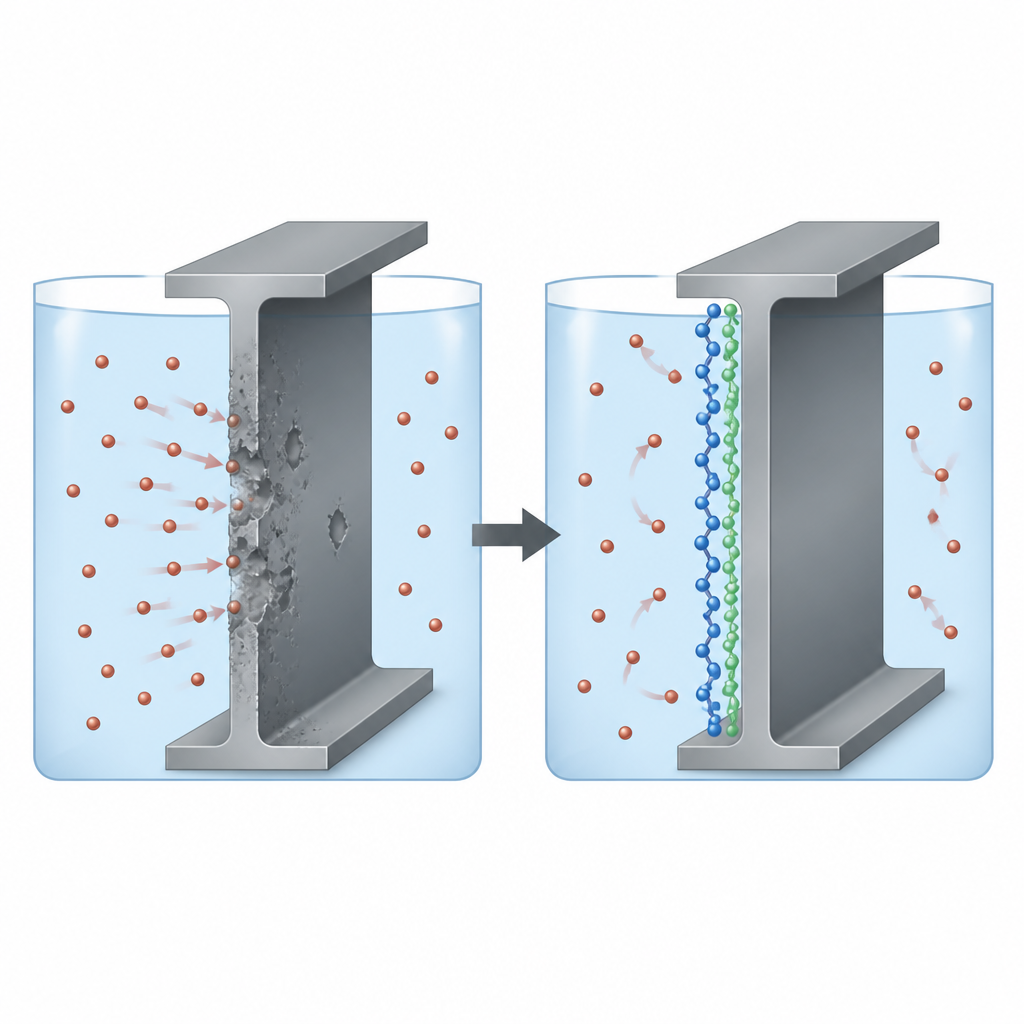
Steel under attack in acidic solutions
In many industries, sulfuric acid is essential for cleaning, pickling, and shaping steel parts, but it also aggressively eats into the metal surface. Engineers often add “corrosion inhibitors” to the acid to slow this damage. The authors focused on two electrically conducting polymers, polythiophene and polypyrrole, chosen because their chain-like structures carry clouds of mobile electrons and reactive atoms that can cling to steel. The central question was whether these polymers could form a thin shield on mild steel in sulfuric acid, and which of the two offers better protection.
Testing how well the molecular shield works
The team first tested real steel samples in sulfuric acid with and without the polymers. They measured how quickly metal dissolved by tracking weight loss over time and by using sensitive electrical methods that reveal how easily current flows during corrosion. In every case, adding either polymer sharply reduced the corrosion rate and slowed both the metal-dissolving and gas-forming reactions at the surface. At higher polymer doses, corrosion dropped by more than 90 percent, meaning only a small fraction of the steel was lost compared with unprotected samples. The experiments also showed that the polymers helped steel resist “pitting,” a dangerous form of highly localized attack that drills deep holes into the metal.
How a thin film blocks the acid
Electrical measurements painted a picture of what happens at the steel surface. With more polymer present, the resistance to charge flow increased and the apparent electrical capacitance at the surface decreased. In simpler terms, a denser, more insulating layer built up between the steel and the acid, forcing electrons and corrosive ions to work much harder to pass through. Analysis of how performance changed with temperature suggested that the polymers are held mainly by physical attraction and electrostatic forces, rather than strong chemical bonds, and that they arrange themselves in a single, fairly uniform layer on the metal. This agrees with classical models of adsorption in which the surface gradually fills with inhibitor molecules until most reactive sites are covered.
