Clear Sky Science · en
A positive association between phosphorylated Tau217 (pTau217) and neural correlations is prevented by human leukocyte antigen allele DRB1*13:01
Why early brain changes matter
Many people worry about memory loss and dementia, but the earliest changes in the brain can start decades before any symptoms appear. This study looked at subtle patterns of brain activity together with blood tests, genes, and past viral infections in older women who did not have dementia. The goal was to see whether certain blood markers of Alzheimer’s disease relate to how flexibly brain networks communicate, and whether the immune system can blunt harmful effects long before disease is diagnosed.
A window into the working brain
To examine brain function, the researchers used a technique called magnetoencephalography, which records tiny magnetic signals from the brain in real time. From these signals they calculated how strongly different brain regions fire together, a measure they call synchronous neural interactions. Healthy brains show a balance: regions can work together when needed but also act independently. When connections become too tightly locked, communication can lose flexibility, which has been linked to lower scores on thinking tests in earlier work. The team studied 348 brain scans from 175 women and paired these data with detailed blood tests and cognitive scores.
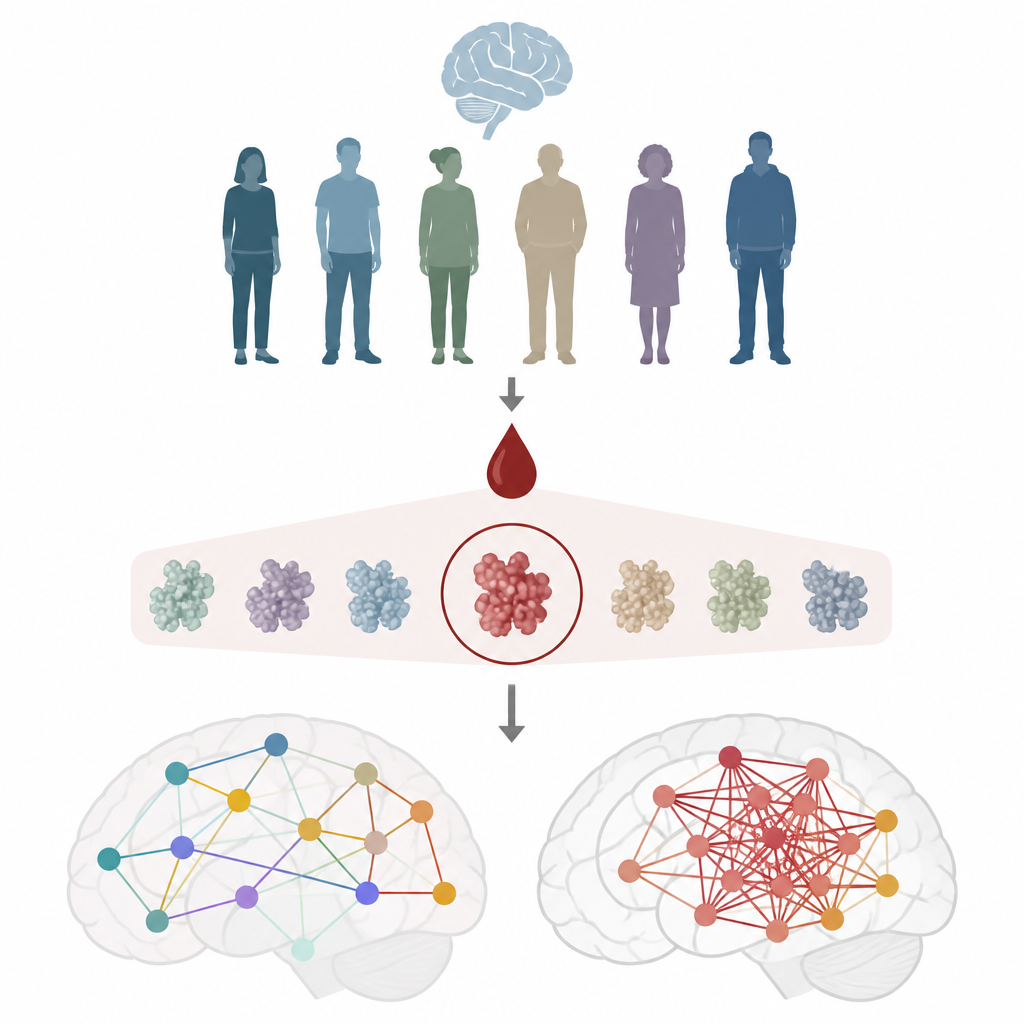
One blood marker stands out
Blood samples were tested for seven proteins tied to Alzheimer’s disease and nerve damage, including different forms of amyloid and tau, as well as a nerve fiber protein. Only one marker, a modified form of tau called pTau217, consistently tracked with brain network changes. Higher pTau217 levels were linked to stronger, more rigid brain correlations and to slightly worse performance on a standard thinking test, even though participants were generally cognitively healthy. Other popular markers, such as amyloid ratios or total tau, did not show this clear relationship with brain activity, underscoring pTau217’s special connection to early brain dysfunction.
