Clear Sky Science · en
An MIEN1-based hexamer peptide (LA3IK) inhibits EGF-driven oncogenic signaling in prostate cancer by disrupting EGFR–ERBB2 heterodimerization
Why This Matters for Prostate Cancer
Men with advanced prostate cancer often run out of effective treatment options, especially when tumors no longer respond to hormones. This study explores a tiny lab-made molecule, a six–amino acid peptide called LA3IK, that aims to shut down one of the cancer’s favorite growth signals without harming healthy tissues. Understanding how this miniature drug candidate works could open the door to safer, highly targeted therapies for aggressive prostate cancer.
A Growth Switch That Cancer Hijacks
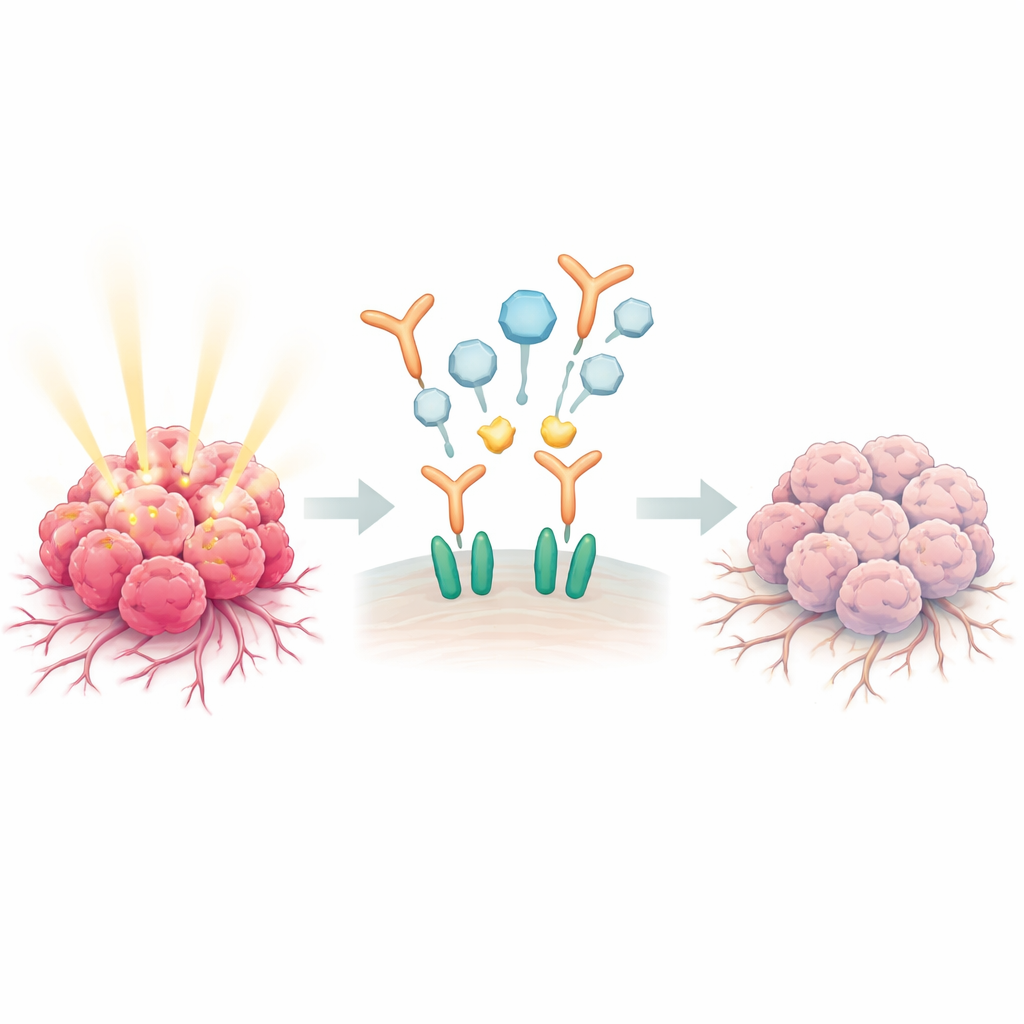
Many prostate tumors become dangerously aggressive by overusing a natural communication system built around epidermal growth factor (EGF) and its receptor, EGFR. When EGF latches onto receptors on the surface of cancer cells, those receptors team up with a partner protein called ERBB2, forming powerful pairs that send commands for cells to move, invade neighboring tissue, and attract new blood vessels. In healthy prostate tissue, male hormones help keep this signaling in check. But in advanced disease, that brake fails, and EGFR–ERBB2 activity is linked to relapse, spread to bone, and resistance to therapy.
A Tiny Peptide with a Precise Target
The researchers built LA3IK from a cancer-associated protein named MIEN1, which is known to boost cell migration and invasion. Earlier work showed LA3IK could dampen harmful signals in breast cancer cells, so the team asked whether it might also tame EGF-driven pathways in prostate cancer. Using PC3 prostate cancer cells, which overproduce EGFR and behave aggressively in the lab, they found that LA3IK does not simply kill cells outright. Instead, it acts like a focused disruptor: it blocks the formation of EGFR–ERBB2 pairs and cuts off the chemical tags that normally flip EGFR and its downstream partners into the “on” position.
