Clear Sky Science · en
Protective role of sodium propionate against glycerol or fractionated doses of gamma rays-induced acute kidney injury via ATF5-induced mitophagy in rats
Why Kidney Damage Matters
Acute kidney injury is a sudden breakdown of the body’s filtration system, and it can strike after exposure to certain drugs, severe muscle damage, or medical radiation. When kidneys fail, waste products build up in the blood within hours to days, often landing patients in intensive care. This study explores whether a simple gut-derived chemical, sodium propionate—a short-chain fatty acid related to dietary fiber fermentation—can shield kidneys from two very different but clinically relevant types of injury in rats: one triggered by a muscle-breakdown toxin (glycerol) and the other by repeated doses of gamma radiation.
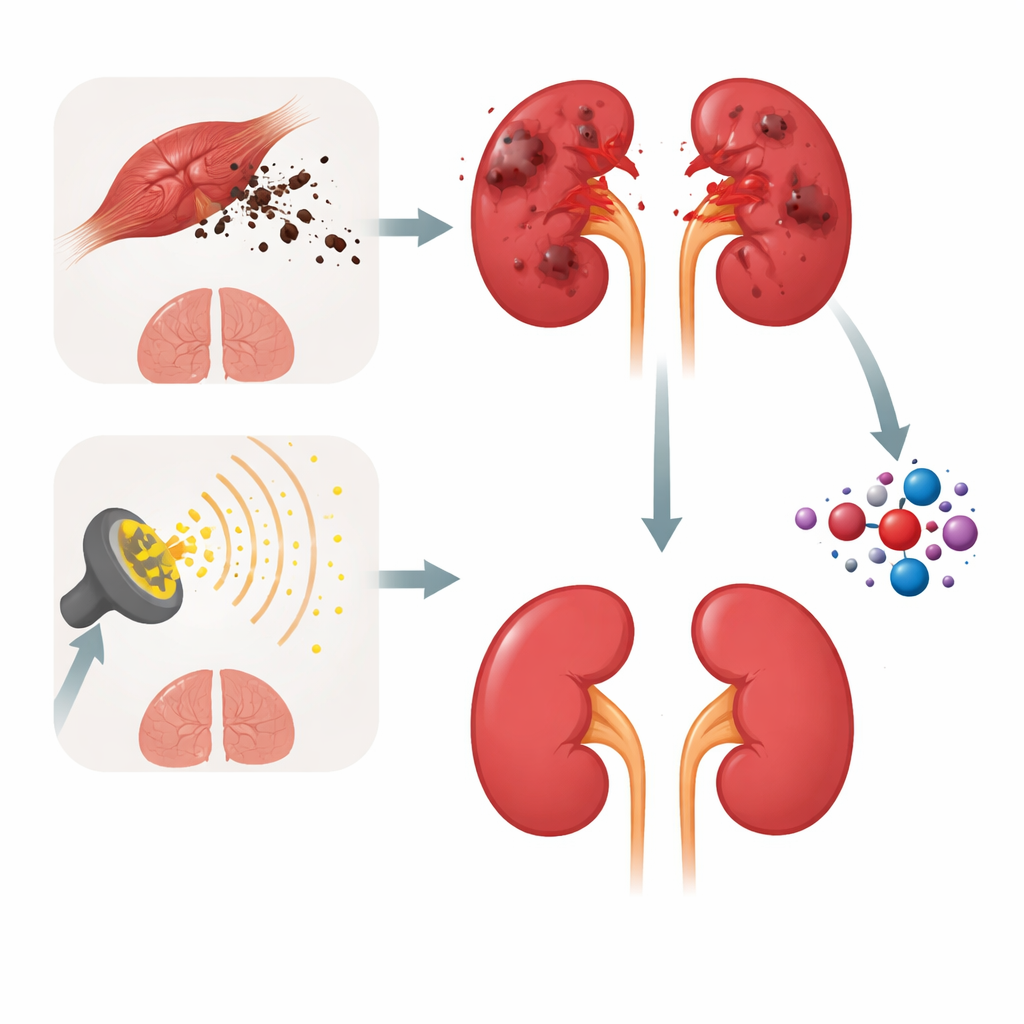
Two Ways to Hurt the Same Organ
The researchers used two established experimental routes to damage rat kidneys. In one, a strong dose of glycerol caused severe muscle breakdown, flooding the bloodstream with muscle pigments and other compounds that clog and poison the kidney’s fine tubules. In the other, the animals received several smaller doses of whole-body gamma radiation, mimicking the fractionated exposures used in radiotherapy. Although these injuries originate in very different places—skeletal muscle versus radiation beams—they converge on the same outcome: inflamed, poorly functioning kidneys, with rising blood urea and creatinine levels and clear signs of structural damage under the microscope.
A Simple Molecule from the Gut
Sodium propionate is normally produced by gut bacteria as they digest dietary fiber. It has been linked to lower inflammation and better antioxidant defenses in various tissues, but its role in kidney protection and, in particular, in the clean-up of damaged mitochondria had not been tested in these types of acute injury. Here, rats received sodium propionate by mouth either during or after exposure to glycerol or gamma rays. The treatment dose was modest and given for two weeks, mimicking a practical supplementation schedule rather than a one-time experimental injection.
Calming Oxidative Fire in Kidney Cells
Both glycerol and radiation flooded kidney cells with reactive oxygen species—highly reactive molecules that damage fats, proteins, and DNA. The team measured several footprints of this oxidative fire: malondialdehyde and lipofuscin (products of damaged fats), protein carbonyls (scarred proteins), and a key repair enzyme called MSRA. Injured rats showed higher levels of damage markers and depleted antioxidant defenses. Sodium propionate treatment reversed much of this pattern. Damage markers dropped, while protective molecules such as glutathione and MSRA rebounded, indicating that the kidneys’ internal chemistry was shifting back toward balance. Under the microscope, kidneys from treated rats showed fewer dead tubular cells, less distortion of the filtering structures, and far less congestion and scarring.