Viruses and a protective immune shield
The story became more complex when the team considered past infections and immune genes. Many common viruses can reach the brain and trigger inflammation. Women who had antibodies indicating prior exposure to herpes virus type 1 or to a group of ancient viral elements called HERVK showed a stronger link between pTau217 and stiffened brain networks. In other words, in those with a history of these infections, rising pTau217 was more strongly tied to less flexible brain activity. Yet this pattern largely disappeared in women carrying a particular immune gene variant known as HLA DRB1*13:01. Computer modeling suggested that this gene can bind pieces of the viral proteins especially well, hinting that it may help the immune system clear or control these viral influences.
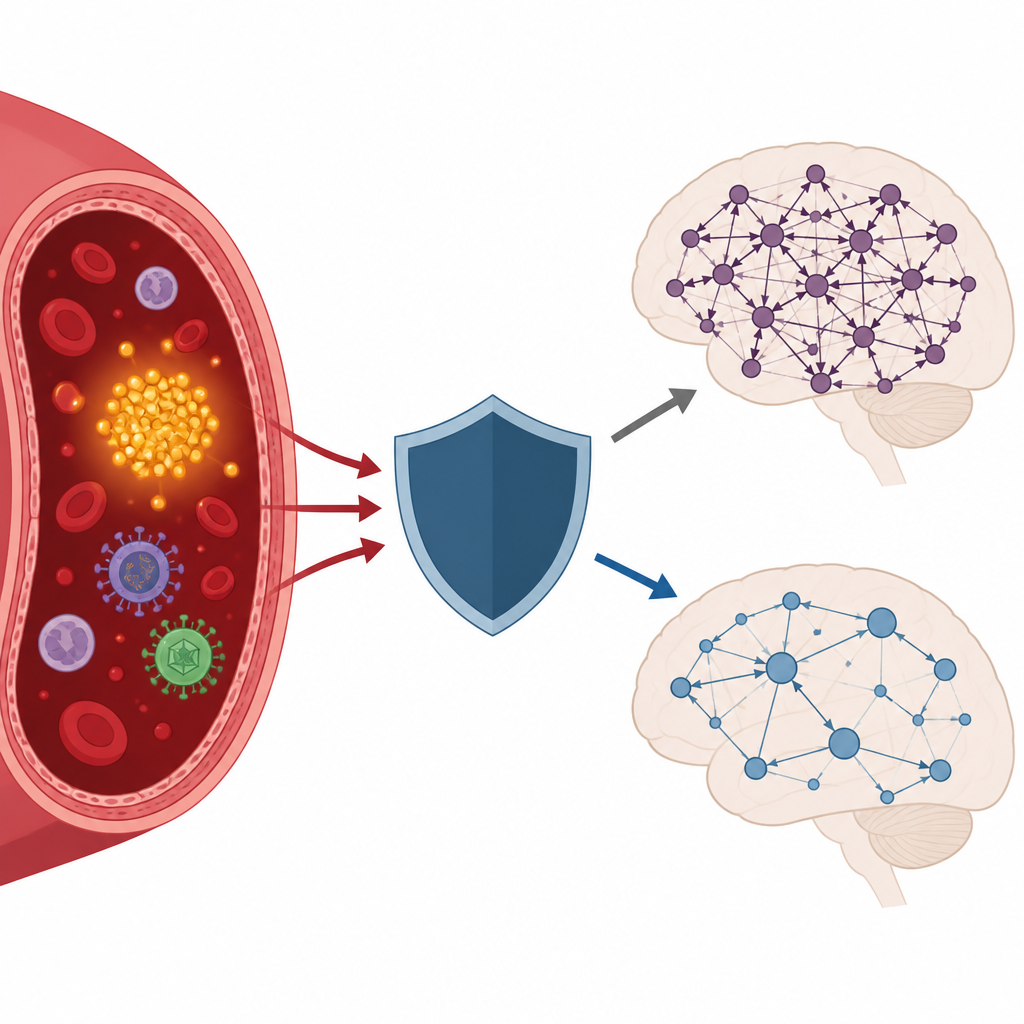
Genes that hurt and genes that help
The researchers also tested another well-known risk gene for Alzheimer’s disease, ApoE. Unlike the HLA gene, ApoE did not change the relationship between pTau217 and brain network measures in this group. This suggests that, at least at these early stages, immune genes that shape how the body handles viral traces may play a more direct role in protecting brain communication than ApoE status alone. Importantly, when either of the two related HLA variants DRB1*13:01 or DRB1*13:02 was present, the harmful combined effect of high pTau217 and rigid brain networks on thinking scores was no longer evident.
What this means for brain health
Taken together, the findings suggest that blood levels of pTau217 reflect subtle disruptions in how brain networks work in everyday, non-demented women, and that these disruptions are linked to slightly poorer thinking ability. Past exposure to certain viruses appears to intensify this link, while specific immune genes can almost switch it off, acting like a protective shield for the brain. For a lay reader, the key message is that dementia risk may emerge from a long-term interplay between infection history, blood markers of nerve stress, and the body’s immune defenses. Understanding this balance could one day help identify people at higher risk earlier and guide prevention strategies tailored to their immune and infection profiles.
Citation: James, L.M., Stratigopoulos, G., Leuthold, A.C. et al. A positive association between phosphorylated Tau217 (pTau217) and neural correlations is prevented by human leukocyte antigen allele DRB1*13:01. Sci Rep 16, 15026 (2026). https://doi.org/10.1038/s41598-026-44894-7
Keywords: pTau217, brain networks, Alzheimer’s risk, HLA DRB1*13:01, viral infections