Clear Sky Science · en
Physcion-8-O-β-D-monoglucoside protects hepatocytes from TNF-α-mediated apoptosis by suppressing the PI3K/AKT/NF-κB signaling pathway
Why this matters for liver health
Liver disease is a growing global problem, and many cases begin with sudden, intense damage to liver cells known as acute liver injury. This kind of injury can quickly lead to liver failure if not controlled, yet current treatments are limited. The study behind this article explores a natural compound, derived from a traditional Chinese medicinal herb, that may help shield liver cells from one of the body’s own most damaging inflammatory signals.
A plant compound with a modern twist
The compound at the center of this work is called physcion-8-O-β-D-monoglucoside (PMG), found in Radix et Rhizoma Rhei, a rhubarb-like root used in traditional Chinese medicine for liver ailments. Earlier research suggested that crude extracts and related molecules could protect the liver, but they were poorly absorbed and their precise actions were unclear. PMG is a more absorbable form, reaching higher levels in the bloodstream after oral dosing. The authors previously saw that PMG could lessen toxin-induced liver damage in mice, and here they set out to uncover exactly how it works at the cellular and molecular level.
Turning down a dangerous alarm signal
A key player in acute liver injury is tumor necrosis factor-alpha (TNF-α), a powerful alarm signal released by immune cells. TNF-α docks onto a receptor on liver cells called TNFR1 and can push those cells toward programmed death and inflammation. Using both cultured mouse liver cells and mice exposed to a liver-damaging chemical (carbon tetrachloride), the researchers showed that PMG helps these cells survive TNF-α–driven stress. In cell dishes, PMG did not harm healthy cells on its own, but when TNF-α and a sensitizing drug were added, PMG restored cell survival, improved cell shape, and reduced leakage of liver enzymes—markers of cell damage—into the culture fluid.
Evidence from injured mouse livers
In live mice, carbon tetrachloride caused classic signs of acute liver injury: disruption of tissue structure, widespread cell death, and surging levels of liver enzymes (AST and ALT) in the blood. Animals pretreated with PMG had markedly healthier liver tissue, lower enzyme levels, and fewer dying cells by multiple microscopic staining methods. PMG also reduced the liver’s production of TNF-α itself and another inflammatory messenger, interleukin-6. When the team forced the liver to make extra TNF-α using a gene-delivery virus, the benefits of PMG largely disappeared. This reversal strongly suggests that PMG’s protective effects depend on keeping TNF-α and its downstream consequences in check.
Blocking a harmful chain reaction inside cells
To understand how PMG tames the TNF-α signal, the researchers looked at a well-known chain of molecular events inside cells, the PI3K/AKT/NF-κB pathway. When overactivated, this pathway boosts inflammatory gene activity and encourages cell death. The team used computer-based molecular docking to show that PMG can bind directly to the TNFR1 receptor, supporting the idea that it interferes at the very first step of signaling. In mouse livers damaged by carbon tetrachloride, levels of activated PI3K and AKT and nuclear NF-κB were all elevated, as was the executioner protein cleaved caspase-3, a hallmark of cells committing suicide. PMG treatment dampened all of these signals and reduced caspase-3 activation. However, when TNF-α was artificially overproduced, these protective changes were lost and the signaling pathway flared back up.
