Clear Sky Science · en
Fingolimod normalizes metabolic signatures associated with synaptic plasticity and memory in APP/PS1 model: Sphingosine-1-phosphate receptor a therapeutic target for Alzheimer’s
Why this research matters for families and aging brains
Alzheimer’s disease robs millions of people of their memories, yet today’s medicines offer only modest relief and little help for the underlying brain changes. This study looks at an unexpected ally: fingolimod, a drug already used for multiple sclerosis. By examining how this medicine reshapes brain chemistry and boosts memory in an Alzheimer’s-like mouse model, the researchers suggest a new way to protect brain cells by restoring their energy supply and calming damaging inflammation.
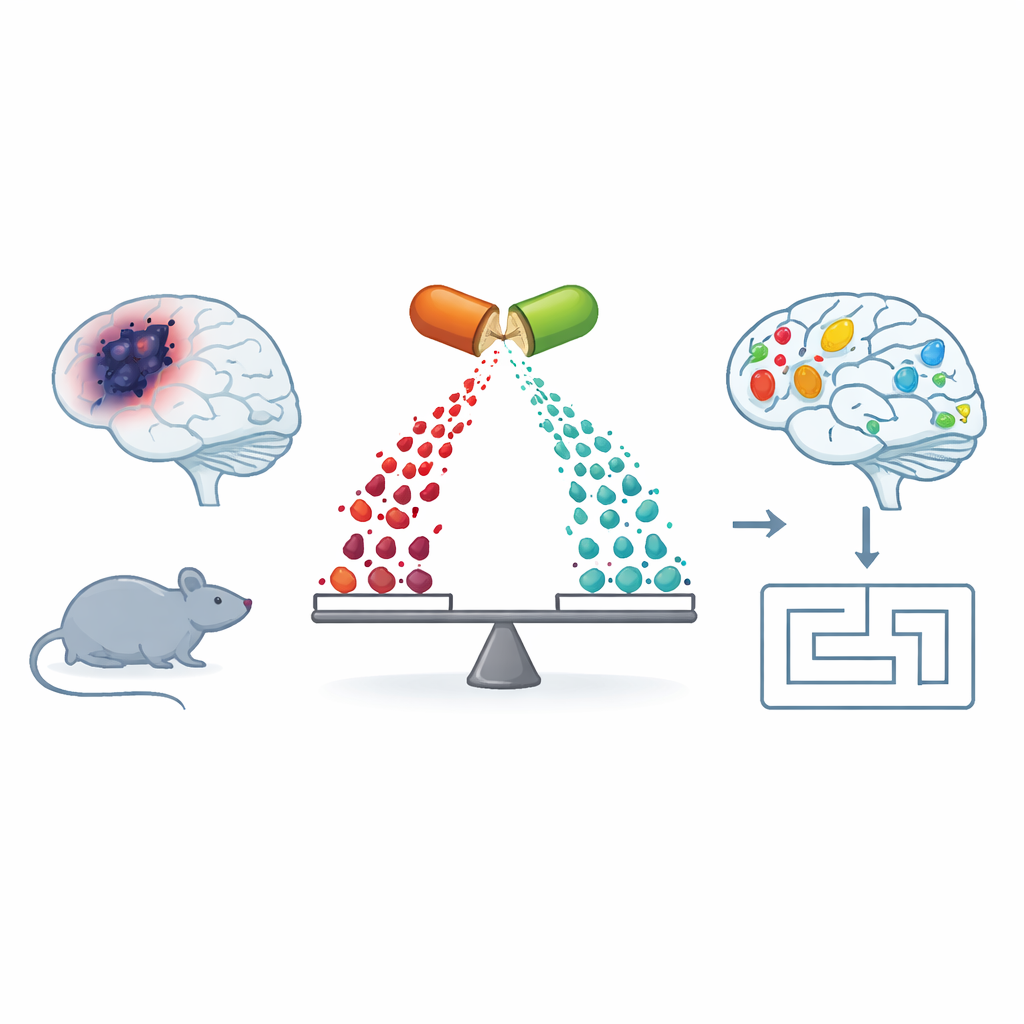
A closer look at Alzheimer’s beyond plaques and tangles
Most people have heard that sticky amyloid plaques and twisted tau tangles are hallmarks of Alzheimer’s disease. But behind these visible signs lies a quieter disturbance in the brain’s chemistry, especially in fats (lipids) that form cell membranes and carry signals between cells. The team studied mice that develop Alzheimer’s-like changes (APP/PS1 mice) and compared them with healthy mice. They measured hundreds of small molecules in the brain, blood, and liver and found that the Alzheimer’s-like mice showed broad imbalances in several groups of molecules, including specialized fats and compounds called polyamines that help keep mitochondria—the cells’ power plants—running smoothly.
When the brain’s fat balance tips the wrong way
A major focus was on a set of fats called sphingolipids, which can either push cells toward survival or toward death, depending on their balance. In the Alzheimer’s-like mice, this “fat balance” was skewed toward molecules linked to cell stress and inflammation, while protective forms were reduced. At the same time, key membrane-building fats in mitochondria-rich regions of brain cells were altered, and the production of spermidine and related polyamines, which support mitochondrial energy production, was reduced. Together, these shifts point to an energy-starved, inflammation-prone brain environment that can undermine synapses—the junctions where learning and memory are encoded.