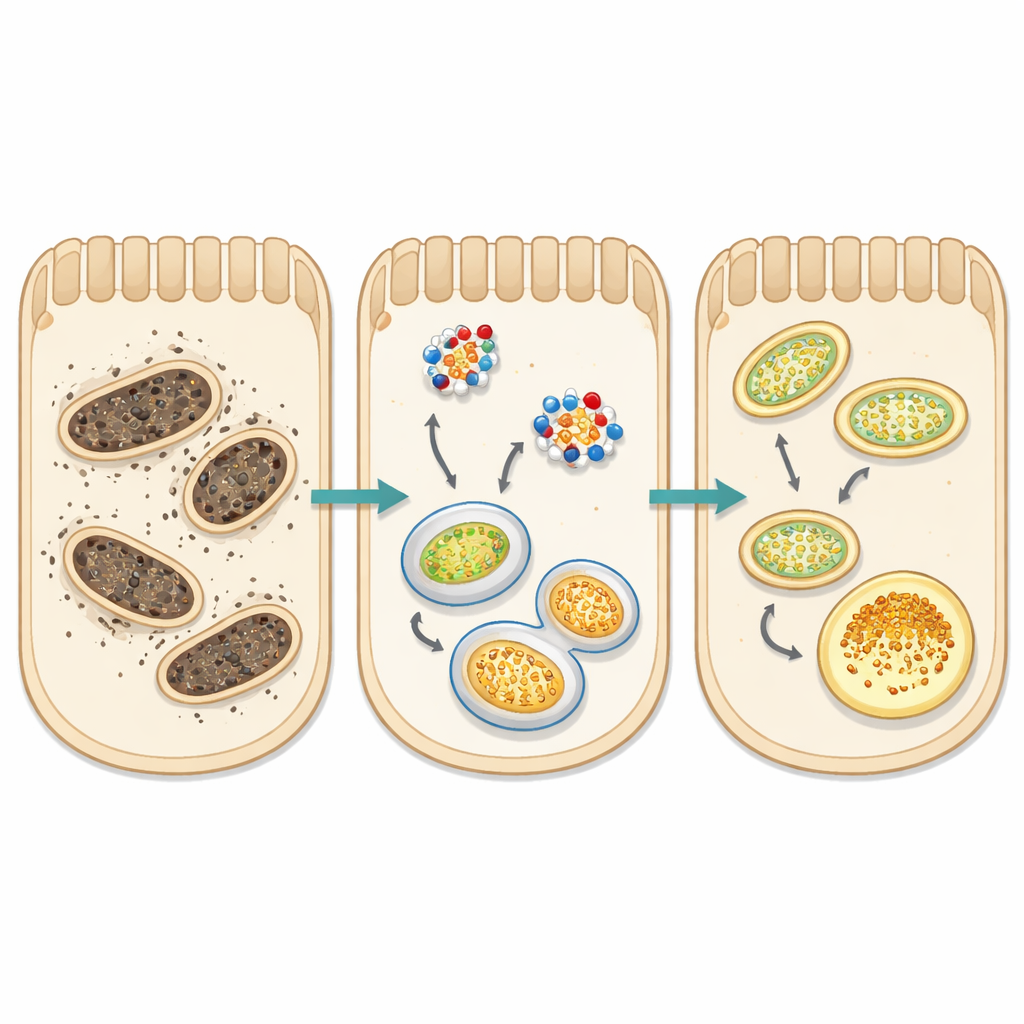
Helping Cells Recycle Their Powerhouses
A central focus of the study was mitophagy—the cell’s selective recycling system for worn-out mitochondria, the energy-producing “powerhouses.” In healthy cells, damaged mitochondria are tagged by proteins such as PINK1, wrapped in membranes marked by LC3, and delivered to cellular recycling centers. In both injury models, this process was disrupted: PINK1 levels dropped, the LC3 balance suggested stalled autophagy, and an adaptor protein called p62 accumulated instead of being cleared. At the same time, levels of ATF5, a stress signal linked to overloaded mitochondria, rose sharply. Sodium propionate largely normalized these signals: it boosted PINK1, restored LC3 balance, reduced p62 build-up, and lowered ATF5. Together with reduced oxidative stress, these changes point to a revived mitochondrial quality-control system, allowing kidney cells to remove damaged powerhouses before they trigger further harm.
What This Could Mean for Patients
To a non-specialist, the key message is that a small, naturally occurring molecule related to fiber digestion helped rat kidneys withstand two very different types of acute attack. By both cooling oxidative stress and restarting the cell’s internal recycling of damaged mitochondria, sodium propionate preserved kidney structure and function in settings that mimic toxin-induced muscle breakdown and radiation exposure. While these results are still in animals and at an early stage, they provide a mechanistic roadmap for exploring sodium propionate, or similar short-chain fatty acids, as supportive therapies to reduce kidney damage in high-risk clinical situations, such as intensive care, major trauma, or cancer radiotherapy.
Citation: Habieb, M.E., Ali, M.M., Abd-ElRaouf, A. et al. Protective role of sodium propionate against glycerol or fractionated doses of gamma rays-induced acute kidney injury via ATF5-induced mitophagy in rats. Sci Rep 16, 12073 (2026). https://doi.org/10.1038/s41598-026-46553-3
Keywords: acute kidney injury, sodium propionate, oxidative stress, mitophagy, gamma radiation