Clear Sky Science · en
A robust mouse liver organoid platform enables sustained multicellular maturation and fibrosis modeling from a single tissue sample
Why Growing Mini Livers Matters
Chronic liver disease is rising worldwide, yet treatment options remain limited, and donor organs for transplant are scarce. Scientists are turning to tiny three-dimensional “mini organs,” or organoids, grown in the lab to better understand liver disease, test new drugs, and explore future regenerative therapies. This study describes a new mouse liver organoid system that, from a single small piece of liver, can reliably grow several key liver cell types together and even mimic the scarring process known as fibrosis. 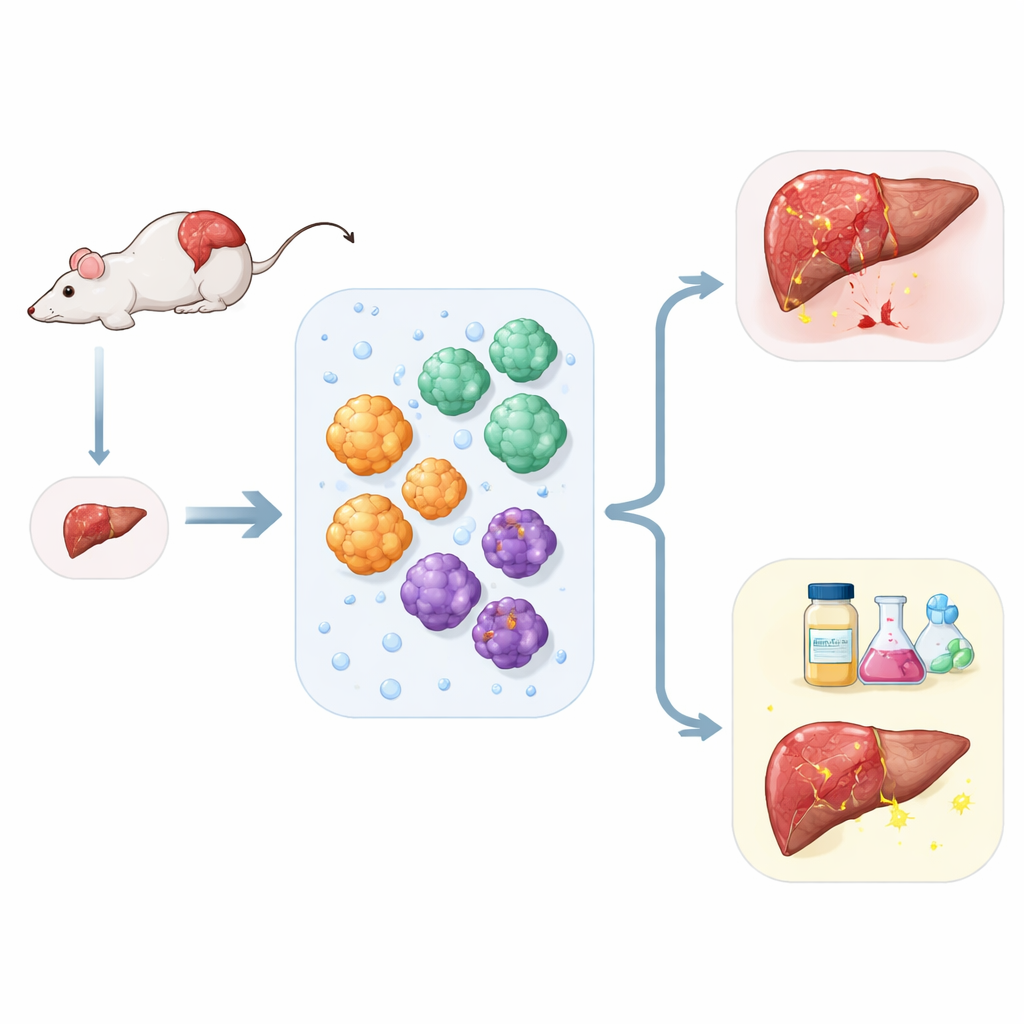
Building Tiny Livers from a Small Sample
The researchers set out to solve a practical problem: how to get the most information from very little liver tissue. Using a streamlined preparation method, they digested mouse liver into individual cells and then separated them based on simple physical and surface properties. From one tissue sample, they could isolate three major players of the liver: working liver cells that handle metabolism, bile duct cells that move digestive fluids, and support cells called stellate cells that drive scarring. Each cell type was grown in a gel-like scaffold, where it self-organized into spherical organoids resembling miniaturized pieces of liver tissue.
Keeping Liver Cells Mature and Active
A long-standing challenge in liver research is that liver cells quickly lose their specialized abilities once taken out of the body. To tackle this, the team fine-tuned the chemical recipe of the growth medium for hepatocyte organoids. By adding a Notch pathway blocker (which nudges cells away from a stem-like state) and the steroid drug dexamethasone, they encouraged the organoids to stay functionally “adult.” Over many rounds of growth, these organoids kept making albumin, storing sugars, processing fats, and carrying out drug-breaking enzymes in ways that closely matched freshly isolated liver cells. Importantly, the cells did not drift back toward a less mature or wrong identity, which is a frequent issue in older organoid systems.
