Clear Sky Science · en
Entorhinal cortex represents task-relevant remote locations independently of CA1
How the brain links places when we pause
Imagine standing at your front door while picturing the kitchen where you left your coffee. Our brains constantly jump between where we are and where we need to go. This study asked how a key navigation area in the mouse brain, the medial entorhinal cortex, handles such mental jumps between locations while the animal is still, and what that might mean for how we learn routes and goals.
A maze that tests memory for place
To probe this question, researchers trained mice on an X-shaped maze where each trial linked two rewarded spots. A mouse first visited a "sample" arm to get a small treat, then chose between two "choice" arms to earn a larger reward according to a rule: go to the arm on the same side of the maze, and later to the arm on the opposite side after the rule flipped. Over many days, mice learned these changing rules while running about a hundred trials per session. As they performed the task, ultra-thin Neuropixels probes recorded the activity of hundreds of individual nerve cells in the medial entorhinal cortex and in a nearby region, hippocampal CA1, letting the team decode where in the maze each population of cells was representing at every moment.
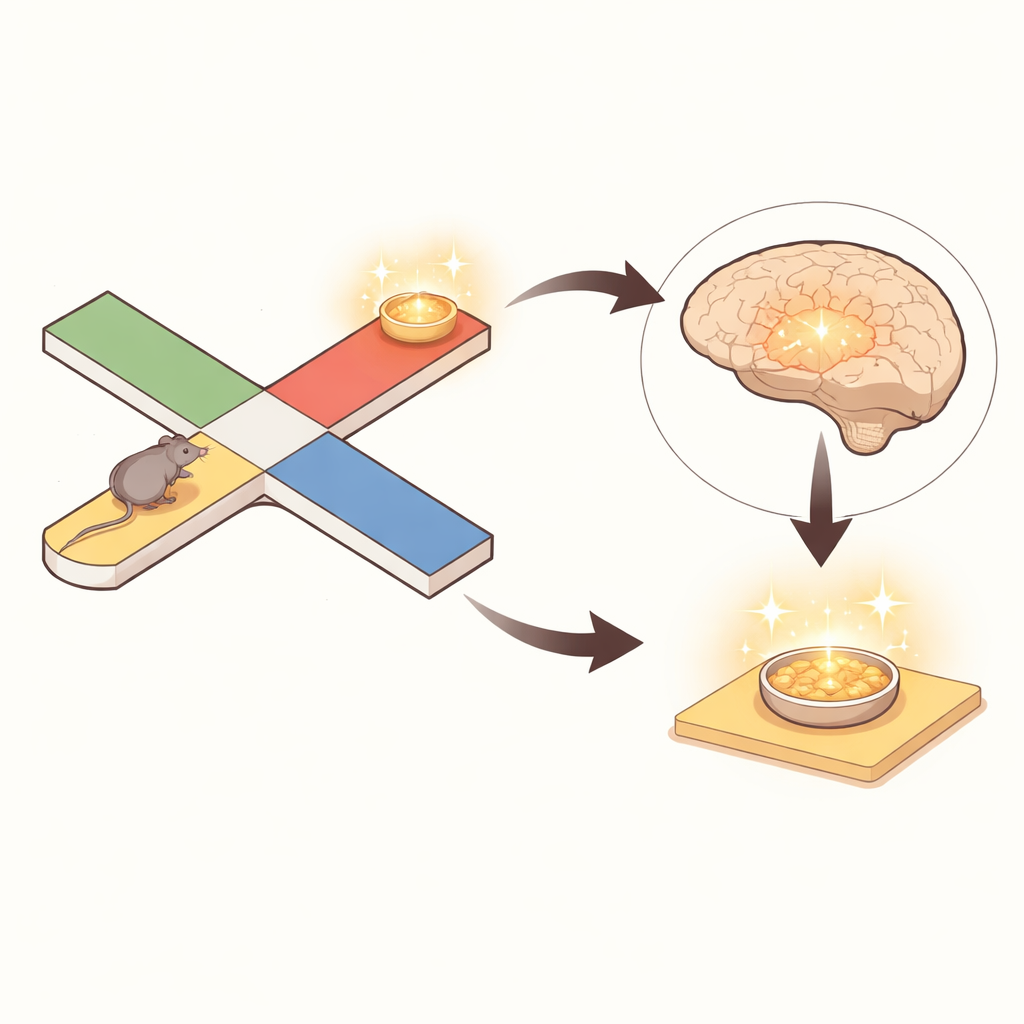
The brain’s map jumps during still moments
During movement, activity patterns in the entorhinal cortex closely tracked the mouse’s actual position along the maze, much like a moving dot on a GPS display. But when the mouse paused, something striking happened: the decoded position from entorhinal activity often “jumped” to locations far away from the animal’s body, frequently to the opposite side of the maze. The authors defined these jumps as "nonlocal" coding when the represented spot lay at least 20 centimeters away. Nearly half of all pauses contained such nonlocal content, and about a quarter of all still-time bins reflected remote spots. Importantly, this was not a decoding accident. Cells tuned to the current physical location kept firing, but cells whose preferred spots lay at remote maze locations increased their firing during these bouts and drove the readout toward those distant positions.
