Clear Sky Science · en
Tellurium-118 as a novel radionuclide for long-term positron emission tomography
Watching Medicine Work Over Time
Modern medical scans can show where drugs and disease-fighting molecules travel inside the body, but most current tracers fade within hours. That makes it hard to follow slow-acting treatments like antibody drugs, which linger in the body for weeks. This study explores a new radioactive element, tellurium-118, that could let doctors and scientists watch such treatments over much longer periods using positron emission tomography (PET) scans.
Why Standard Scans Are Not Enough
PET scanners detect tiny bursts of energy created when special radioactive tracers break down inside the body. These tracers are attached to molecules that seek out tumors or other targets, allowing doctors to see where those molecules go. The problem is that the most common PET tracers lose their radioactivity quickly, often within a few hours. Many new cancer therapies, especially antibody-based drugs, move slowly and stay in the body for weeks. With short-lived tracers, important parts of their journey remain invisible, making it harder to predict how well a treatment will work or where side effects might appear.
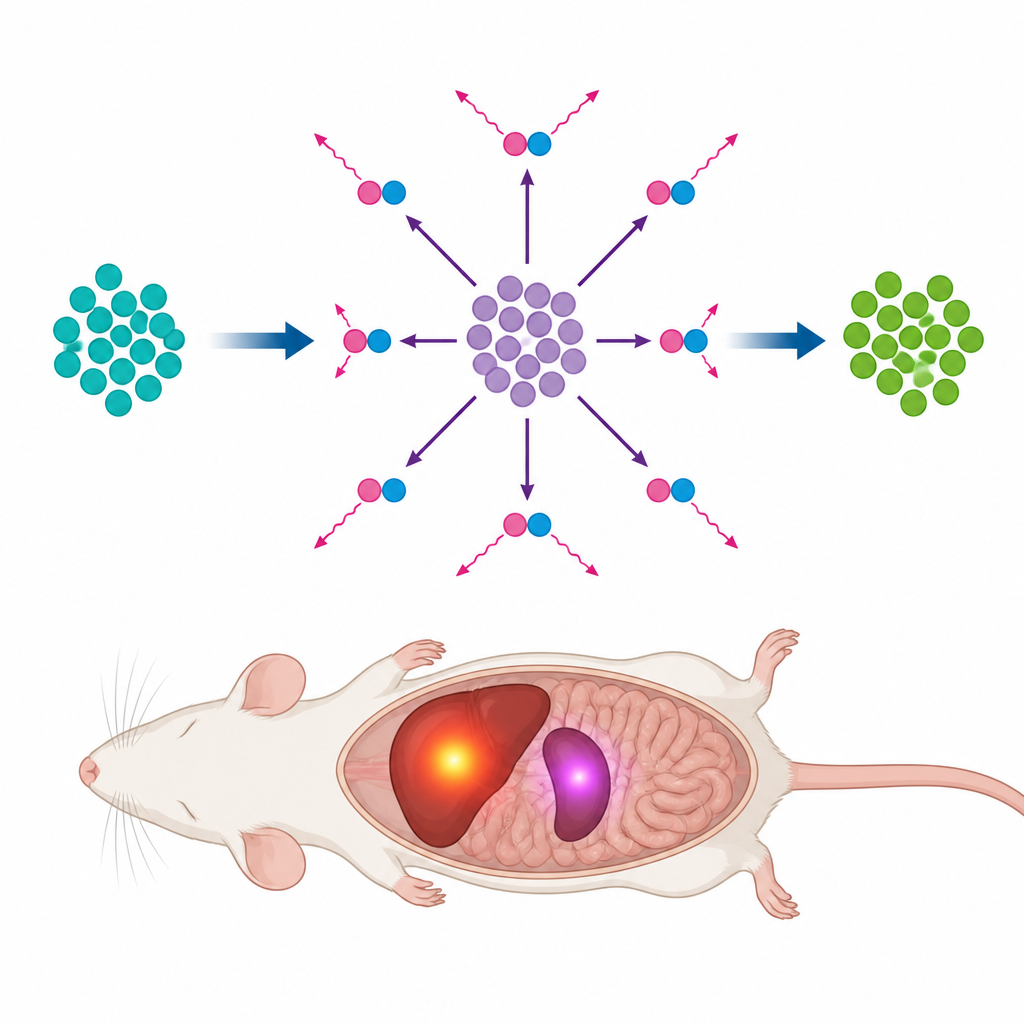
A New Long-Lasting Tracer Idea
The researchers turned to an uncommon element, tellurium-118, which stays radioactive for about six days. On its own it does not emit the signals PET scanners see. Instead, it quietly transforms into another element, antimony-118, which does emit the needed signals but only for a few minutes before turning into a stable, non-radioactive form. Because tellurium-118 keeps feeding this short-lived antimony inside the body, a single dose behaves like a tiny built-in generator that produces PET signals over many days without the extra high-energy radiation that can blur images or increase unwanted exposure.