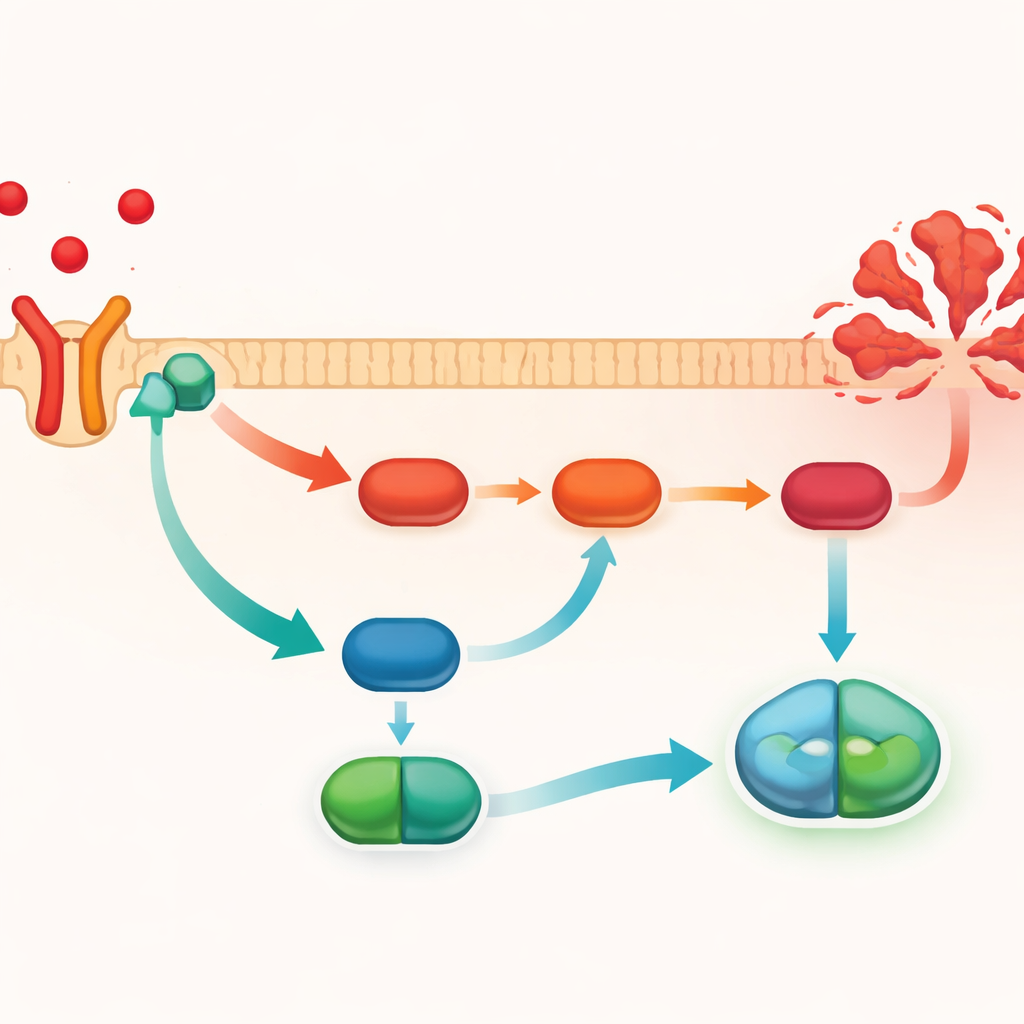
What this could mean for future treatments
Taken together, the findings paint a clear picture: PMG appears to shield liver cells from acute inflammatory injury by blocking the TNF-α signal at its receptor and by calming the PI3K/AKT/NF-κB pathway that would otherwise drive inflammation and cell death. While this work was done in mice and cultured cells, it offers a mechanistic basis for a plant-derived molecule as a potential drug candidate for acute liver injury. Further studies will be needed to test safety, dosing, and whether similar benefits occur in humans, but the study highlights how traditional remedies can inspire targeted modern therapies for protecting one of the body’s most vital organs.
Citation: Hu, R., Chen, Z., Chen, T. et al. Physcion-8-O-β-D-monoglucoside protects hepatocytes from TNF-α-mediated apoptosis by suppressing the PI3K/AKT/NF-κB signaling pathway. Sci Rep 16, 12716 (2026). https://doi.org/10.1038/s41598-026-38701-6
Keywords: acute liver injury, inflammation, tumor necrosis factor, traditional Chinese medicine, liver protection