Clear Sky Science · en
Microfluidic process-property correlations of dsRNA lipid nanoparticle formulations
New tools for safer crop protection
Farmers around the world struggle to protect crops from insects without harming bees, butterflies, and other helpful creatures. This study explores a new way to safeguard plants using a natural gene-silencing signal, wrapped in tiny fat-based particles so it can survive long enough to work in the field. The goal is a spray that targets only harmful insects, reduces reliance on broad poisons, and can be made cheaply at industrial scale.
Why a genetic whisper can replace a chemical blast
Many modern insecticides kill a wide range of species and can linger in soil, water, and food chains. In contrast, RNA interference (RNAi) acts like a genetic whisper: double-stranded RNA (dsRNA) molecules are designed to match a vital gene only in the pest insect. When ingested, they trigger the breakdown of that gene’s messenger, eventually killing the pest while sparing most other species. However, these dsRNA strands are fragile. Sunlight, enzymes on leaf surfaces, and harsh conditions in the insect gut can destroy them within hours. To make RNAi into a practical field spray, the dsRNA must be shielded just long enough to be eaten and released inside insect cells.
Tiny protective shells made from affordable ingredients
Pharmaceutical companies already protect medical RNA with lipid nanoparticles—nanoscale spheres made from fatty molecules. But the specialty lipids used in vaccines are far too expensive for use on large fields. The authors therefore built a “toolbox” of three technical-grade lipids already produced on the ton scale: a positively charged fatty amine to grab the negatively charged dsRNA, a PEG-containing stabilizer to keep particles dispersed, and a lecithin mixture similar to food emulsifiers. Using controlled mixing in narrow microfluidic channels, they tuned how much lipid they added and how fast they mixed, and then measured particle size, surface charge, and uniformity with light-scattering methods and electron microscopy. Faster mixing and more lipid generally yielded smaller, more spherical particles—mostly below 100 nanometers—while too little lipid led to clumps and irregular shapes.
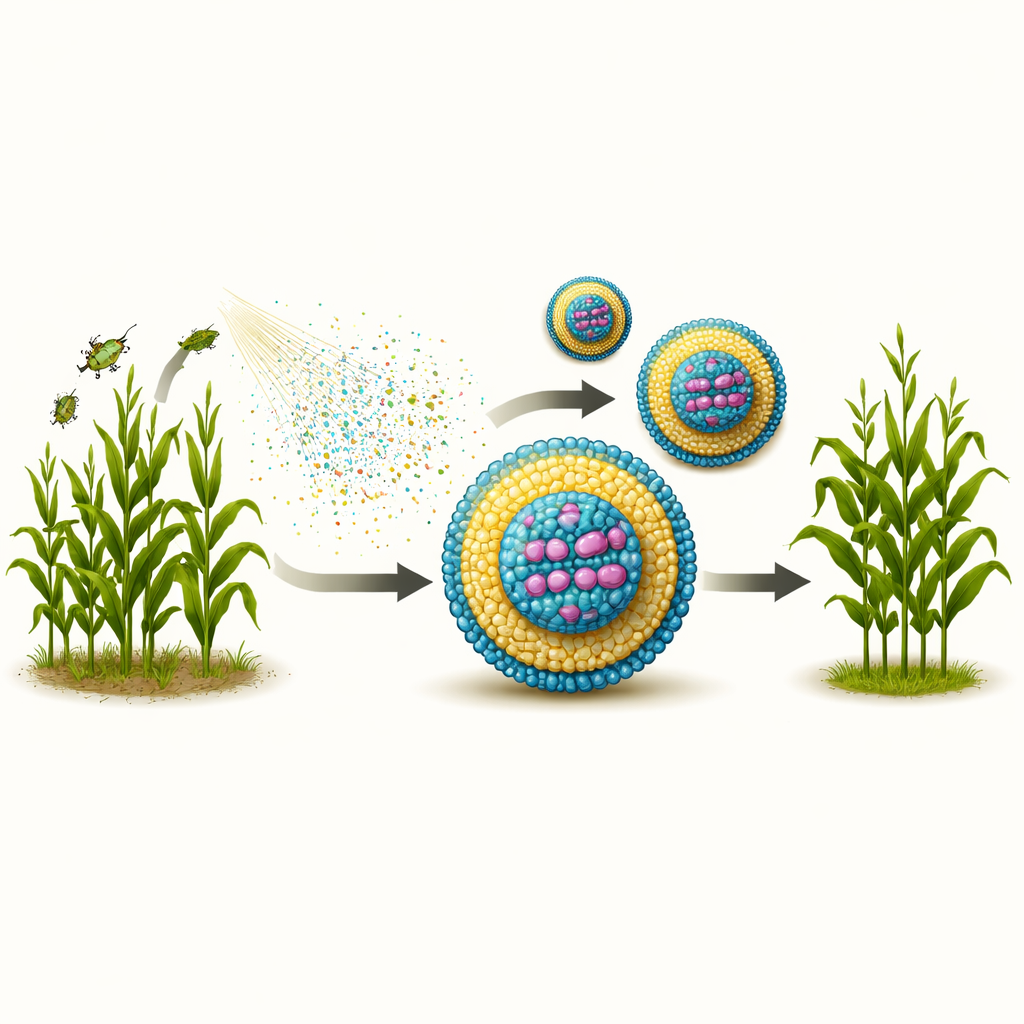
Keeping the genetic message intact in harsh conditions
