Clear Sky Science · en
TCF4 trinucleotide repeat expansion drives distinct proteomic signatures in Fuchs endothelial corneal dystrophy
Why this eye disease matters
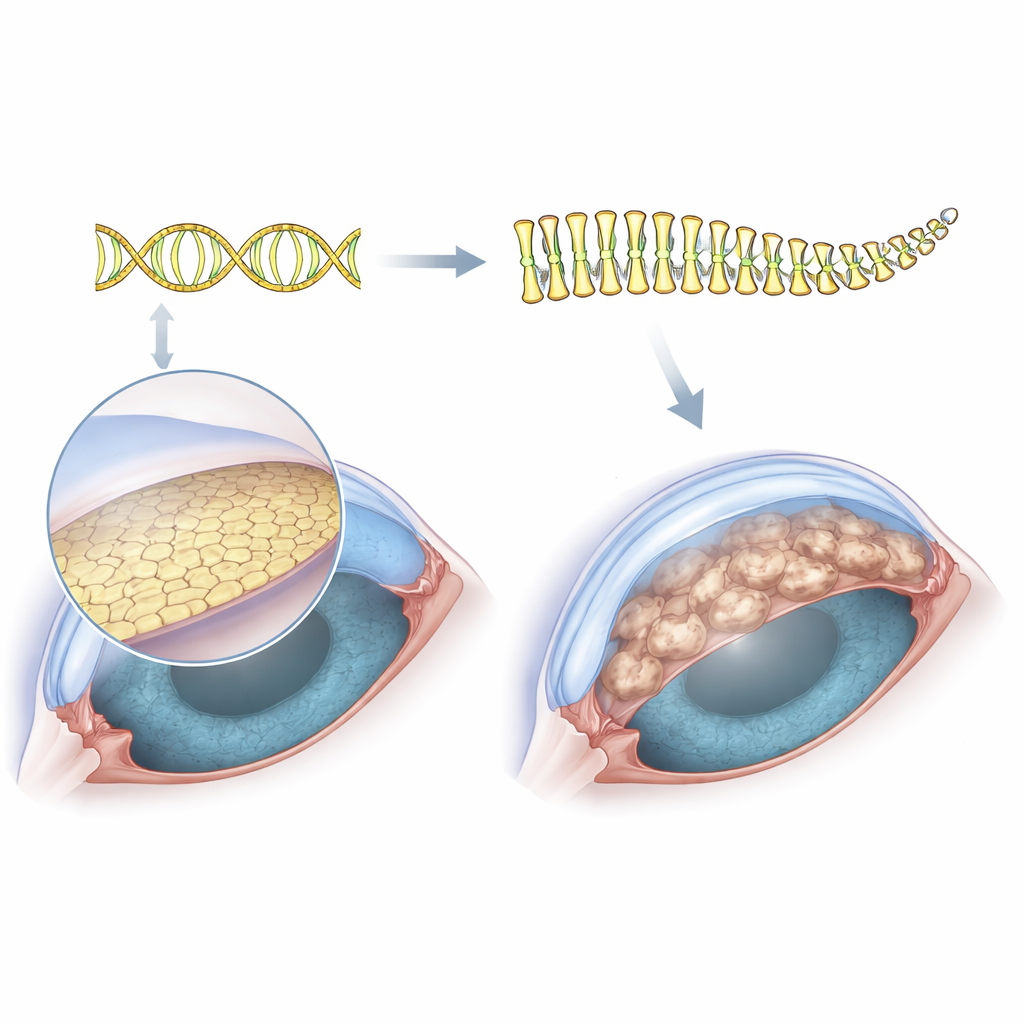
As people age, one of the most common reasons to need a corneal transplant is a condition called Fuchs endothelial corneal dystrophy. In this disease, the inner surface of the cornea—normally a clear, smooth window—develops tiny bumps and swelling that cloud vision. Doctors have long known that a stretch of repeated DNA code in a gene named TCF4 strongly raises the risk of Fuchs, but they have not understood how this extra genetic “stutter” actually damages the cells that keep the cornea clear. This study tackles that question by looking directly at the proteins inside affected cells, aiming to connect a mysterious DNA change to the everyday workings of the eye.
