Clear Sky Science · en
Targeting oxidative stress and mitochondrial dysfunction via URG7 overexpression in an in vitro Parkinson’s disease neuronal model
Why protecting brain cells matters
Parkinson’s disease slowly robs people of movement and independence, and today’s treatments can only ease symptoms rather than stop the damage. Many scientists suspect that a major culprit is an internal "rusting" of brain cells, driven by harmful oxygen-based molecules that batter the cell’s energy factories, the mitochondria. This study explores a little-known protein called URG7 and asks a simple but important question: if we boost this protein inside nerve-like cells, can it help them withstand this kind of internal wear and tear?
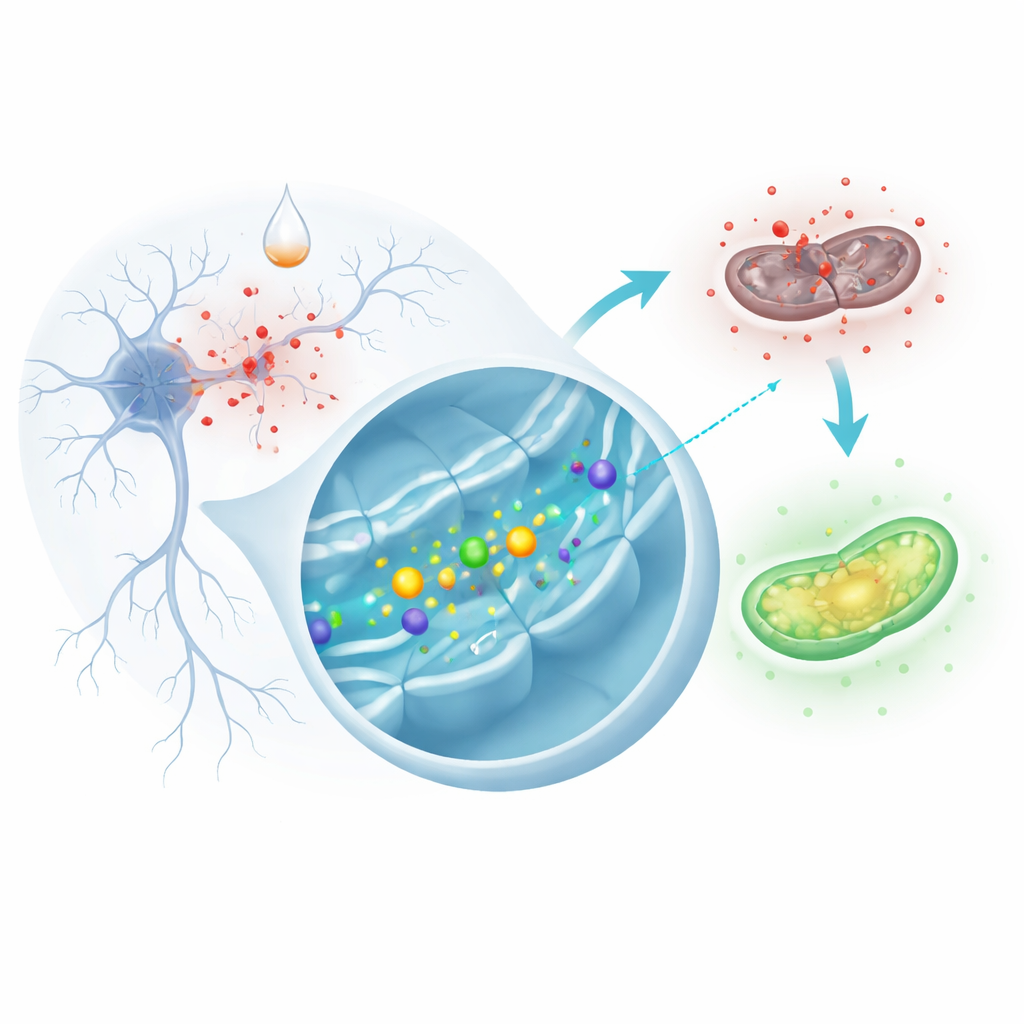
A lab stand‑in for Parkinson’s damage
Because we cannot easily watch living human brain cells degenerate, researchers often use cultured cells as a stand‑in. Here, the team used SH‑SY5Y cells, a human cell line that can mimic many features of neurons. They exposed these cells to a chemical called 6‑hydroxydopamine, which is widely used to reproduce key aspects of Parkinson’s disease in the lab. Once inside the cells, this compound rapidly generates reactive oxygen species—short‑lived, highly reactive molecules that can damage proteins, fats, and DNA—leading to mitochondrial failure and ultimately cell death. The scientists compared ordinary cells with cells engineered to make extra URG7, a small protein that sits in the endoplasmic reticulum, the cell’s protein‑processing and signaling hub.
Fewer dying cells and calmer oxidative stress
When both cell types were challenged with increasing doses of the toxic compound, the URG7‑rich cells survived significantly better. Under the microscope, control cells began to round up, lose their branch‑like extensions, and detach from the dish—a classic picture of stressed, dying neurons—while URG7‑overexpressing cells largely kept their shape and numbers. Measurements of reactive oxygen species confirmed this visual impression: in control cells, oxidative signals rose steeply with toxin dose, but in URG7 cells they stayed close to baseline, especially at the dose chosen for deeper study. This suggested that URG7 does not just passively sit in the cell; it actively helps restrain the surge of damaging oxidants.