To test whether these particles truly protect their cargo, the team exposed both bare dsRNA and dsRNA inside lipid nanoparticles to a degrading enzyme (RNase III) and to a wide range of acidity and alkalinity. On gels, unprotected dsRNA essentially vanished after 24 hours with the enzyme or at very low and very high pH. In contrast, all nanoparticle recipes kept the dsRNA intact for at least a day under enzyme attack, and the genetic strands could be recovered by adding a detergent that pulls apart the particles. The formulations also greatly improved dsRNA survival in acidic conditions, similar to those found in the guts of important beetle pests. At extremely alkaline conditions, the particles lost their stabilizing charge and precipitated, so protection was limited, but these extremes are less relevant for typical field use and insect digestion.
From lab bench trickle to farm-scale flow
Protection alone is not enough; any practical solution must be manufactured in large volumes at low cost. The researchers therefore adapted their microfluidic process to run at much higher flow rates, producing hundreds of milliliters of formulation in a single run—an important step toward pilot-scale batches. By systematically varying the proportions of the three lipids while keeping the overall lipid-to-dsRNA ratio constant, they mapped how recipe changes affected particle size and surface charge. They then used a calorimetry technique to examine how readily a detergent could pull dsRNA back out of the particles. The energy measurements showed that release was moderate and driven largely by disordering of the lipid–water system, suggesting that dsRNA can be firmly protected yet still liberated under the right conditions.
Toward smarter, targeted pest control
In plain terms, this work shows that it is possible to build small, well-behaved protective shells around gene-silencing signals using inexpensive, scalable ingredients and high-throughput mixing methods. These particles keep the dsRNA message safe from enzymes and harsh acidity long enough to reach insect pests, while still allowing its release when triggered. Although further refinements are needed to reduce particle clumping and test performance on real crops and insects, the study lays essential groundwork for field-ready RNA sprays that could one day replace many broad-spectrum insecticides with a more precise and environmentally friendly tool.
Citation: Geisler, P., Knorr, E., Steiniger, F. et al. Microfluidic process-property correlations of dsRNA lipid nanoparticle formulations. Sci Rep 16, 9653 (2026). https://doi.org/10.1038/s41598-026-44095-2
Keywords: RNA interference, lipid nanoparticles, biopesticide, microfluidics, sustainable agriculture