Looking at the cornea’s hidden workforce
The cornea’s clarity depends on a single layer of cells on its inner surface, the corneal endothelium. These cells sit on a thin support sheet and constantly pump fluid out of the tissue to keep it transparent. In Fuchs dystrophy, that inner sheet becomes studded with wart-like outgrowths called guttae, the cells gradually die off, and the cornea swells and clouds. Because this cell layer is so thin, it has been technically difficult to collect enough material from patients to study all of the proteins they make. To get around this, the researchers used an immortalized cell line from a person with Fuchs that naturally carries the expanded TCF4 DNA repeat, and then used CRISPR gene editing to cut out just that expanded segment, creating an almost perfectly matched “before and after” cell system.
Switching off the repeat and watching proteins change
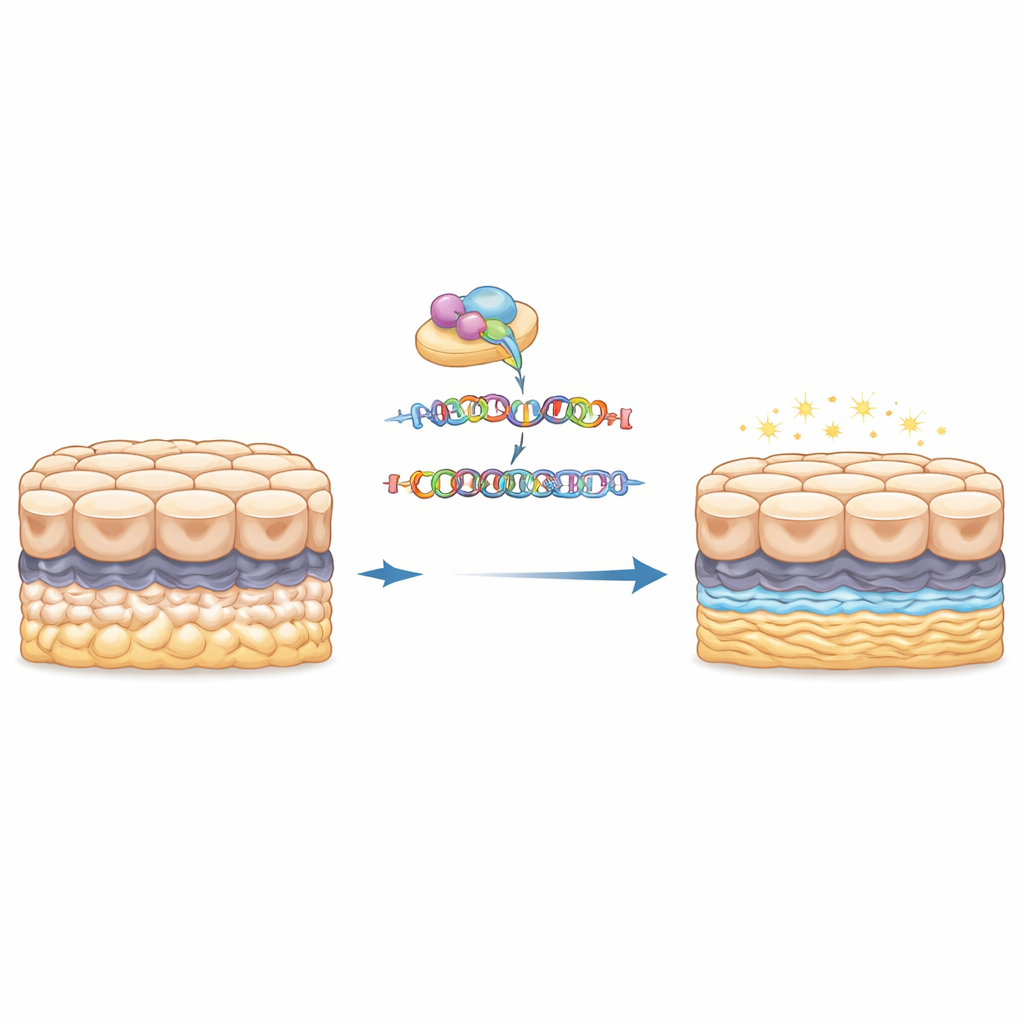
With the expanded DNA repeat removed, the edited cells looked normal under the microscope: they kept their typical polygon shape and formed neat sheets, suggesting that simply erasing the repeat does not harm basic cell structure. The real differences emerged when the team measured thousands of proteins using a sensitive mass spectrometry method that tags and compares samples in parallel. Between the original and the edited cells, 201 proteins changed their abundance in a statistically meaningful way. Some became more common, others less so, but together they formed a distinct molecular fingerprint that clearly separated the “repeat-on” and “repeat-off” states when the data were clustered and visualized.
