Clear Sky Science · en
M2 macrophage–derived extracellular vesicles induce EMT-like transcriptional reprogramming in colorectal cancer cells via upregulation of FAM83A
Why our own defenses can sometimes aid cancer
Cancer does not grow in isolation; it develops within a bustling neighborhood of immune and support cells that can either restrain or unknowingly assist the tumor. This study explores how a particular set of immune cells can send tiny biological “packages” that push colon cancer cells toward a more mobile, invasive state linked to metastasis, the spread of cancer throughout the body.
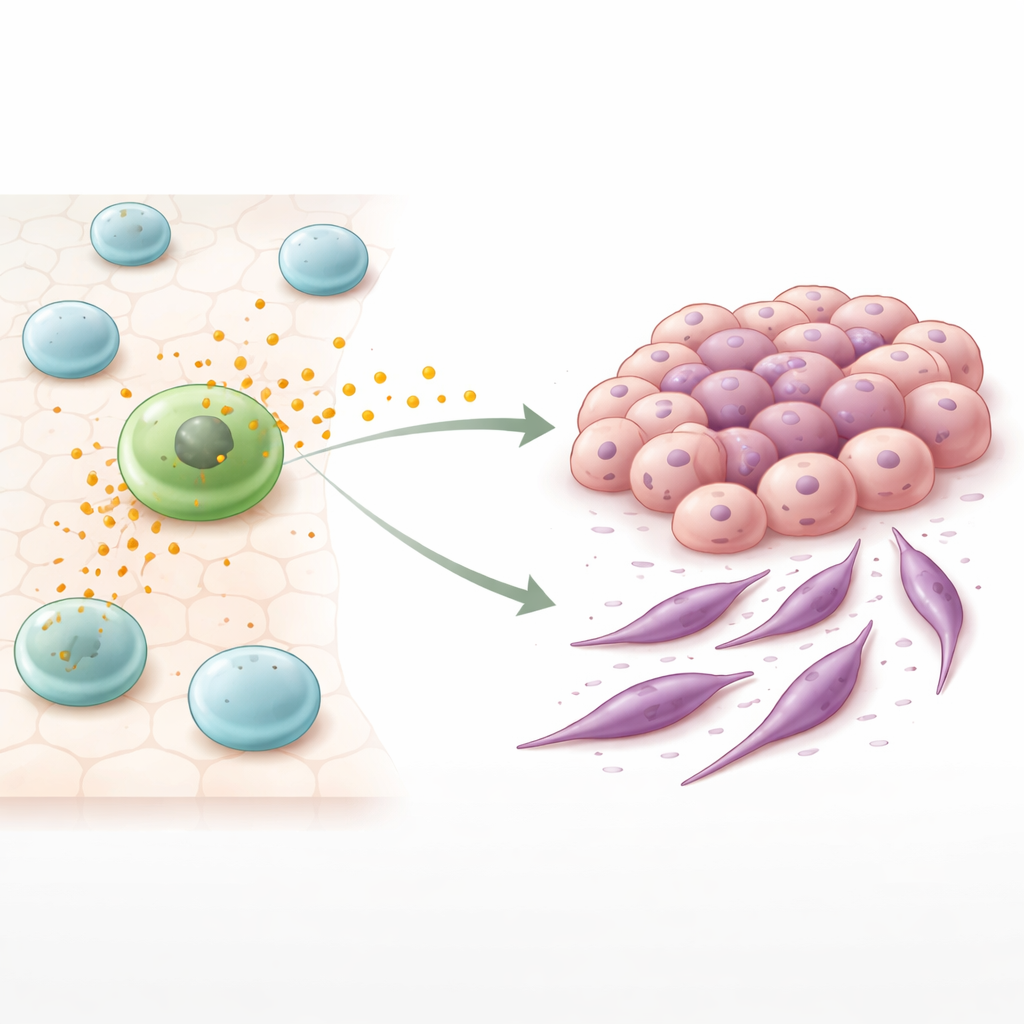
The hidden messages sent by immune cells
Among the immune cells that gather around tumors, macrophages are key players. Some macrophages adopt a warrior-like mode that can attack tumors, while others, called M2-like macrophages, take on a more nurturing, wound-healing role. In many cancers, these M2-like cells accumulate and are associated with tumor growth and poor outcome. The authors focused on tiny membrane bubbles released by these cells, known as extracellular vesicles or exosomes. Packed with proteins and genetic material, these vesicles travel between cells and can reprogram the behavior of their targets.
Turning settled tumor cells into wanderers
To test how these vesicles affect colon cancer, the researchers grew human colorectal cancer cells together with M2-like macrophages or exposed the cancer cells only to purified macrophage vesicles. In both situations, the cancer cells began to lose features of tightly connected, orderly tissue cells and gained traits of more flexible, spindle-shaped cells that can move more easily. This shift resembles a process called epithelial–mesenchymal transition, in which formerly stable cells loosen their attachments and become more migratory. Strikingly, the changes were even stronger when cancer cells were treated with isolated vesicles than when they were simply co-cultured with macrophages, suggesting that these vesicles are a major driver of the effect.
