Clear Sky Science · en
Targeting STAT3 pathway attenuates macrophages inflammation and cardiovascular injury in a model of Kawasaki disease
Why a childhood heart illness matters
Kawasaki disease is a sudden inflammatory illness that mainly affects children under five years old and can leave lasting scars on the heart’s blood vessels. Even with today’s standard treatment of antibodies (IVIG) and aspirin, some children still go on to develop bulges or narrowing in their coronary arteries, raising their risk of heart attacks later in life. This study explores a new way to calm the overactive immune cells that attack blood vessels in Kawasaki disease, pointing toward a possible future medicine that could better protect children’s hearts.
The immune cells that crowd heart vessels
Doctors have long known that Kawasaki disease is driven by runaway inflammation in blood vessels, especially the coronary arteries that feed the heart muscle. When researchers examine heart tissue from affected patients or from mouse models that mimic the disease, they find large numbers of macrophages—immune cells that normally help clear infections—clustered in and around the vessel wall. In Kawasaki disease, many of these macrophages switch into a highly aggressive state, releasing torrents of inflammatory molecules that weaken and damage the vessel. The authors used a well-established mouse model, triggered by a bacterial cell-wall extract called LCWE, to reproduce these vessel changes and study how macrophages contribute to the damage.
A molecular switch that turns inflammation up
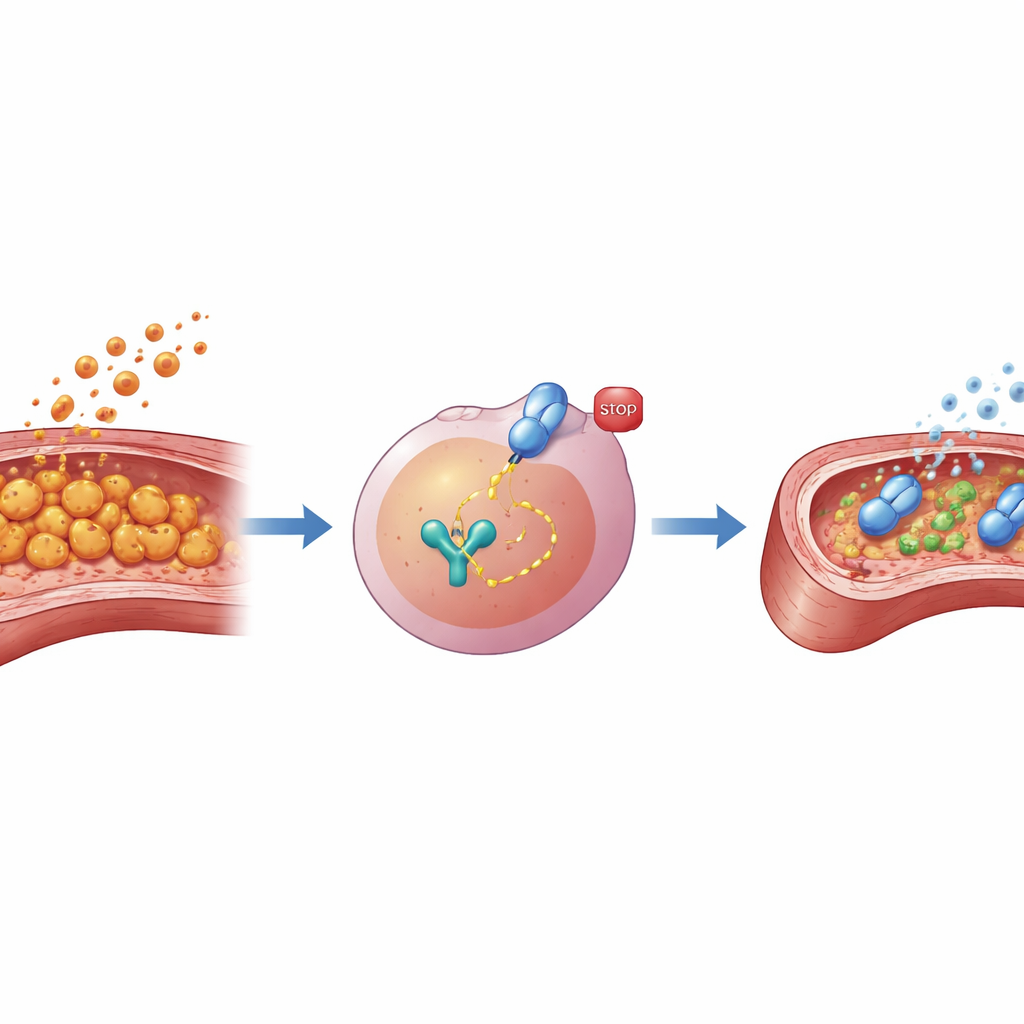
Inside macrophages, a protein called STAT3 acts like a switchboard operator, turning on genes that drive inflammation. The team first asked whether this switch is flipped on in their Kawasaki-like model. In cultured mouse macrophages exposed to LCWE, they found that the activated, phosphorylated form of STAT3 rose sharply over time, while the total amount of STAT3 protein stayed the same. Similar activation appeared in heart tissue from LCWE-treated mice, where vessel inflammation and thickening were pronounced and levels of several inflammatory signals were high. These findings suggested that STAT3 is not just present but actively pushing macrophages toward a damaging, overexcited state in this disease model.
Turning down the signal to protect vessels