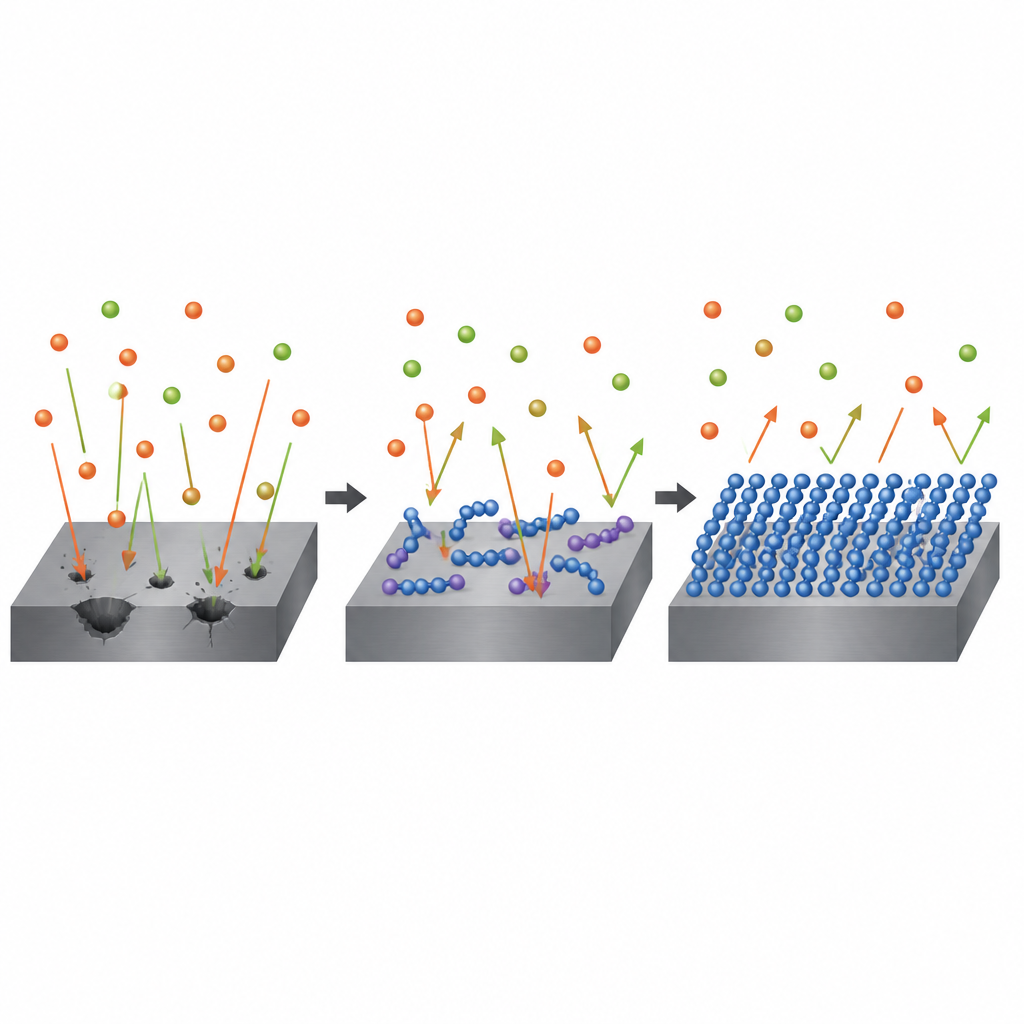
Peering into the molecules on the computer
To understand why one polymer works better than the other, the researchers turned to computer chemistry. They used quantum calculations to map where electrons sit on each molecule and how easily they can move to or from the steel surface. Polypyrrole was predicted to donate more electron density to iron and to present more strongly charged regions that can grip the metal. Simulations that virtually “land” the polymers on a model iron surface found that polypyrrole binds a little more strongly and lies flat, maximizing contact. These factors help it build a tighter, more cohesive protective film than polythiophene, even though polythiophene appears slightly more reactive in some simple electronic measures.
Why polypyrrole comes out on top
Taken together, the lab tests and the computer models tell a consistent story. Both polythiophene and polypyrrole can act like a self-organizing coat that clings to mild steel in sulfuric acid, greatly slowing the rate at which the metal dissolves and helping to prevent deep pits from forming. However, polypyrrole interacts more strongly with iron and packs more efficiently on the surface, so it delivers higher protection at every concentration tested. For industries that must use strong acids but want equipment to last longer and fail less often, such polymer-based inhibitors offer a promising route to safer, more durable steel infrastructure.
Citation: Abdallah, M., Al-Gorair, A.S., Al Jahdaly, B.A. et al. Experimental and theoretical insights into the inhibitory capabilities of polythiophene and polypyrrole molecules for protecting mild steel from corrosion in sulfuric acid. Sci Rep 16, 15045 (2026). https://doi.org/10.1038/s41598-026-50293-9
Keywords: steel corrosion, corrosion inhibitors, conducting polymers, sulfuric acid, polypyrrole