How LA3IK Disrupts the Signal
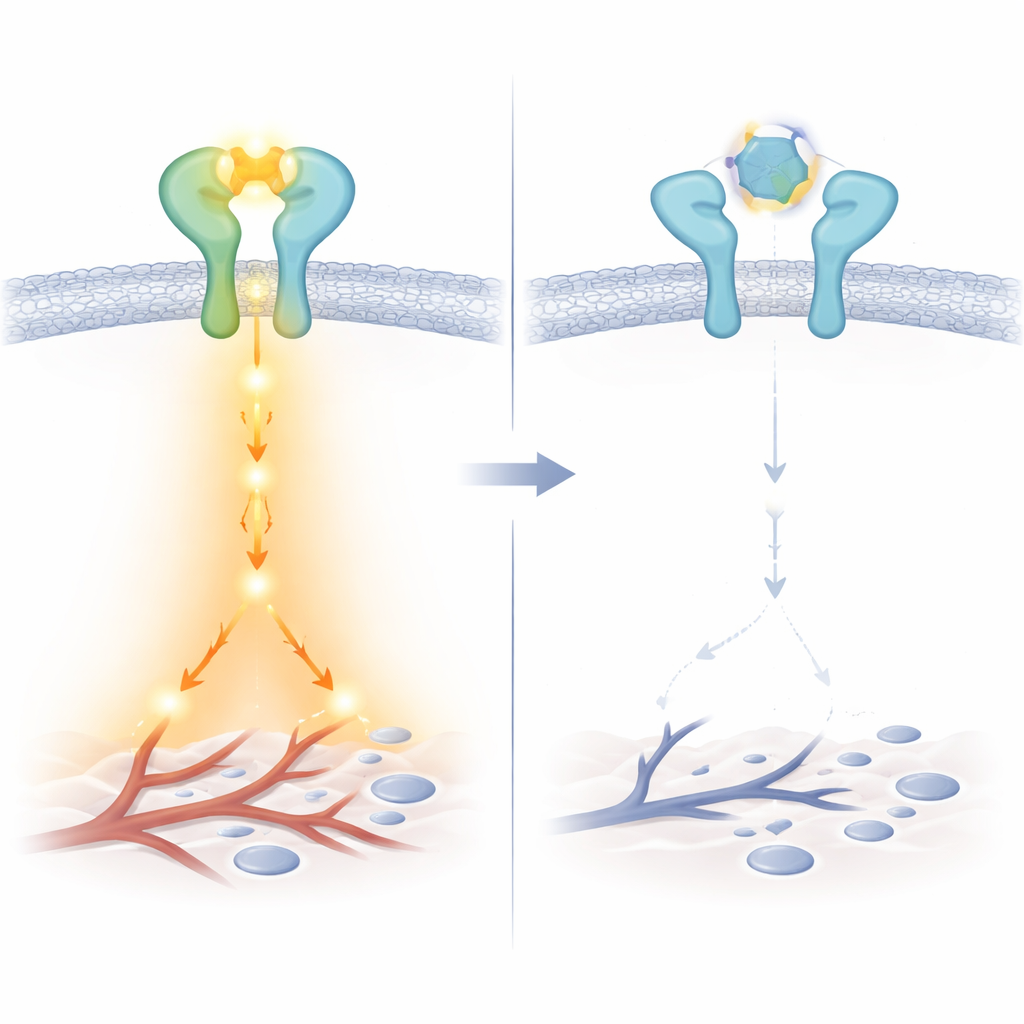
Closer biochemical experiments revealed a subtle but important twist: LA3IK binds directly to EGF itself and alters the local environment around two of EGF’s tryptophan building blocks. This tiny structural nudge appears to weaken how effectively EGF can engage the EGFR–ERBB2 pair, even though the receptors can still form larger clusters on the cell surface. As a result, key messengers inside the cell—such as Src, STAT3, NF-κB, and beta-catenin—are much less activated. In functional tests, this translated into sharply reduced cancer cell movement and invasion through artificial barriers, without reducing the number of living cells. LA3IK also turned down genes and secreted factors that drive blood vessel growth, including ANGPTL4 and VEGF family members, suggesting that it can dampen both spread and the tumor’s ability to feed itself.
Strong Action on Tumors, Gentle Touch on Healthy Tissue
An important concern with any drug that targets EGFR is collateral damage: this receptor also protects normal tissues, especially the liver. In mice given EGF injections, LA3IK did not disturb protective EGFR signals in liver tissue, nor did it change levels of EGFR circulating in the blood. In cell-based tests, the peptide was more selective for prostate cancer cells than a standard EGFR-blocking pill, erlotinib, particularly in some widely used cancer lines. This pattern suggests that LA3IK acts preferentially in the cancer setting, likely because it was designed from MIEN1, a protein that is abundant in tumors but scarce in normal cells.
What the Findings Mean Going Forward
Taken together, the results introduce LA3IK as an unusually short peptide that can blunt an important growth and spread pathway in advanced prostate cancer while largely sparing healthy tissue, at least in early tests. Rather than smashing the receptor machinery outright, LA3IK subtly reshapes the growth factor that feeds it and prevents the most aggressive receptor pairings from forming. For patients, this strategy points toward future medicines that slow invasion and angiogenesis with fewer side effects, offering a more precise way to disarm tumors that have outgrown standard hormone-based treatments.
Citation: Tripathi, A.K., Ahamad, N., Tyagi, A. et al. An MIEN1-based hexamer peptide (LA3IK) inhibits EGF-driven oncogenic signaling in prostate cancer by disrupting EGFR–ERBB2 heterodimerization. Sci Rep 16, 12295 (2026). https://doi.org/10.1038/s41598-026-41933-1
Keywords: prostate cancer, growth factor signaling, peptide therapy, tumor invasion, angiogenesis