Repurposing a multiple sclerosis drug to reset brain chemistry
The researchers then treated the Alzheimer’s-like mice with fingolimod, a drug that targets receptors for a lipid messenger called sphingosine‑1‑phosphate. In earlier work, the same group had shown that this treatment improved learning and synaptic plasticity (the ability of synapses to strengthen with experience). In the new study, they confirmed that fingolimod restored performance in a spatial memory task (the Barnes maze) and revived long-term potentiation, a key electrical signature of learning, in a major hippocampal pathway. At the chemical level, fingolimod increased protective sphingolipid signals, reduced markers of damaging ceramides, and largely reversed many of the abnormal lipid saturation patterns in brain membranes, especially in temporal cortex regions linked to memory.
Powering up mitochondria and stabilizing memory circuits
Beyond these fat molecules, fingolimod boosted indicators of spermidine synthesis and increased acylcarnitines, intermediate carriers that ferry fats into mitochondria for energy production. These changes were tightly linked to improvements in both memory performance and synaptic strength, suggesting that the drug helps rescue mitochondrial function in brain cells. The treatment also shifted other metabolic pathways in ways that fit with known cell-signaling effects of the drug: promoting pro-survival pathways, raising serotonin, supporting anti-inflammatory immune states in the brain, and reducing by-products associated with oxidative stress. In short, the drug seemed to push the entire network of signals toward a healthier, more resilient state.
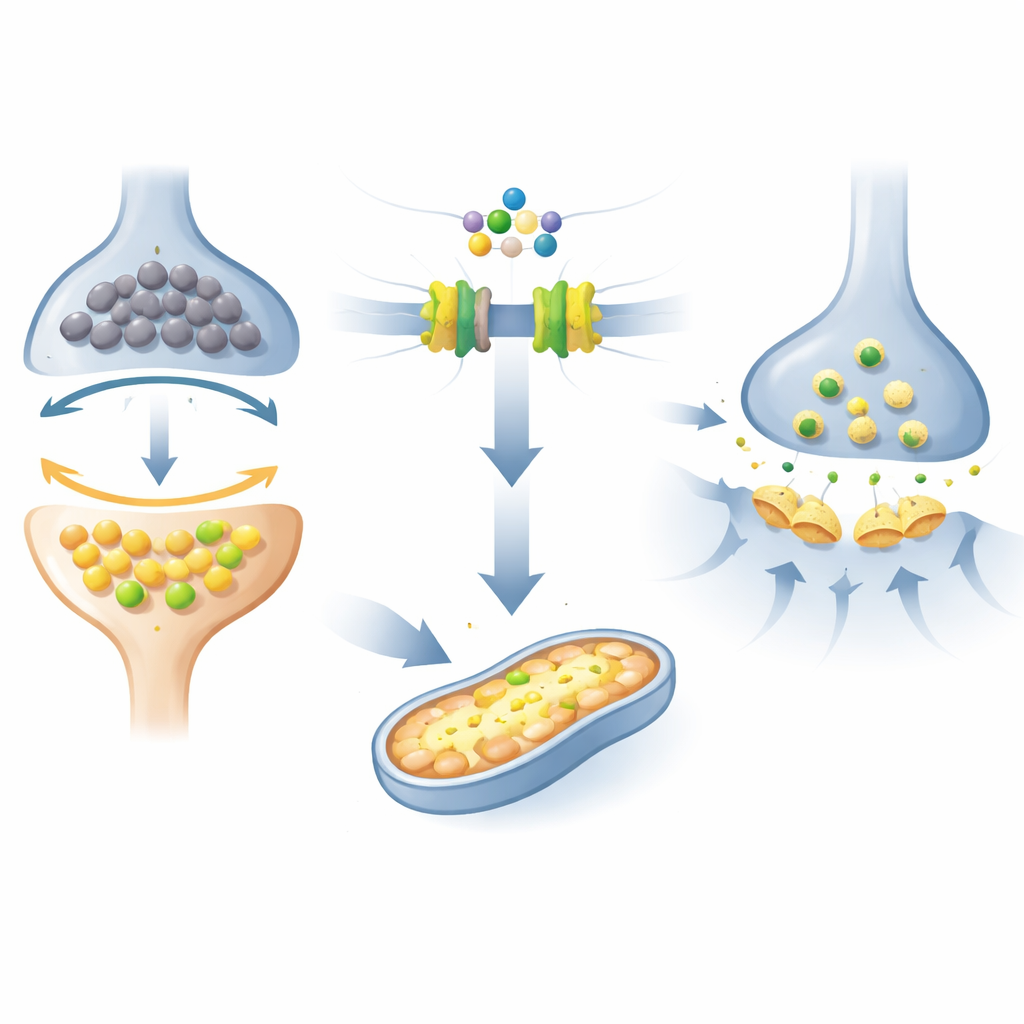
What this could mean for future Alzheimer’s treatment
In everyday terms, this work suggests that helping brain cells rebalance their internal chemistry—especially the fats that control cell survival and the fuels that feed mitochondria—may restore some of the memory functions damaged by Alzheimer’s-like changes. Fingolimod did not simply mask symptoms; in mice, it corrected underlying metabolic disruptions tied to synaptic health and learning. While mice are not people and more mechanistic and clinical studies are needed, these findings highlight a promising new therapeutic angle: targeting lipid and energy metabolism, via sphingosine‑1‑phosphate receptors and related pathways, to protect brain circuits long before they fail.
Citation: Kalecký, K., Buitrago, L., Alarcon, J.M. et al. Fingolimod normalizes metabolic signatures associated with synaptic plasticity and memory in APP/PS1 model: Sphingosine-1-phosphate receptor a therapeutic target for Alzheimer’s. Sci Rep 16, 12835 (2026). https://doi.org/10.1038/s41598-026-42518-8
Keywords: Alzheimer’s disease, brain metabolism, fingolimod, sphingolipids, mitochondrial function