Clear Sky Science · en
A computational analysis of biophysical and geometric constraints refutes existing hypotheses of cross-excitation in dorsal root ganglia
Why neighboring nerve cells matter
Every touch, ache, or change in temperature you feel starts its journey in tiny clusters of nerve cells called dorsal root ganglia, tucked just outside the spinal cord. For decades, scientists have seen an odd behavior there: when one of these cells fires, its neighbors often become easier to excite, as if the signal were quietly spreading sideways. This “cross-excitation” could reshape how we think about pain and sensation. The new study uses detailed computer models to ask a simple but uncomfortable question: do our current explanations for this sideways chatter actually work?
A crowded neighborhood of nerve cells
Dorsal root ganglia are tightly packed neighborhoods of sensory neurons. Each neuron’s cell body is wrapped in a thin shell of support cells called satellite glial cells, forming a capsule that largely insulates one neuron from another. Classic textbooks describe these neurons as simple relays, passing signals forward to the spinal cord only when inputs are strong enough. Yet experiments have shown that when one neuron is stimulated, many neighbors become more excitable within about half a second to a second. This slow, delayed boost suggests a chemical or diffusion-based process rather than a fast electrical one, and it appears in most large, touch-related fibers, hinting at a fundamental role in sensory processing.
Testing the idea of leaking ions
One leading explanation is that when a neuron fires repeatedly, it releases potassium ions into the tiny fluid space between its surface and the surrounding glial shell. If enough potassium accumulates and seeps through the surrounding tissue, it could nudge nearby neurons closer to firing. The authors rebuilt a highly detailed model of a sensory neuron and its glial capsule, including dozens of ion channels and pumps, and then simulated potassium levels rising from normal to values measured in experiments. While a simple textbook formula suggested the neuron’s voltage might shift by a few millivolts—close to what experiments report—the full biophysical model told a different story. Once realistic ion regulation by both neuron and glia was included, the voltage shift was less than one millivolt, far too small to explain the observed cross-excitation.
Following the trail through tiny spaces