Capturing Bile Duct and Support Cells
The same liver fragments also yielded robust bile duct organoids. These structures formed hollow spheres lined with a single layer of cells that resembled tiny tubes. They showed stable expression of markers typical of bile duct tissue and demonstrated an active transport pump that can move compounds into the central cavity, a key feature for handling bile and drugs. In parallel, the team purified stellate cells to very high purity by exploiting their natural stores of vitamin A–rich droplets. In their resting state, these stellate cells quietly stored lipids, but when exposed to a signaling molecule associated with injury, they transformed into a more aggressive, fiber-producing form, echoing what happens during scarring in real livers.
Modeling Liver Scarring in a Dish
To see whether these components could recreate early liver damage, the scientists brought the organoids and stellate cells together in a shared culture. When stellate cells were switched into their activated, scar-forming state, they released soluble factors that reached the nearby hepatocyte and bile duct organoids. The once-healthy organoids slowed their growth, lost some of their characteristic liver features, and began to show signs of a cellular identity shift linked to tissue stiffening and loss of function. These changes mirror the way scarring in chronic liver disease gradually undermines the organ’s ability to regenerate and detoxify the body. 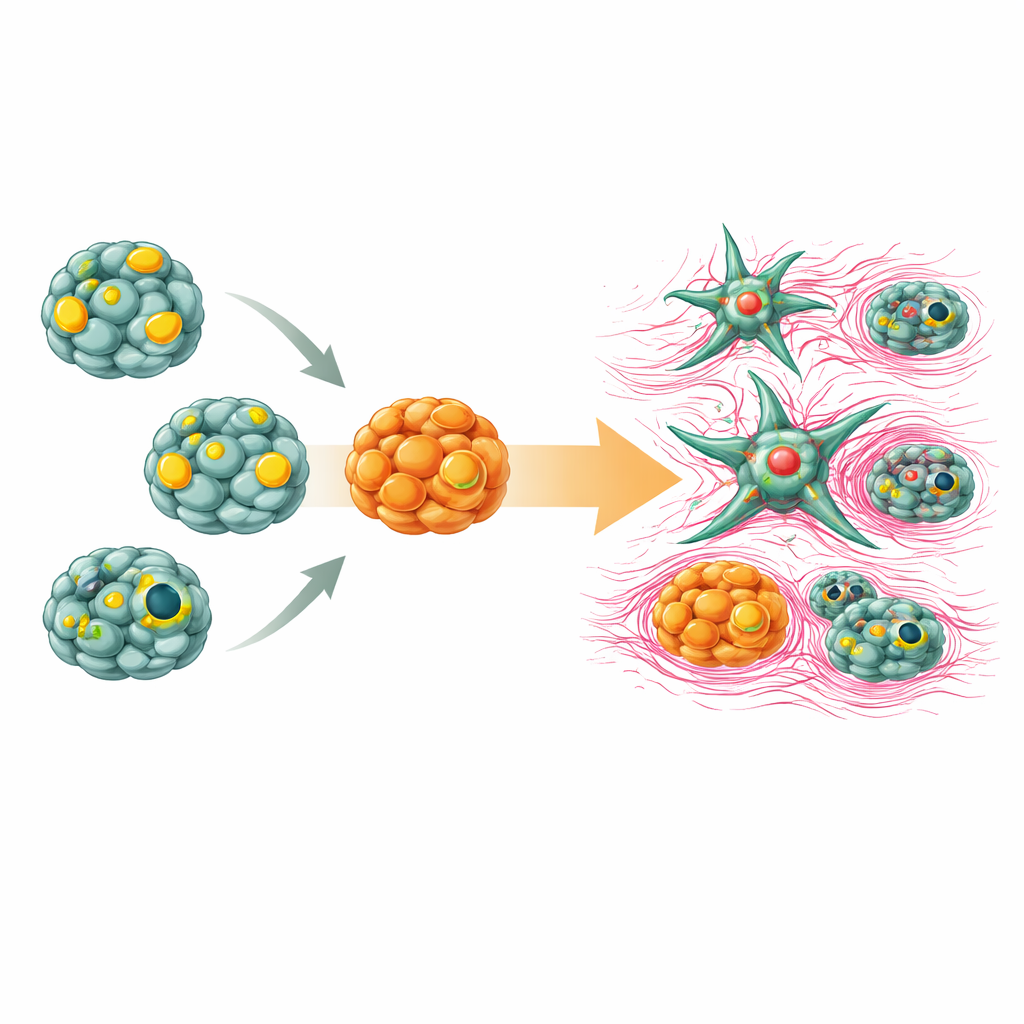
What This Means for Future Liver Care
By building a stable, multicellular mini-liver system from just one small tissue piece, this study offers a powerful new tool for liver research. The platform faithfully maintains the behavior of several liver cell types and can reproduce key steps of fibrosis, all in a controlled dish. For non-specialists, the takeaway is that scientists can now study liver injury, recovery, and drug responses in a much more realistic yet scalable way, without relying solely on animal models or scarce human organs. In the long run, such systems could speed up the discovery of anti-fibrosis drugs and help personalize treatments, bringing us closer to therapies that halt or even reverse chronic liver damage.
Citation: Liang, Y., Ye, Y., Xie, H. et al. A robust mouse liver organoid platform enables sustained multicellular maturation and fibrosis modeling from a single tissue sample. Sci Rep 16, 14137 (2026). https://doi.org/10.1038/s41598-026-42990-2
Keywords: liver organoids, fibrosis modeling, hepatic stellate cells, regenerative medicine, drug screening