Uncovering a key switch inside cancer cells
To understand what was happening inside the cancer cells, the team compared the activity of thousands of genes after vesicle exposure. They found widespread rewiring of gene activity, including a strong rise in a gene called FAM83A, previously linked to aggressive behavior in several cancers. When FAM83A levels went up, the colon cancer cells strongly reduced molecules that help them stick to their neighbors and increased molecules and enzymes associated with movement and tissue breakdown. When the researchers used small interfering RNA, a precise molecular tool, to reduce FAM83A levels, the vesicle-driven shift toward a more mobile state was largely blocked. This pinpointed FAM83A as a crucial switch that connects the vesicle signal from immune cells to the internal reprogramming of cancer cells.
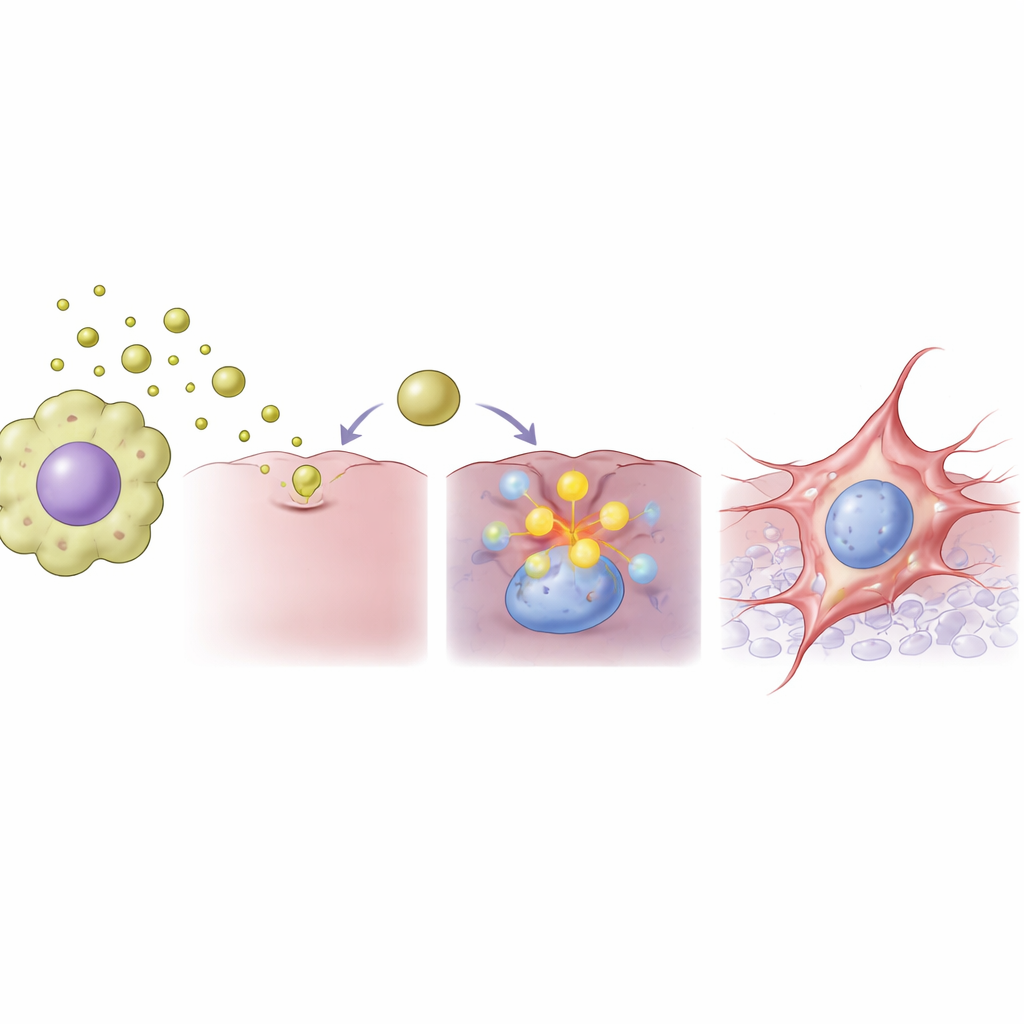
How these findings may shape future cancer treatments
Because these vesicles proved more potent than the macrophages themselves at pushing cancer cells toward invasive behavior, they emerge as promising new targets. The study suggests that blocking vesicle release from tumor-associated macrophages, or directly turning down FAM83A in tumor cells, might help prevent early steps of metastasis. The authors also saw hints that this vesicle–FAM83A pathway could matter in other cancers, although its role seems to vary between tumor types, underscoring the need for more research before therapies can be designed.
What this means for people facing cancer
In simple terms, this work shows that certain immune cells can send microscopic packages that “coach” colon cancer cells to become more mobile and potentially more dangerous, and that a single gene inside the cancer cells acts as a key relay for this message. By mapping out this chain—from macrophage vesicles to FAM83A activation to a more invasive cell state—the study offers a clearer picture of how the tumor environment can fuel progression. In the long run, therapies that disrupt these signals could complement existing treatments, aiming not only to shrink tumors but also to keep them from spreading.
Citation: Isik, M., Akkulak, M. & Derkus, B. M2 macrophage–derived extracellular vesicles induce EMT-like transcriptional reprogramming in colorectal cancer cells via upregulation of FAM83A. Sci Rep 16, 9829 (2026). https://doi.org/10.1038/s41598-026-39262-4
Keywords: colorectal cancer, tumor microenvironment, macrophage exosomes, epithelial mesenchymal transition, FAM83A