Clear Sky Science · en
Synthesis, antimicrobial evaluation, molecular docking, and drug-likeness assessment of novel phenothiazine chromene hybrid compounds
Why New Germ-Fighting Molecules Matter
Antibiotic‑resistant infections are turning once‑routine illnesses into serious medical threats. This study reports a new family of laboratory‑made molecules that blend three known drug‑like building blocks into a single structure. By doing so, the researchers aim to outsmart bacteria and fungi that have learned to shrug off many existing medicines. The work not only tests how well these hybrids kill microbes, but also asks whether they look “drug‑like” enough to be taken by mouth and whether they latch onto a key resistance enzyme inside microbes.
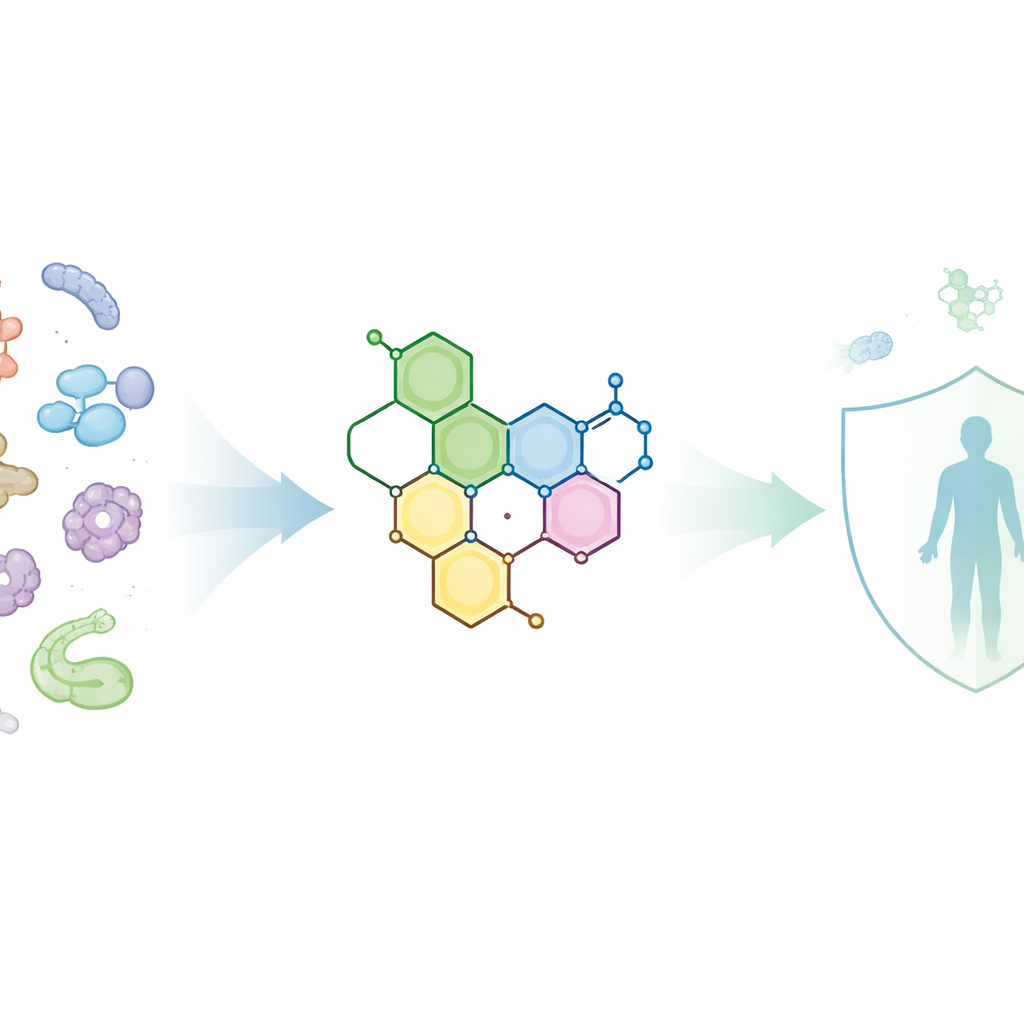
Building a Smarter Molecule
The research team started from a chemical core called phenothiazine, already known to disturb microbial cell membranes and interfere with their defenses. To this, they grafted ring‑shaped units called chromenes and related structures, plus smaller fragments able to form additional rings such as thiazoles, thiophenes, and pyrazoles. Stepwise reactions joined these parts under controlled conditions, and each product was checked by standard techniques that read out atomic vibrations and magnetic behavior to confirm that the desired structures had formed. The end result was a small library of related molecules, each with a slightly different arrangement of rings and side‑groups.
Testing How Well They Fight Germs
The new compounds were then challenged against a panel of disease‑causing microbes: common Gram‑negative bacteria such as E. coli, Gram‑positive species like Staphylococcus aureus and Bacillus subtilis, and two troublesome fungi, Candida albicans and Fusarium oxysporum. The team measured the minimum amount of each compound needed to stop growth in liquid culture. Several hybrids stood out. Two candidates, labeled 4 and 7, were especially effective against E. coli, needing only tens of micrograms per milliliter to halt growth—on par with or better than a reference antibiotic in some tests. Another compound, 10, showed the strongest activity against Fusarium, a plant and opportunistic human pathogen. In contrast, some simpler intermediates in the synthesis were almost inactive, underscoring that the final ring‑rich frameworks are crucial for potency.
