Clear Sky Science · en
Detection of inflammation-related blood–brain barrier dysfunction using PET and MR imaging: a pilot study
Why tiny leaks in the brain’s shield matter
Many brain diseases, from Alzheimer’s to traumatic brain injury, are tied to a slow-burning form of inflammation inside the brain. A key player in this story is the blood–brain barrier, a protective wall of cells that normally keeps harmful substances out of the brain. When this barrier starts to leak, trouble follows—but catching those early, subtle leaks has been very difficult. This study tests a new, non-invasive scan in mice that aims to spot such small barrier defects long before obvious brain damage or symptoms appear.
Watching the brain’s protective wall
The blood–brain barrier acts like a highly selective filter between the blood and the brain. It allows in nutrients but blocks many large molecules, including most blood proteins. In chronic brain inflammation, immune cells and their chemical messengers can weaken this filter, letting big molecules seep into brain tissue. Clinicians currently rely on blood tests and spinal fluid samples to infer that the barrier is compromised, but these methods cannot precisely show where in the brain the leak occurs or how severe it is. Imaging tools such as PET and MRI can see inside the living brain, yet most existing PET tracers slip through the barrier too easily, making it hard to tell whether the barrier itself is damaged.
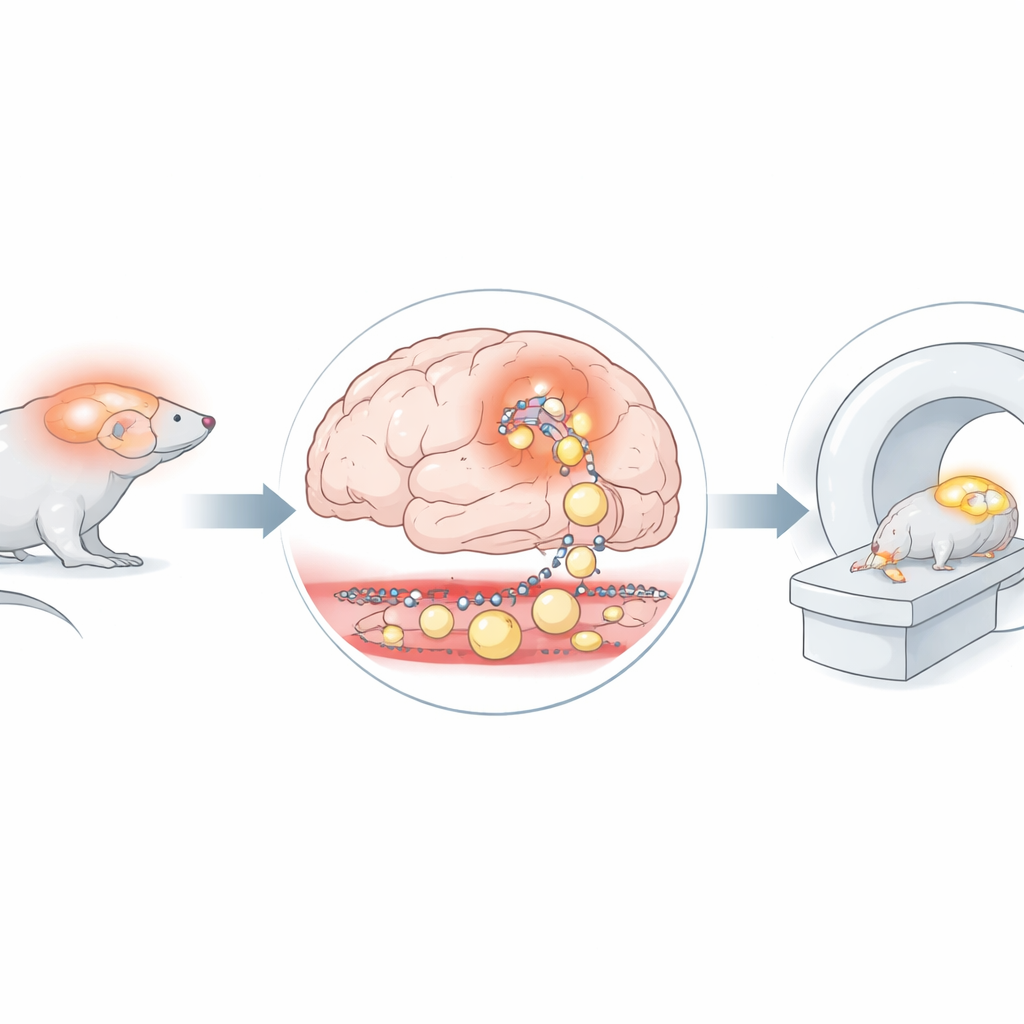
A tagged protein as a visible guest
The researchers explored a new strategy: taking human serum albumin—a common, large blood protein that normally stays within the bloodstream—and attaching a weakly radioactive tag to it. Because albumin is bulky and does not usually cross into the brain, any signal from this tagged protein inside brain tissue should signal a genuine leak. The team used a mouse model engineered to develop chronic brain inflammation through a specific signaling pathway in support cells called astrocytes. These mice, along with healthy control mice, were injected with the radiolabeled albumin and scanned using combined PET and MRI at one hour and again 24 hours after injection. MRI provided detailed pictures of brain structure, while PET showed where the tagged albumin accumulated over time.
