Clear Sky Science · en
Rheumatic immune-related adverse effects of immune checkpoint treatment: a retrospective multicentre study
Why cancer breakthroughs can bring new joint pains
Immune checkpoint inhibitors are a newer class of cancer drugs that help the body’s own defenses recognize and attack tumors. For many people, they have turned once‑grim diagnoses into longer, better lives. But revving up the immune system also has a downside: in some patients, it begins to attack healthy tissues, including joints, muscles, and blood vessels. This study follows hundreds of people treated at two Spanish hospitals to understand how often these rheumatologic side effects appear, what they look like, and how doctors can best manage them without losing the life‑saving benefits of the cancer treatment.
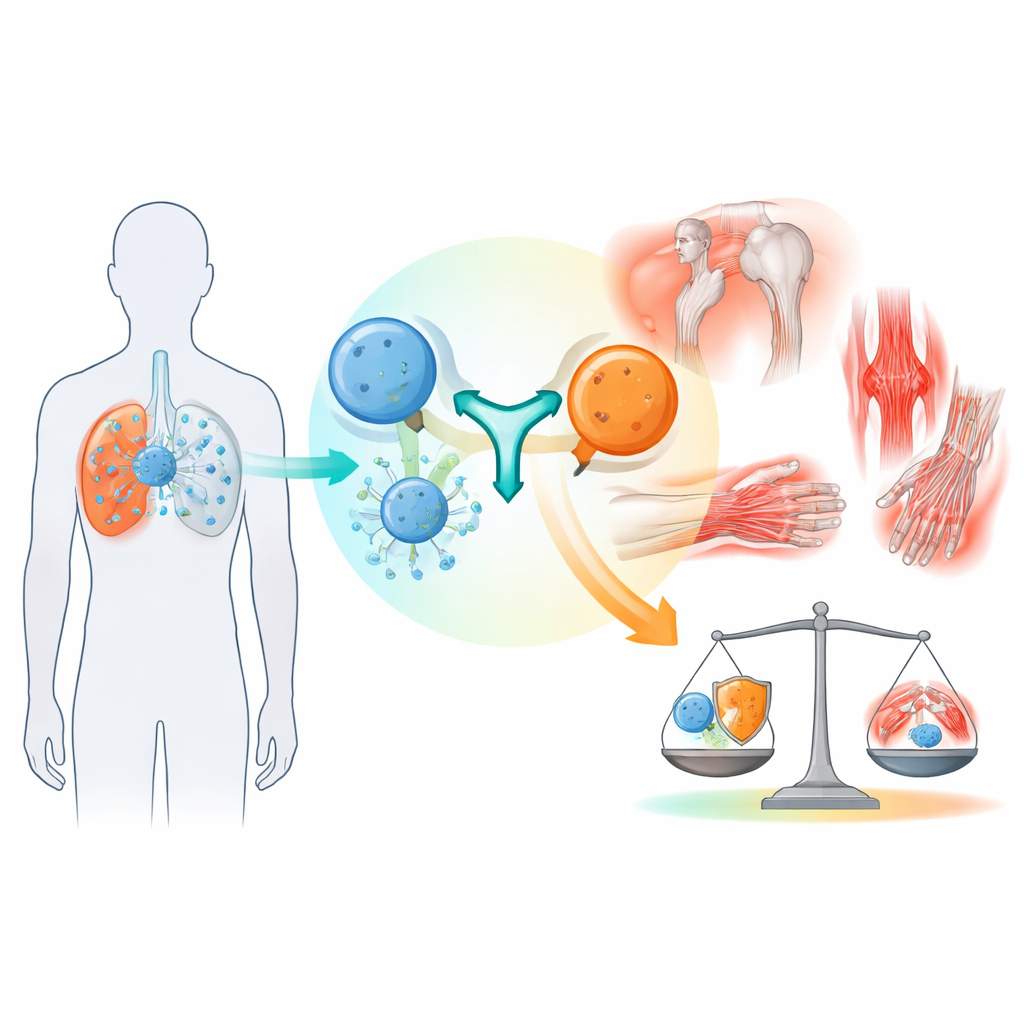
Stronger immunity, new kinds of side effects
The cancer drugs examined in this research block molecular “brakes” that normally keep immune cells from becoming overactive. By lifting these brakes, the drugs can unleash T cells to more effectively destroy cancer. Yet the same unleashed immune response can spill over into normal organs, leading to so‑called immune‑related adverse events, which can affect the skin, gut, hormone‑producing glands, liver, lungs, nerves, and the musculoskeletal system. While rashes and bowel problems are relatively well recognized, joint and muscle complications have been less clearly described and are often harder for oncologists to spot and classify.
Who was studied and what was found
The researchers reviewed the medical records of 734 people who received immune checkpoint drugs between 2016 and 2022. Nearly one in three developed some kind of immune‑related side effect. Among them, 54 patients—about 7% of everyone treated and almost a quarter of those with any immune complication—developed problems involving joints, muscles, or related tissues. Most had lung cancer or melanoma and were receiving drugs that block the PD‑1/PD‑L1 pathway. Only a handful had known autoimmune diseases beforehand, suggesting that serious rheumatic reactions can arise even in people without a prior history of such conditions.
How the body’s support structures were affected
The pattern of symptoms was varied but followed some recurring themes. The most frequent problems were forms of arthritis and joint pain, which together made up around 70% of rheumatic cases. Some patients developed a rheumatoid arthritis‑like illness with multiple joints involved, while others had clear inflammation in joints or more diffuse inflammatory joint pain without obvious swelling. Another common picture resembled polymyalgia rheumatica, a condition that causes aching and stiffness in shoulders and hips, especially in older adults. Less frequent but particularly serious were muscle inflammation and related conditions that can involve weakness, swallowing difficulty, or even the heart, as well as dryness of the eyes and mouth and inflammation of large blood vessels supplying the head and limbs.