Boosting the cell’s own defenses
To understand how URG7 exerts this protective effect, the researchers looked at key components of the cell’s internal defense system. They examined enzymes such as catalase and superoxide dismutase 2, which safely break down harmful oxygen species, and the master control protein Nrf2, which switches on many detoxifying genes. In URG7‑overexpressing cells, all three were elevated at the RNA and protein levels, and catalase activity itself increased instead of dropping after toxin exposure. Markers of damage told the same story from the opposite angle: levels of malondialdehyde, a by‑product of fat molecules damaged by oxidation, rose sharply in control cells but were much lower when URG7 was present. Together, these findings indicate that URG7 acts as an upstream coordinator, turning up the volume on the cell’s own antioxidant toolkit rather than directly neutralizing oxidants.
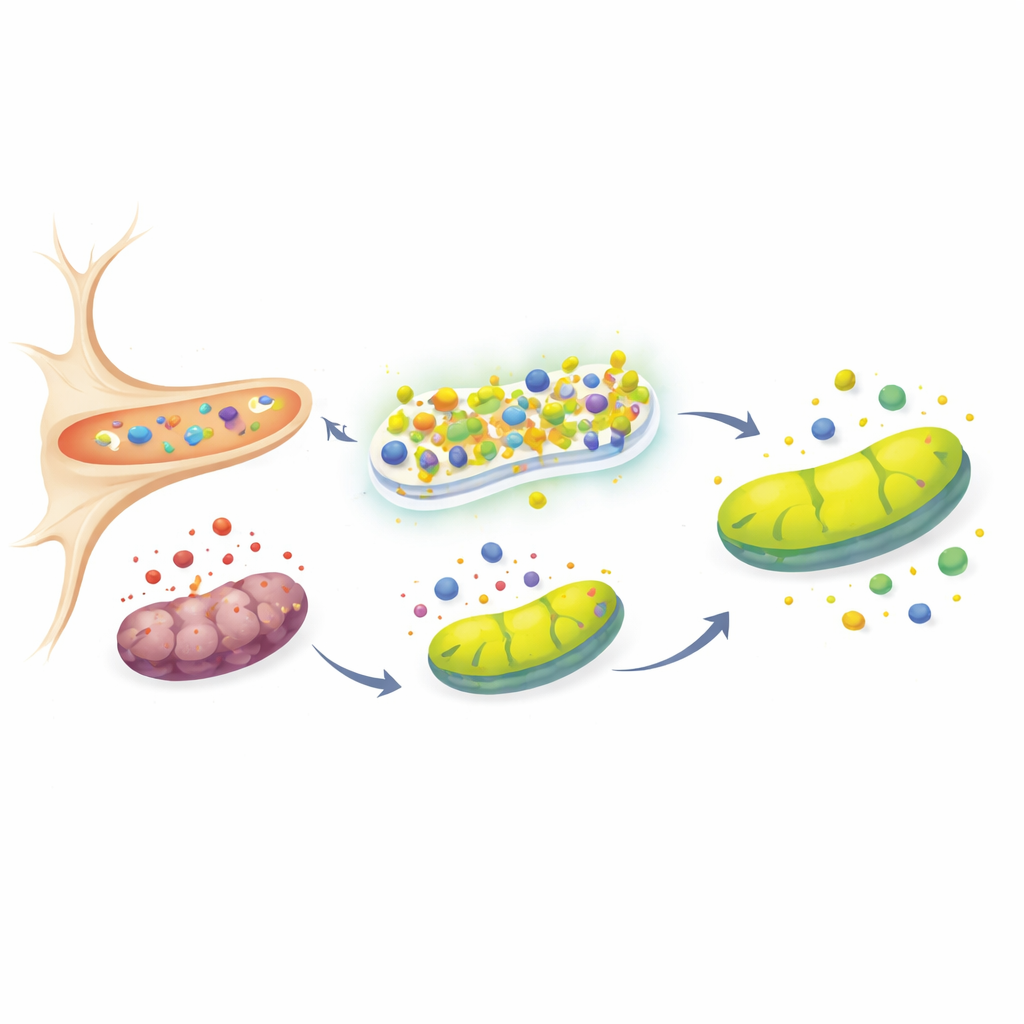
Keeping cellular power plants running
Mitochondria, often called the cell’s power plants, are both a major source and a prime victim of oxidative stress. A key indicator of their health is the electrical potential across their membrane, which drops when they are damaged. After toxin treatment, control cells showed a clear loss of this potential, signaling mitochondrial distress, whereas URG7‑rich cells maintained near‑normal values. The team then examined proteins closely tied to mitochondrial quality control and Parkinson’s disease: PINK1, Parkin, and DJ‑1. In healthy cells, PINK1 is continuously cleared from mitochondria; it builds up only when these organelles are damaged. In URG7‑overexpressing cells, PINK1 remained low despite the toxin, consistent with preserved mitochondrial integrity. At the same time, Parkin and DJ‑1—both associated with supporting mitochondrial function and antioxidant defense—were upregulated, hinting that URG7 helps sustain a broader protective network that stabilizes mitochondria and limits cell death.
What this could mean for future therapies
Put simply, this work suggests that URG7 behaves like an early‑warning sentinel inside nerve‑like cells. When oxidative stress rises, URG7‑rich cells are better able to switch on protective genes, ramp up detoxifying enzymes, and keep their energy factories intact, greatly improving their chances of survival in a Parkinson’s‑like environment. While these experiments were done in a dish and not in living brains, they highlight URG7 as a promising new target for treatments designed to shield neurons from oxidative damage and mitochondrial failure—two central drivers of Parkinson’s disease and other neurodegenerative disorders.
Citation: Nigro, I., Miglionico, R., Lela, L. et al. Targeting oxidative stress and mitochondrial dysfunction via URG7 overexpression in an in vitro Parkinson’s disease neuronal model. Sci Rep 16, 9955 (2026). https://doi.org/10.1038/s41598-026-38925-6
Keywords: Parkinson’s disease, oxidative stress, mitochondrial protection, neuroprotection, URG7 protein