Hidden changes that scans can see
On MRI, the brains of inflamed and healthy mice looked essentially the same: no shrinkage, no obvious lesions, and no visible breakdown of brain tissue, even in areas known to be vulnerable. However, PET told a different story. In the mice with chronic neuroinflammation, the tagged albumin signal was consistently higher in the brain, and especially in the cerebellum, a region involved in coordination and balance. Over 24 hours, the amount of tracer in the blood steadily dropped in both groups, but the ratio of brain signal to blood signal rose more strongly in the inflamed mice. This pattern indicates that more of the large protein was slipping through the barrier and remaining within brain tissue in these animals.
Checking the brain after the scan
To confirm the scans, the researchers examined the organs after the final imaging session. They measured radioactivity in the brain, blood, and major organs and again found higher levels of the tagged albumin in the brains of inflamed mice, with similar levels in peripheral organs across both groups. Thin slices of brain tissue were stained and inspected under a microscope, revealing that the basic architecture of the cerebellum remained intact—cell layers were preserved and there were no obvious signs of cell loss. Yet digital autoradiography, a technique that maps radioactivity in the tissue slices, showed stronger signal in the cerebellum of inflamed mice, mirroring the PET findings. Together, these results suggest that barrier leaks were already present even though standard structural measures still looked normal.
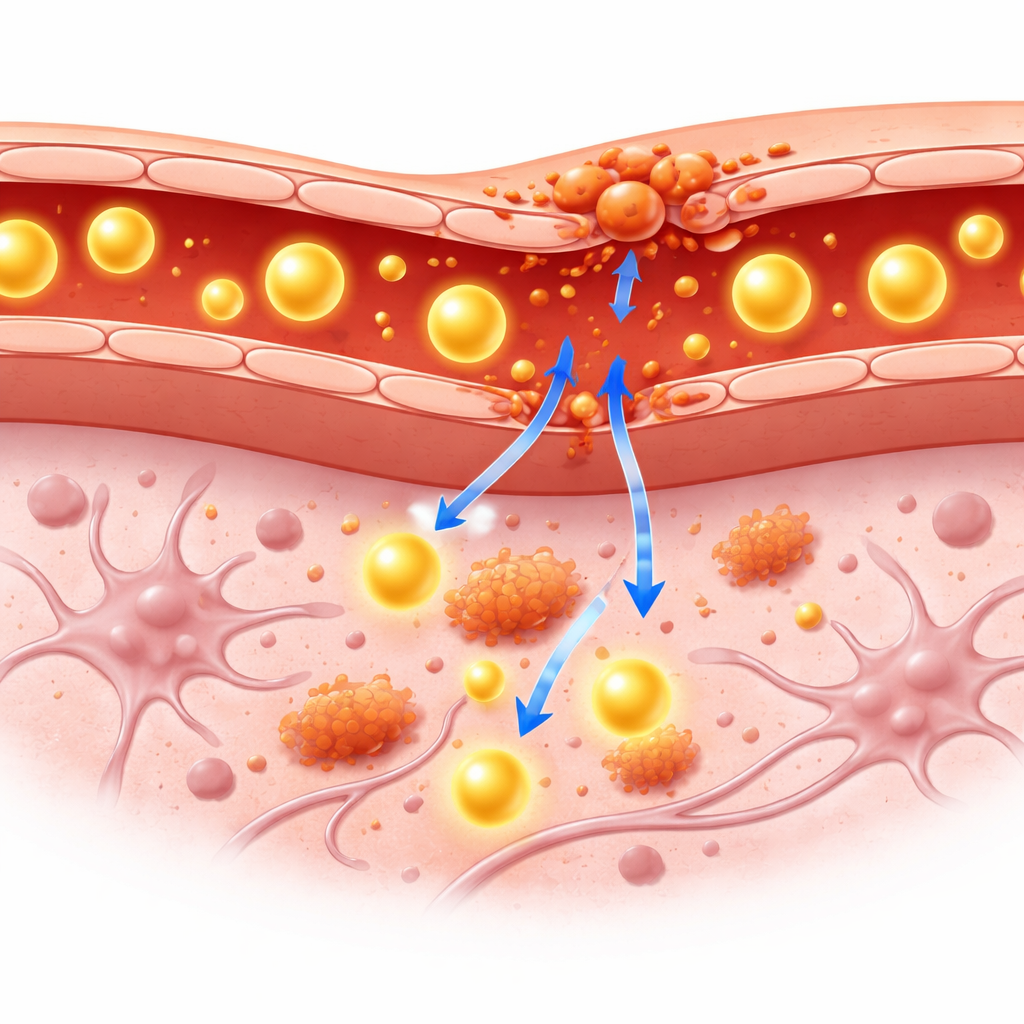
What this means for future brain care
This pilot study suggests that a radiolabeled albumin tracer, combined with PET/MR imaging, can sensitively detect early changes in the brain’s protective barrier during chronic inflammation, particularly in regions like the cerebellum. The method picked up subtle leakage of large molecules before any clear tissue damage appeared on MRI or in routine microscopic examination. While the work was done in a small number of mice and faces some technical challenges—especially in very small blood vessels—the approach may eventually help doctors monitor barrier health in people with neuroinflammatory diseases or brain injury. In simple terms, it offers a way to see the brain’s shield starting to fail, opening the door to earlier diagnosis and potentially earlier, more targeted treatment.
Citation: Hilbrig, C.F., Baumann, B., Sievert, W. et al. Detection of inflammation-related blood–brain barrier dysfunction using PET and MR imaging: a pilot study. Sci Rep 16, 12014 (2026). https://doi.org/10.1038/s41598-026-47352-6
Keywords: blood–brain barrier, neuroinflammation, PET MRI, albumin tracer, brain imaging