Clear Sky Science · en
Nrf2/SLC7A11/GPX4 pathway, a potential target of oridonin in inhibiting ferroptosis during cerebral ischemia-reperfusion injury progression
Why a Stroke Can Hurt Twice
When a person has an ischemic stroke, a clot blocks blood flow to part of the brain. Doctors race to reopen the vessel, but the very act of restoring blood can cause its own wave of damage, a phenomenon called reperfusion injury. This study explores whether oridonin, a naturally occurring compound from a traditional Chinese medicinal herb, can shield brain cells from this second hit of injury by calming a destructive, iron-driven form of cell death.
A Plant Compound Under the Microscope
Oridonin is extracted from the herb Rabdosia rubescens, long used in traditional Chinese medicine and already known for anti-inflammatory and antioxidant effects. Researchers suspected it might also protect the brain during the critical window after a stroke. To test this idea, they used two complementary models: rats subjected to a brief blockage of a major brain artery followed by restored blood flow, and cultured nerve-like cells exposed to a period without oxygen and sugar followed by reoxygenation. Together, these systems mimic what happens to brain tissue when a clot is first starved of blood and then suddenly reperfused in the clinic.
When Iron and Oxidants Turn Deadly
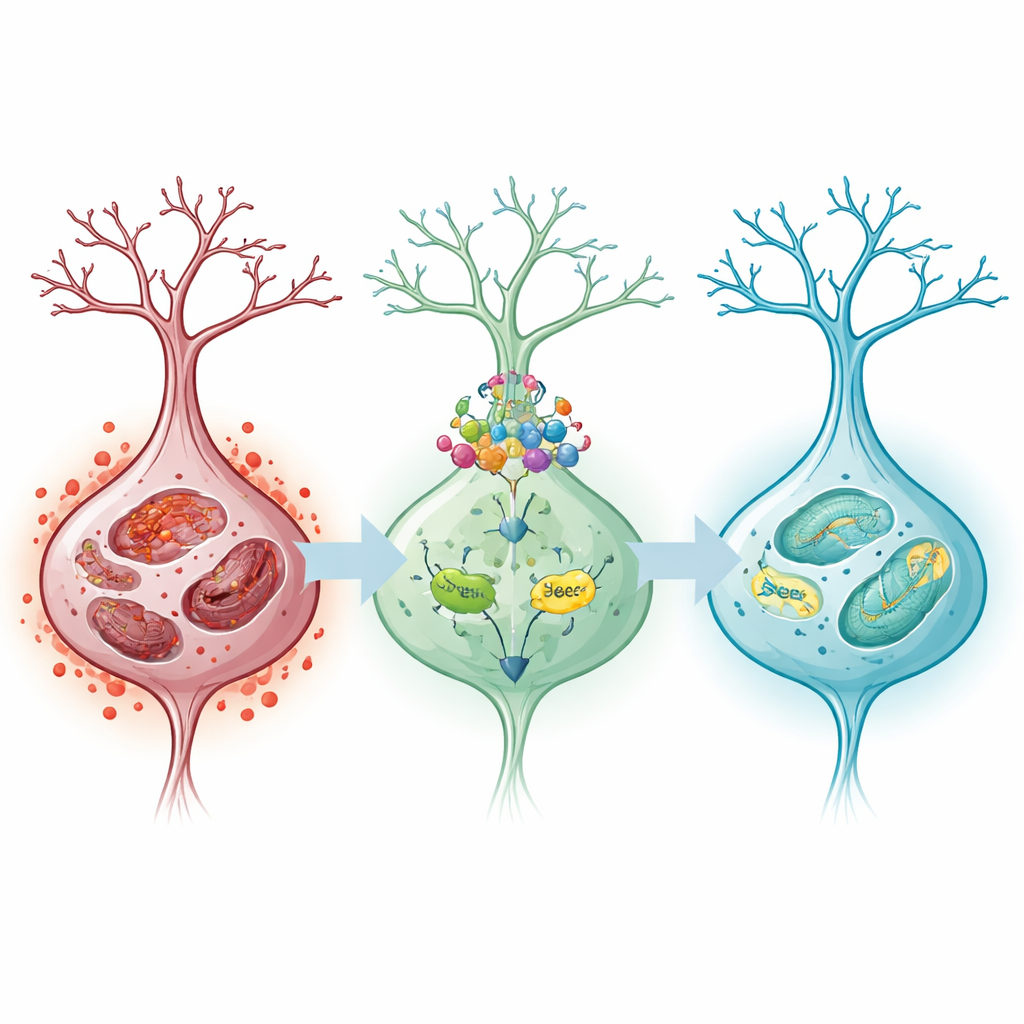
The team focused on ferroptosis, a recently recognized type of cell death fueled by iron buildup and runaway oxidation of fats in cell membranes. In rats, longer periods of restored blood flow led to worse neurological scores, larger brain infarcts, and more disorganized, dying neurons. The brain tissue accumulated iron and chemical hallmarks of oxidative damage, while the mitochondria—the cell’s energy organelles—became distorted and vacuolated. At the same time, a key protective signaling route, involving the proteins Nrf2, SLC7A11, and GPX4, steadily weakened. This pathway normally helps cells keep iron and oxidants in check; when its activity fell, pro–ferroptosis markers rose, indicating that ferroptosis was strongly engaged after reperfusion.
How Oridonin Changes the Picture