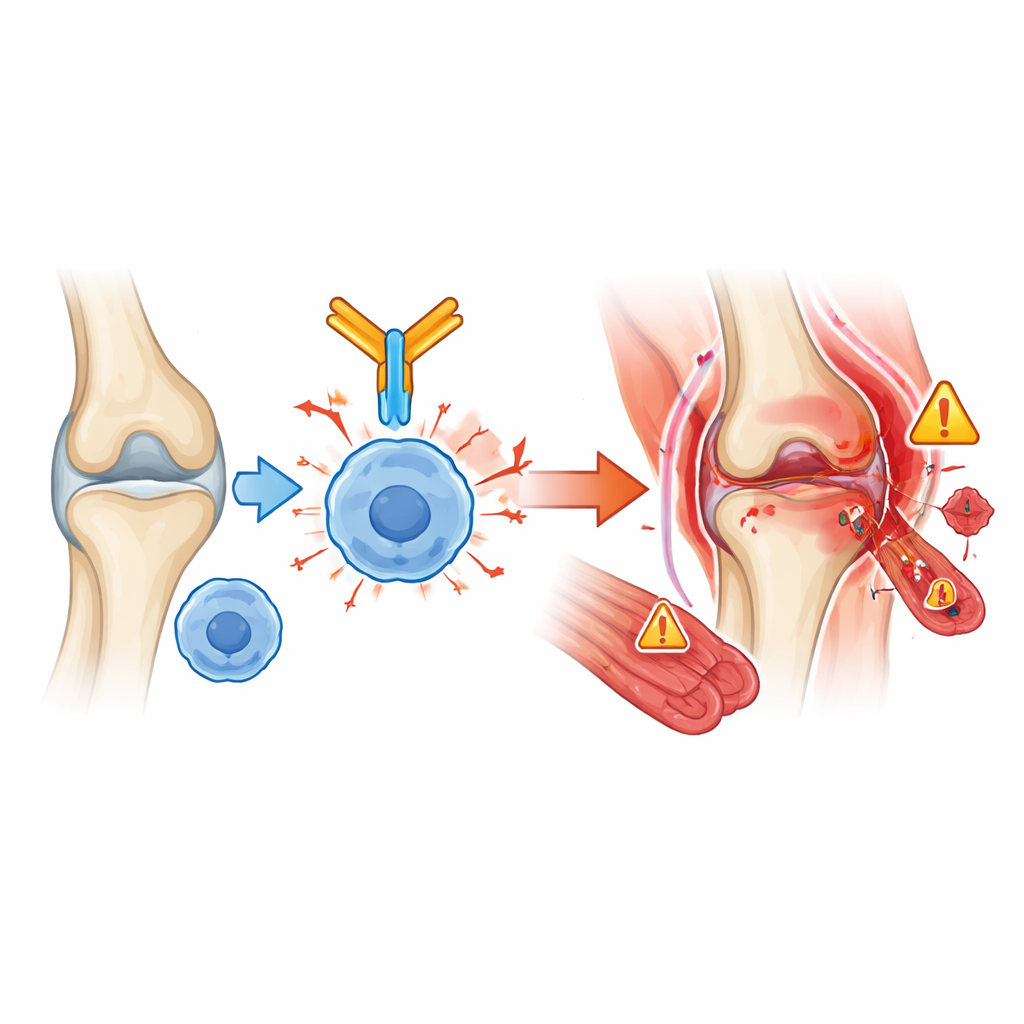
Timing, tests, and treatments
Most joint and muscle problems appeared within six months of starting cancer immunotherapy, but there were exceptions. Muscle inflammation tended to strike early, often within the first few weeks and sometimes alongside nerve‑muscle or heart involvement, making it one of the most dangerous complications. By contrast, dryness of eyes and mouth could arise much later, even nearly two years into treatment. Blood tests, when available, frequently showed signs of general inflammation, and some patients developed new autoantibodies, although these did not always match classic autoimmune diseases. Doctors mainly used corticosteroids—powerful anti‑inflammatory drugs—to treat these reactions. A few patients needed additional immune‑suppressing pills, biologic therapies, or intravenous antibodies, especially in severe muscle disease. While about 70% improved clinically, one in three had to stop their cancer immunotherapy because the rheumatic problems were too severe.
Balancing cancer control and quality of life
For people facing cancer, these findings carry a double message. On the one hand, rheumatic side effects from immune checkpoint drugs are not rare, and some, particularly those affecting muscles and large blood vessels, can be life‑threatening if not recognized quickly. On the other hand, many of these conditions respond well to timely treatment, and not all require permanently stopping the cancer therapy. The authors conclude that closer collaboration between oncologists and rheumatologists, clearer guidelines, and better awareness of early warning signs could help patients stay on effective cancer drugs while limiting damage to joints, muscles, and blood vessels. In the long run, better monitoring and research into risk markers may allow doctors to predict who is most likely to develop these side effects and to tailor treatment accordingly.
Citation: Llobell-Uriel, A., González-Mazón, Í., Gratacós, J. et al. Rheumatic immune-related adverse effects of immune checkpoint treatment: a retrospective multicentre study. Sci Rep 16, 13159 (2026). https://doi.org/10.1038/s41598-026-43606-5
Keywords: cancer immunotherapy, immune checkpoint inhibitors, autoimmune side effects, arthritis and myositis, rheumatology oncology