From Test Tube to Potential Pill
Having promising activity is only half the battle; a useful drug also needs to move through the body in the right way. The researchers therefore checked each molecule against the widely used “rule‑of‑five” guidelines, which estimate whether a compound is likely to be absorbed after oral dosing. Most of the hybrids passed these filters, with molecular weights close to or just above the preferred limit and a balance of oily and water‑friendly features that should allow them to cross cell membranes without becoming too sticky or too bulky. Calculations of polar surface area—a measure related to how strongly a molecule interacts with water—suggested that several candidates, particularly compounds 5, 6, 8a, 8b, and 11, would be well absorbed in the gut.
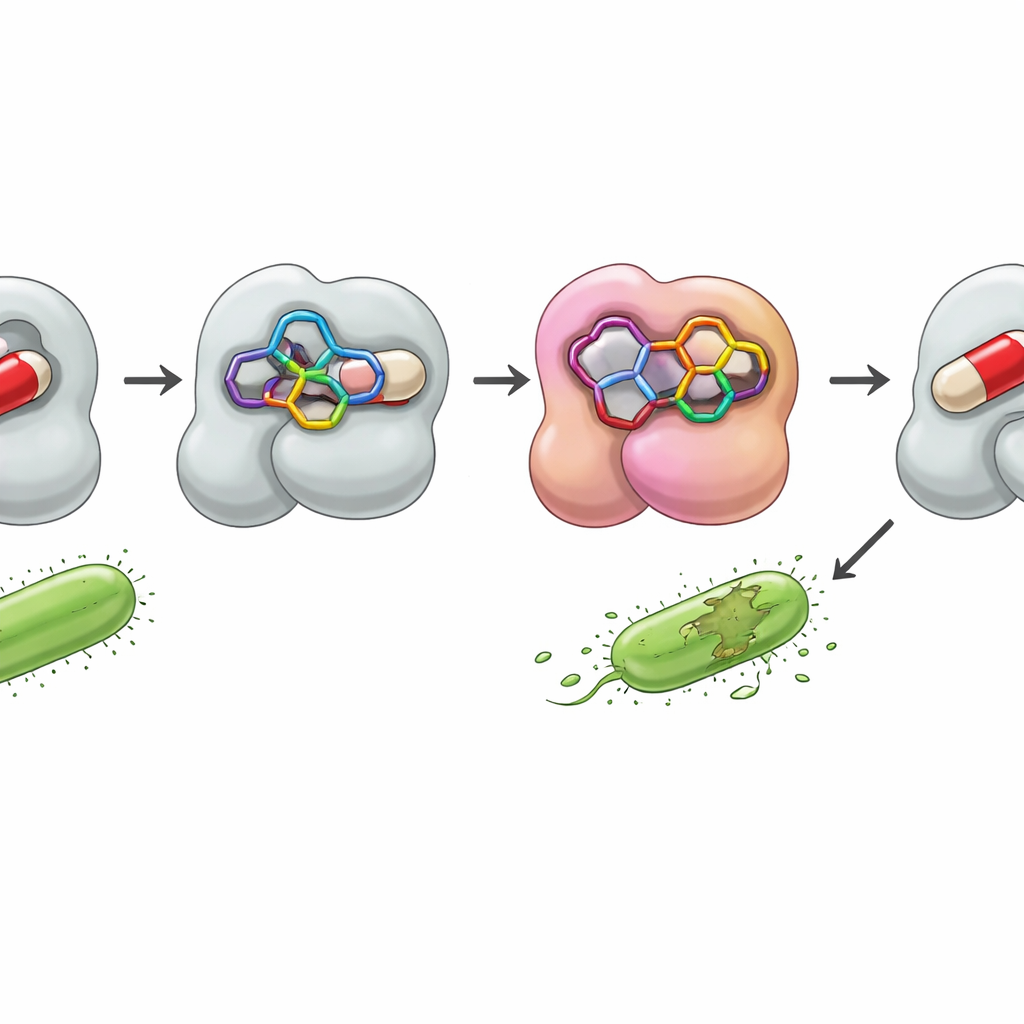
Peeking Inside the Bacterial Shield
To explore how these hybrids might beat resistance, the team turned to computer docking studies with a bacterial enzyme called β‑lactamase. Many bacteria use this protein as a shield, chopping up β‑lactam antibiotics before they can do damage. By modeling how the new molecules sit in the enzyme’s active pocket, the researchers could estimate binding strength and map key contacts. One compound, 7, nestled particularly snugly into the site, forming stabilizing interactions with several amino acids and achieving a calculated binding energy even better than that of the reference drugs ampicillin and clotrimazole. Another active molecule, 4, also showed favorable interactions. Statistical analyses of the biological data backed up these trends, grouping compounds 4, 7, and 10 together as the most consistently potent across the tested microbes.
What This Could Mean for Future Treatments
In everyday terms, this study shows that carefully “wiring together” multiple drug‑friendly fragments into a single, flat, ring‑rich molecule can produce new candidates that both hit resistant microbes hard and look suitable for development as pills. The most successful hybrids not only slowed the growth of a range of bacteria and fungi, but also appeared to wedge themselves into a key resistance enzyme more firmly than existing drugs in computer models. While these molecules are still at an early, laboratory stage and must be tested for safety and performance in animals and humans, they offer a promising blueprint for next‑generation antibiotics designed to keep pace with rapidly evolving, multidrug‑resistant pathogens.
Citation: Bayoumy, N.M., Fadda, A.A., Gaffer, H.E. et al. Synthesis, antimicrobial evaluation, molecular docking, and drug-likeness assessment of novel phenothiazine chromene hybrid compounds. Sci Rep 16, 10592 (2026). https://doi.org/10.1038/s41598-026-43195-3
Keywords: antibiotic resistance, antimicrobial agents, phenothiazine hybrids, molecular docking, drug design