Pre-treating rats with oridonin before the artery blockage changed these outcomes in a dose-dependent fashion. Animals receiving higher doses had better blood flow restoration, smaller stroke areas, and improved short-term neurological behavior. Their neurons retained more normal structure and fewer iron deposits, and the biochemical signals of ferroptosis—reactive oxygen molecules, iron overload, and lipid breakdown products—were all reduced. In both rat brains and cultured cells, oridonin boosted the levels of Nrf2, SLC7A11, and GPX4 while dampening proteins that promote ferroptosis. Mitochondria appeared healthier and more intact under the electron microscope, suggesting that the compound helped preserve core cellular machinery during the stress of reperfusion.
Probing the Cell’s Defense Switch
To probe whether this protective pathway was truly central to oridonin’s action, the researchers selectively reduced Nrf2 in the cultured nerve-like cells. When Nrf2 was silenced, oridonin could no longer fully restore the balance of iron and antioxidants: damaging molecules and iron levels climbed again, cell survival dropped, and cell death increased. This reversal indicated that oridonin’s benefits depend strongly on Nrf2’s ability to turn on downstream defenses, including SLC7A11 and GPX4, which together help maintain glutathione, a crucial cellular antioxidant, and prevent membrane fats from being peroxidized.
What This Could Mean for Future Stroke Care
For a layperson, the core message is that this work identifies a natural molecule that helps brain cells weather the violent chemical storm that follows restored blood flow after a stroke. By strengthening an internal defense circuit that controls iron handling and antioxidant reserves, oridonin reduces a specific, iron-dependent form of cell death and limits early brain damage in animal and cell models. The study does not yet prove that oridonin is safe or effective in people, nor does it address long-term recovery, but it offers a promising blueprint: targeting the Nrf2–SLC7A11–GPX4 pathway to tame ferroptosis may one day complement clot-busting and clot-removal therapies, turning lifesaving reperfusion into a safer rescue rather than a double-edged sword.
Citation: Zhang, D., Shao, L., He, M. et al. Nrf2/SLC7A11/GPX4 pathway, a potential target of oridonin in inhibiting ferroptosis during cerebral ischemia-reperfusion injury progression. Sci Rep 16, 14597 (2026). https://doi.org/10.1038/s41598-026-38658-6
Keywords: ischemic stroke, reperfusion injury, ferroptosis, oridonin, neuroprotection