Testing Image Quality in the Lab
To find out whether this new tracer could produce usable pictures, the team first tested it with plastic “phantoms,” stand-ins for the body that contain small holes and patterns used to judge image sharpness. They filled these phantoms with a solution containing tellurium-118 and scanned them for several hours. The images clearly showed structures a few millimeters wide, although fine details looked a bit fuzzy. This blur likely comes from the relatively energetic particles released by antimony-118, which travel farther before creating the signal that the scanner detects. Even so, the level of sharpness was judged good enough for tracking where tracers go in both small animals and, potentially, people.
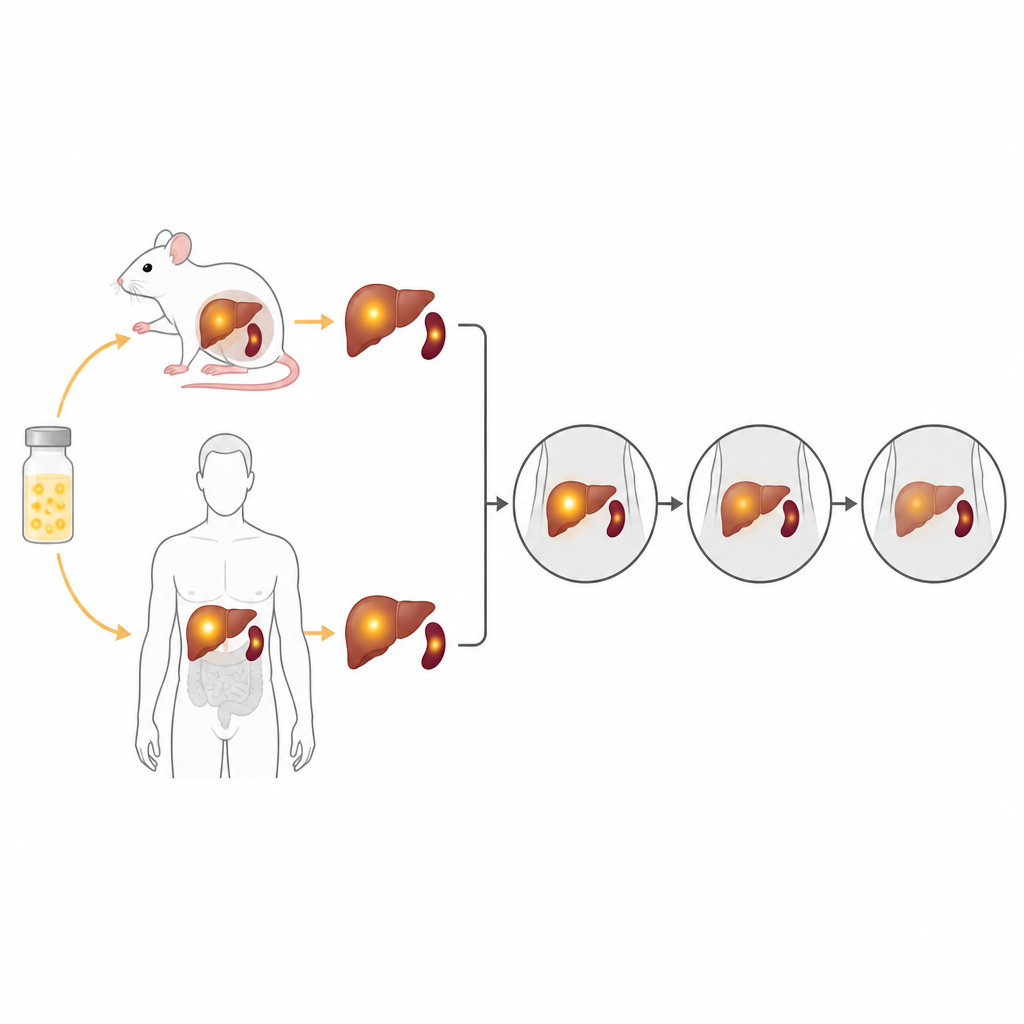
Following the Tracer Inside Mice
The next step was to see how the tracer behaved in living animals. Mice were injected with the tellurium-118 solution and then scanned one, two, and three weeks later using combined PET and CT imaging. Strong signals remained visible in the abdominal area for the full three-week period. After scanning, the scientists measured radioactivity directly in the organs. They found that most of the tracer settled in the liver and spleen and stayed there over time, with only tiny amounts in blood and other tissues. These findings matched the scan images and suggest that the decay from tellurium-118 to antimony-118 does not drastically change where the tracer ends up in the body.
What This Could Mean for Future Care
The work shows that tellurium-118 can act as a long-lasting source of PET signal from a single injection, opening the door to imaging that spans weeks instead of hours or days. There are still hurdles, including refining the chemistry needed to attach this element to specific drugs, checking safety over longer periods, and comparing it carefully with other long-lived tracers. If these challenges are met, this new approach could help doctors watch slow-moving therapies more closely, better understand how radiation-based treatments behave in the body, and fine-tune doses to balance benefit and risk for each patient.
Citation: Miyao, S., Momose, T., Kawabata, M. et al. Tellurium-118 as a novel radionuclide for long-term positron emission tomography. Sci Rep 16, 13909 (2026). https://doi.org/10.1038/s41598-026-46505-x
Keywords: positron emission tomography, long-lived radionuclide, tellurium-118, molecular imaging, radiotheranostics