The team then asked whether potassium could simply diffuse from a firing neuron to its neighbors through the maze-like connective tissue between capsules. They connected two modeled neuron–glia units with a simulated extracellular corridor and drove one neuron with rapid electrical pulses, mimicking past experiments. The voltage change in the silent neighbor was vanishingly small—on the order of ten-millionths of a millivolt. Even when they packed neurons unrealistically close together or reduced the available space to a narrow labyrinth, diffusion only produced noticeable effects when two cell bodies were almost sharing a common envelope, a rare occurrence under healthy conditions. From a physical standpoint, diffusion just could not carry a strong enough signal across typical distances.
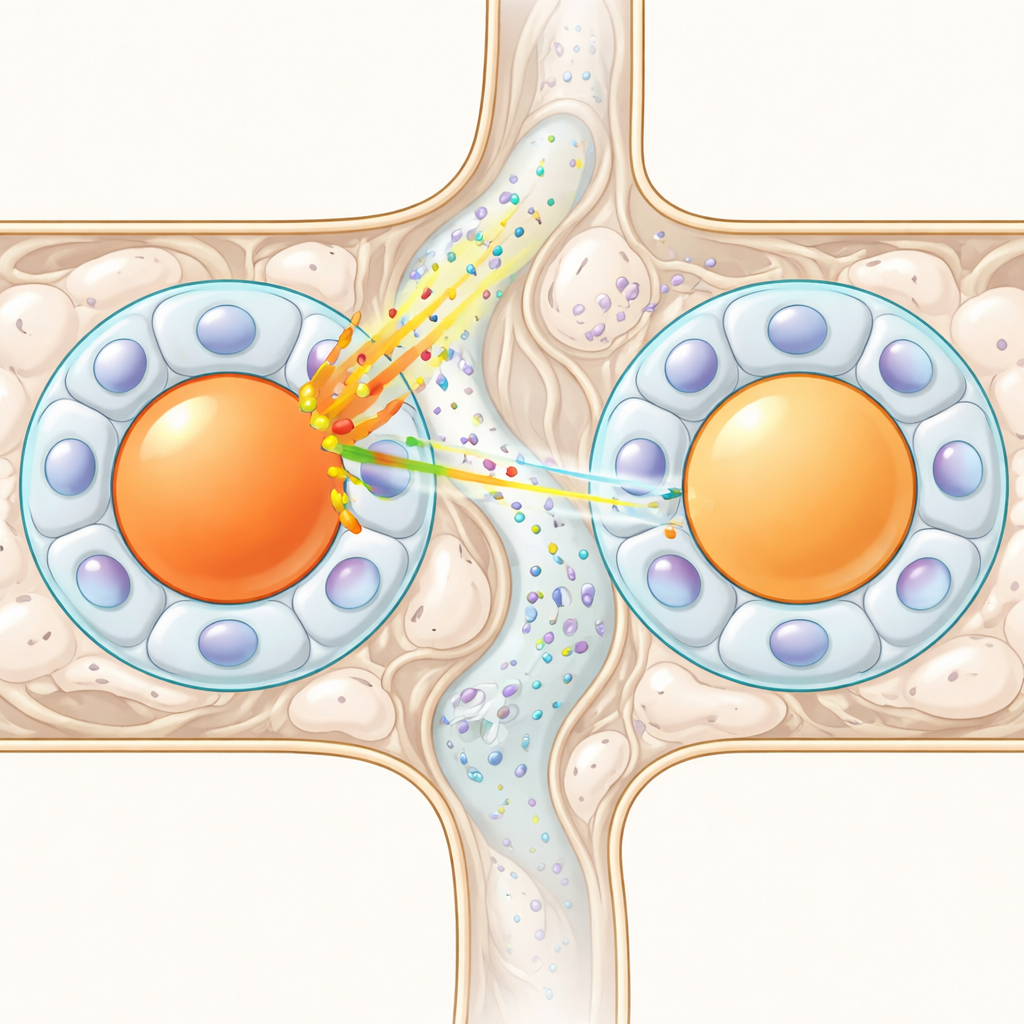
When structure makes connection unlikely
Another set of hypotheses relies on physical bridges between cells. Some satellite glial cells form direct connections called gap junctions, or special “sandwich” arrangements where one glial cell sits between two neuron bodies and can transmit chemical signals. If such links were common, they could form a network that spreads excitation. To test this, the researchers built a 3D statistical model of how neuron–glia modules of different sizes can be packed inside a ganglion, then overlaid known probabilities for these connections. Experiments suggest that gap junctions between distinct capsules are rare—typically a few percent—and sandwich-like arrangements occur in only about one in ten neurons. The simulations showed that, given these low odds and the limited number of physical neighbors (typically about seven per neuron), you cannot reach the very high rates of cross-excitation seen experimentally. Even combining all known structural mechanisms still falls far short.
What this means for feeling and treating pain
By pushing both the physics and the anatomy to their most generous limits—maximizing ion leaks, packing cells as tightly as possible, and assuming best-case configurations—the authors tried to give existing theories every chance to work. Yet diffusion of ions, rare gap junctions, and sandwich-like contacts together still could not reproduce the strength and prevalence of cross-excitation observed in real tissue. For a lay reader, the takeaway is straightforward: the sideways “echo” of nerve activity in these ganglia is real and common, but our current explanations are incomplete. Something important about how sensory neurons talk to each other before signals even reach the spinal cord is still missing. Understanding this hidden layer of processing could reshape ideas about normal sensation and open new paths for pain therapies, especially as doctors increasingly use electrical stimulation near these ganglia to treat chronic pain and aid recovery after spinal cord injury.
Citation: Perevozniuk, D., Gorskii, O., Musienko, P. et al. A computational analysis of biophysical and geometric constraints refutes existing hypotheses of cross-excitation in dorsal root ganglia. Sci Rep 16, 14306 (2026). https://doi.org/10.1038/s41598-026-40355-3
Keywords: dorsal root ganglion, cross-excitation, sensory neurons, neuropathic pain, computational modeling