The cornea’s scaffold and cell “grip” are disrupted
When the researchers grouped these altered proteins by function, two major themes stood out. Many of the proteins that went down after removing the repeat are involved in building and organizing the extracellular matrix—the mesh-like scaffold beneath the cells—and in how cells attach to that scaffold and to each other. These include components of collagen-rich structures and proteins that link cell surfaces to their surroundings. Because excess or abnormal matrix is a hallmark of guttae in Fuchs dystrophy, this pattern suggests that the TCF4 repeat expansion actively drives the overbuilding and mis-shaping of the cornea’s inner surface. The fact that deleting the repeat tones down these matrix and adhesion proteins supports a direct cause-and-effect link between the genetic change and the physical bumps seen in patients’ corneas.
Unexpected signals from the immune alarm system
Surprisingly, many of the proteins that increased after the repeat was removed were tied to interferon signaling and antigen processing, pathways usually associated with antiviral defense and immune surveillance. Proteins such as neuropilin-1, a molecule connected to blood vessel growth and matrix remodeling, and enzymes that help crosslink matrix proteins or regulate the cell cycle, all rose in the edited cells. At the same time, several protective stress-buffering proteins, including a small heat-shock–like protein and a signaling adaptor that helps control cell survival, dropped. Together, these shifts hint that the repeat expansion not only alters the cornea’s physical scaffold but also dampens or reshapes stress and immune-like responses inside the endothelial cells, potentially making them more vulnerable over time.
From DNA repeat to cloudy vision
For a layperson, the key message is that this study draws a clearer line from a small quirk in the TCF4 gene to the clouded cornea seen in Fuchs dystrophy. The extra DNA repeats act like a hidden switch that rewires the protein landscape of corneal endothelial cells, pushing them to lay down too much, or the wrong kind of, supporting material and to change how tightly they cling to their base. Removing that repeat in a controlled cell model largely normalizes those matrix-related proteins and unmask shifts in stress and immune pathways, strengthening the idea that therapies targeting the repeat itself—such as antisense drugs or precise gene-editing tools—could ease both the structural guttae and the functional decline of the corneal endothelium. While further work in patient tissue is needed, this proteomic map brings researchers closer to rational, gene-guided treatments that might someday delay or prevent the need for corneal transplants in people with Fuchs dystrophy.
Citation: Yuasa, T., Nakagawa, T., Honda, T. et al. TCF4 trinucleotide repeat expansion drives distinct proteomic signatures in Fuchs endothelial corneal dystrophy. Sci Rep 16, 14446 (2026). https://doi.org/10.1038/s41598-026-43789-x
Keywords: Fuchs endothelial corneal dystrophy, TCF4 repeat expansion, corneal endothelium, extracellular matrix, proteomics