Clear Sky Science · en
Edelfosine induces cell cycle arrest and apoptosis in vascular smooth muscle cells to suppress neointimal hyperplasia
Why clogged arteries matter
When an artery is opened with a balloon or stent, the goal is to restore blood flow to the heart or brain. But the vessel often responds by growing a new inner layer of muscle and scar tissue that slowly reclogs the passage. This study explores whether a drug called edelfosine, originally designed to kill cancer cells, can instead be used locally on blood vessels to curb this overgrowth and keep arteries open longer.
A closer look at vessel wall overgrowth
After procedures like angioplasty or stenting, smooth muscle cells in the vessel wall can switch into a growth mode. They multiply and move inward, building a thick layer called neointima that narrows the channel where blood should flow. Current stents release drugs that slow cell growth, but they do not always fully prevent this problem and can affect other cell types. The authors reasoned that a drug able to selectively push overactive smooth muscle cells into a controlled death program might offer a stronger and more focused way to prevent re-narrowing.
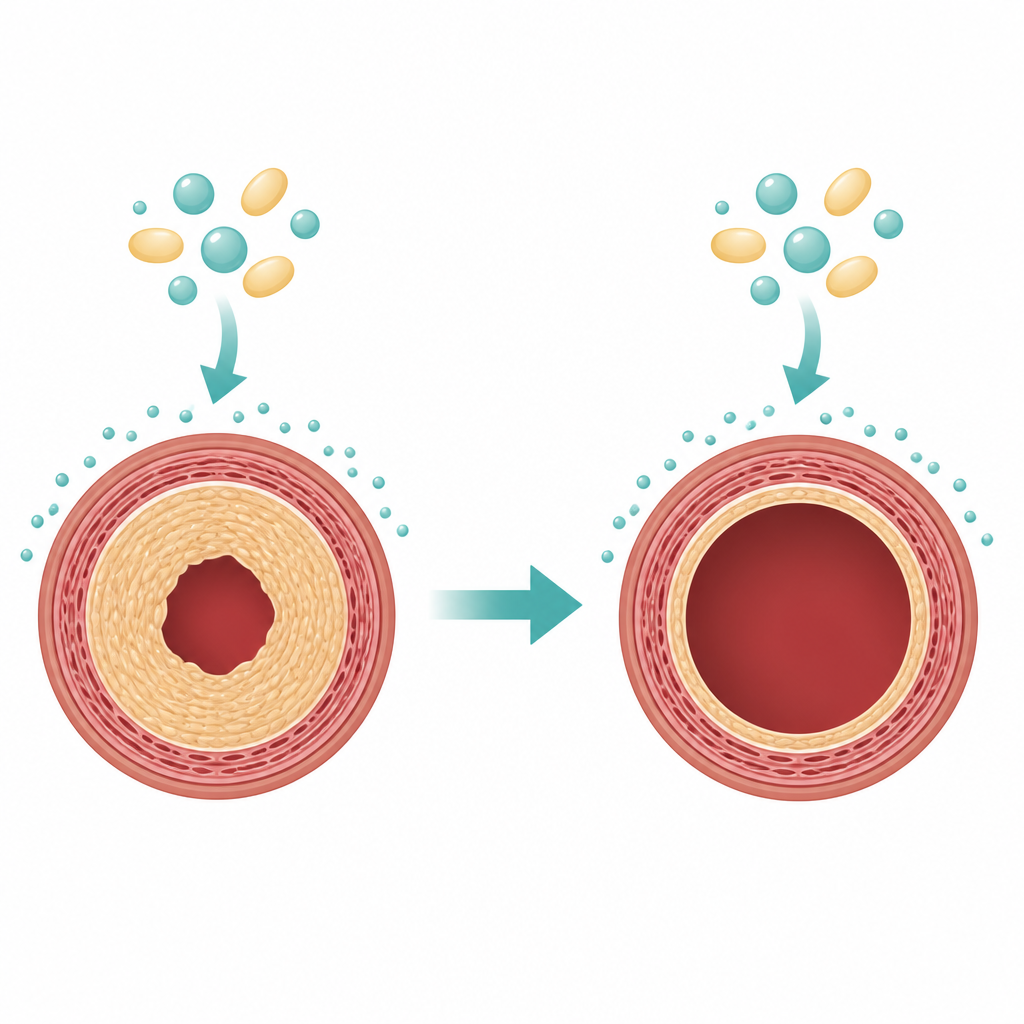
Borrowing a cancer drug for blood vessels
Edelfosine is a synthetic fat-like molecule that slips into cell membranes and is known to trigger self-destruct programs in rapidly dividing cancer cells while sparing many resting cells. The team tested edelfosine on smooth muscle cells taken from rat and mouse arteries and grown in dishes. At moderate doses, the drug sharply reduced how many cells stayed alive and how much new DNA they made, a sign that they had stopped dividing. These effects were stronger when growth factors from serum were absent, suggesting edelfosine hits especially hard when cells rely on active growth signals.
How edelfosine halts and removes problem cells
To find out what happens inside the cells, the researchers tracked their progress through the cell cycle, the stages a cell passes through as it prepares to divide. With edelfosine, many cells piled up in the stage just before and during division and often failed to split properly, ending up with two nuclei. This pointed to a roadblock at the division checkpoint. At the same time, molecular markers of a stress response in an internal structure called the endoplasmic reticulum increased, and key "executioner" proteins that drive programmed cell death were switched on. Blocking these death enzymes rescued many cells, confirming that edelfosine was not just slowing growth but actively triggering a tidy form of cell suicide.
The role of calcium inside the cells
The study also examined the flow of calcium ions, which act as powerful internal signals. Using fluorescent dyes, the team showed that edelfosine caused a rapid rise in calcium levels inside smooth muscle cells. Drugs that blocked calcium entry from outside the cell or interfered with calcium release from internal stores reduced this rise and partly lessened the early wave of cell death. This suggests that disturbed calcium handling, linked to internal stress, is one of the early steps by which edelfosine pushes these cells toward shutdown and removal.
Testing the approach in live arteries
To see if these lab findings matter in a living body, the researchers used a mouse model in which tying off a carotid artery reliably causes the inner layer to thicken. They wrapped the injured artery with a hydrogel containing edelfosine or a harmless vehicle. After two weeks, arteries treated with edelfosine showed much thinner inner layers and a wider open channel, while the middle muscle layer remained intact. Staining for fragmented DNA revealed more cells undergoing programmed death in the vessel wall of treated animals, consistent with the dish experiments and without signs of obvious vessel damage or clot formation in this short-term study.
What this could mean for patients
The work suggests that edelfosine can both stop overactive smooth muscle cells from dividing and nudge them into a controlled self-destruct process, easing the thickening that threatens to reclog treated arteries. By acting through membrane and internal stress pathways rather than classic cell division targets, it differs from drugs now used in drug-coated stents and may spare more of the surrounding healthy tissue. While much remains to be tested, especially in human vessels and over longer periods, the findings raise the possibility that a cancer-inspired lipid drug could one day help keep reopened arteries clearer for longer by pruning back the very cells that cause them to narrow again.
Citation: Sun, J., Gui, Y., Liu, Y. et al. Edelfosine induces cell cycle arrest and apoptosis in vascular smooth muscle cells to suppress neointimal hyperplasia. Sci Rep 16, 14944 (2026). https://doi.org/10.1038/s41598-026-44632-z
Keywords: edelfosine, vascular smooth muscle, neointimal hyperplasia, restenosis, apoptosis