Clear Sky Science · en
Assessment of antimicrobial activity of [1,2,4]triazolo[4, 3-a]quinoxaline derivatives individually and in combination with levofloxacin
Why new germ-fighting partners matter
Antibiotic resistance is eroding the power of many trusted drugs, leaving doctors with fewer options to treat everyday infections. This study explores a strategy that does not rely on inventing a completely new antibiotic, but instead builds small partner molecules that can boost the performance of an existing drug, levofloxacin. By pairing this common antibiotic with specially designed helper compounds, the researchers aim to knock down dangerous bacteria, including drug‑resistant strains, using lower doses and with limited harm to human cells. 
Building new chemical helpers
The team focused on a family of ring‑shaped molecules called triazoloquinoxalines, structures already known to interact well with biological targets. Using a five‑step laboratory route, they crafted ten new versions of these molecules, each carrying a different small chemical tail. These tails ranged from simple carbon chains to alcohol‑containing groups and bulkier side arms. The researchers then used a suite of analytical tools—such as infrared light absorption and nuclear magnetic resonance—to confirm that each step of the synthesis had produced the intended structure and that the new rings were correctly assembled.
Testing the fight against common microbes
Once the structures were confirmed, the compounds were tested against several medically important microbes: two staph bacteria (Staphylococcus aureus and Staphylococcus epidermidis), two gut‑related bacteria (Escherichia coli and Pseudomonas aeruginosa), and the yeast Candida albicans. Three members of the family—labeled 5b, 5d, and 5h—showed the strongest ability to slow or stop growth, although they were less potent than standard antibiotics on their own. Subtle changes in their chemical tails made a big difference: a branched carbon side chain and an alcohol group helped these molecules slip through bacterial defenses and interact more effectively, while long straight chains or bulky ring systems tended to blunt their activity.
Working together with an existing antibiotic
Because none of the new compounds alone matched the strength of current drugs, the researchers turned to combination therapy. They mixed 5b, 5d, and 5h with levofloxacin and measured how much of each was needed to halt bacterial growth. The results revealed strong synergy: when paired, far lower amounts of both the helper molecules and levofloxacin were required to control bacteria, including hospital‑derived strains of methicillin‑resistant S. aureus (MRSA) and resistant P. aeruginosa. In some cases, the effective dose of levofloxacin dropped by more than an order of magnitude. Electron microscope images backed this up, showing that bacteria exposed to the combinations had crumpled, ruptured surfaces and leaking contents, in stark contrast to the smooth, intact cells seen in untreated samples. 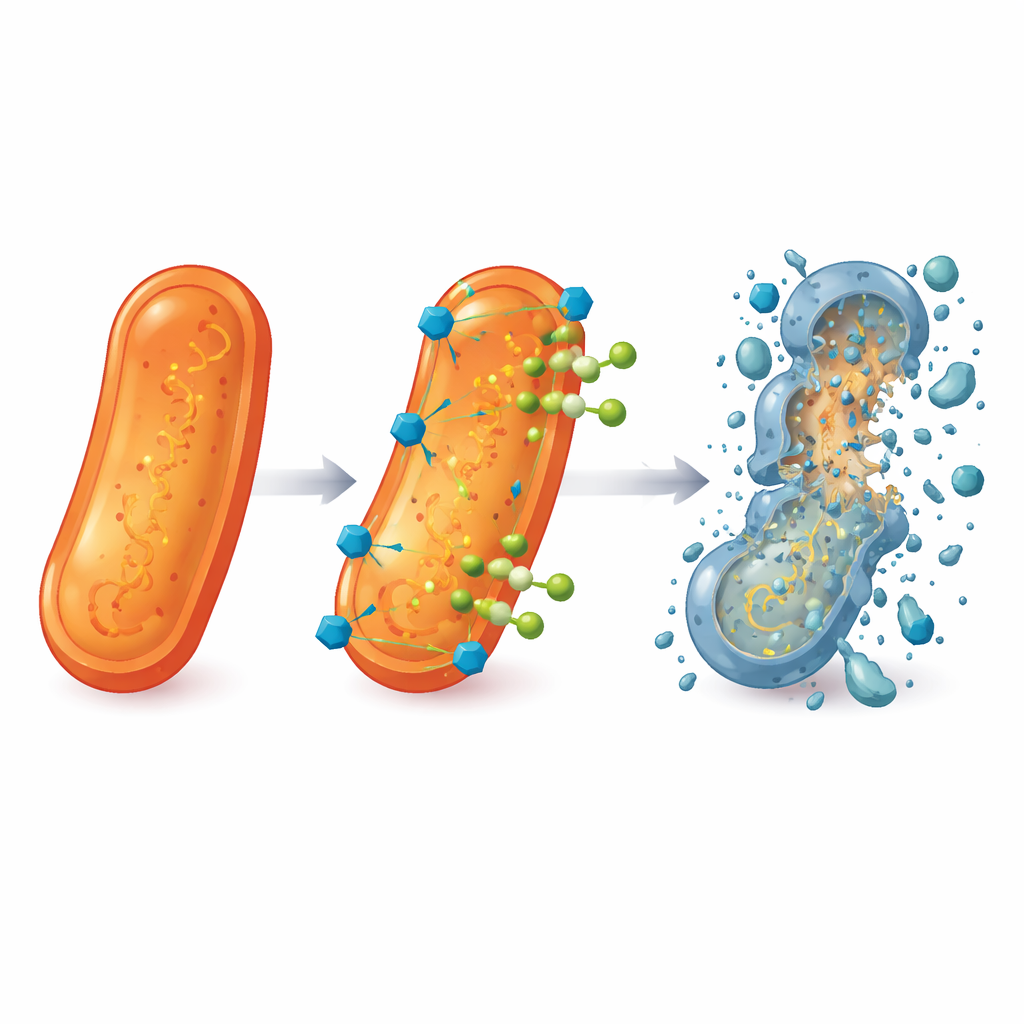
Checking safety for human cells
Any potential helper drug must harm microbes more than it harms us. To gauge this balance, the team exposed mouse connective‑tissue cells and red blood cells to various doses of the three best compounds. At levels equal to or twice those needed to inhibit bacteria, compounds 5b and 5d left most mammalian cells alive and caused almost no damage to red blood cells. Compound 5h was somewhat more irritating at these doses but still showed limited harm at the concentrations used for antibacterial tests. Only at much higher levels did all three compounds become clearly toxic, suggesting there is a useful window where bacteria are hit hard while human cells remain mostly unharmed.
What this means for future treatments
Put simply, the study shows that carefully designed chemical partners can make an existing antibiotic work better against both ordinary and drug‑resistant microbes. The triazoloquinoxaline helpers 5b, 5d, and 5h do not replace levofloxacin; instead, they appear to soften up bacterial defenses—likely by disturbing cell walls or targeting other internal systems—so that the antibiotic can finish the job at lower doses. For patients, such combinations could one day mean more effective treatments with fewer side effects and a slower march toward resistance. Before that happens, however, these promising helpers must still be tested in animals and, eventually, in clinical settings to confirm their safety, stability, and real‑world impact on hard‑to‑treat infections.
Citation: Harooni, N.S., Naimi-Jamal, M.R., Dekamin, M.G. et al. Assessment of antimicrobial activity of [1,2,4]triazolo[4, 3-a]quinoxaline derivatives individually and in combination with levofloxacin. Sci Rep 16, 9902 (2026). https://doi.org/10.1038/s41598-026-39141-y
Keywords: antimicrobial resistance, combination therapy, levofloxacin, heterocyclic compounds, drug synergy