To test whether blocking STAT3 could rein in this harmful response, the researchers used AG490, a small molecule that prevents STAT3 from becoming activated. In both a macrophage cell line and primary macrophages taken from mice, pretreatment with AG490 before LCWE exposure sharply reduced the cells’ output of key inflammatory factors, including well-known cytokines and enzymes that can injure tissue or break down vessel structure. When the team collected the “soup” of substances released by these macrophages and applied it to mouse coronary artery endothelial cells—the cells that line the inside of heart vessels—the difference was striking. Medium from untreated, LCWE-stimulated macrophages reduced endothelial cell survival and boosted markers of injury, whereas medium from AG490-treated macrophages caused less damage, suggesting that dialing down STAT3 in macrophages indirectly shields the vessel lining.
From cells in a dish to hearts in living mice
The group then moved to living animals to see whether STAT3 blockade could ease heart vessel damage in the full Kawasaki-like syndrome. Mice were injected with LCWE to trigger vasculitis and treated for two weeks with either AG490 or a neutral vehicle. By four weeks after LCWE exposure, vehicle-treated mice showed dense immune cell infiltrates and thickened coronary arteries, along with elevated levels of inflammatory proteins in heart tissue. In contrast, mice receiving AG490 had milder vessel inflammation, thinner arterial walls and lower levels of the same inflammatory signals. Importantly, additional tests suggested that AG490’s protective effect worked mainly through macrophages, rather than directly acting on endothelial cells, underscoring the central role of these immune cells and their STAT3 switch in driving vessel injury.
What this could mean for children with Kawasaki disease
Taken together, the results show that STAT3 acts as a key amplifier of macrophage-driven inflammation and heart vessel damage in a mouse model of Kawasaki disease, and that blocking this pathway can lessen both the inflammatory signals and the structural injury to coronary arteries. While AG490 itself targets an upstream protein and may not be the final drug used in patients, the work positions STAT3 as a promising target for future therapies that could complement current treatment and better prevent long-term heart complications. Further studies in animal models and, eventually, in children will be needed to refine safer, more precise STAT3 blockers and to confirm that dampening this pathway can safely reduce Kawasaki-related heart risks.
Citation: Zheng, F., Xu, J., Bi, Y. et al. Targeting STAT3 pathway attenuates macrophages inflammation and cardiovascular injury in a model of Kawasaki disease. Sci Rep 16, 14358 (2026). https://doi.org/10.1038/s41598-026-45051-w
Keywords: Kawasaki disease, macrophages, STAT3, vascular inflammation, pediatric heart disease