Independent snapshots, not classic replay
Previous work has shown coordinated “replay” of past paths during brief electrical events in the hippocampus called sharp-wave ripples, which are thought to support memory. Here, entorhinal cortex sometimes represented far-away positions during these ripples, but most of the nonlocal coding occurred outside them. When the team compared entorhinal and CA1 activity, they found that the two regions were less synchronized during nonlocal periods than during local ones. CA1 was less likely to represent the same location as entorhinal cortex, pairs of cells between regions fired together less often, and a fast rhythm thought to carry entorhinal input into CA1 was weaker. These results suggest that, during many of these mental jumps, entorhinal cortex is running its own internal map with limited influence on hippocampal output.
Thinking about the right place at the right time
The content of these remote representations was far from random. Mice tended to pause longer at reward spots, but nonlocal coding was especially likely when they were elsewhere on the maze, such as in the central arm. Regardless of where the animal actually sat, remote representations favored rewarded locations much more than chance. When a mouse lingered at a sample reward, its entorhinal activity more often represented the choice reward that was correctly paired with that sample on successful trials, and shifted toward the unpaired reward on errors. Likewise, when resting at a choice reward after a correct decision, the entorhinal map more often jumped back to the corresponding sample reward. When the rule of the task was flipped so that the “correct” partnership between arms changed, the preferred remote representations flipped in step with the new rule.
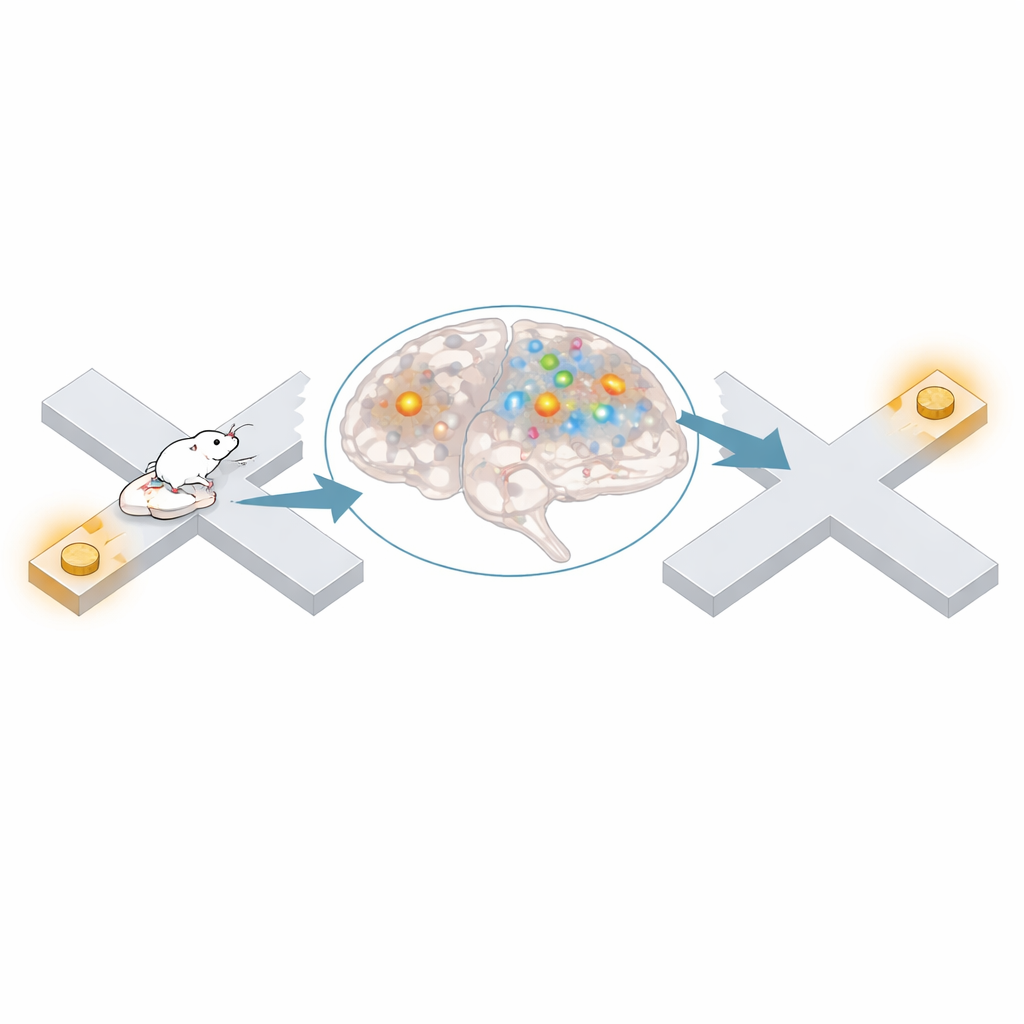
Why this matters for everyday navigation
These results indicate that the medial entorhinal cortex often represents task-relevant, but physically distant, places when an animal is still, and that these mental jumps are tuned to the correct partnerships between locations. While another brain area, CA1, largely disengages during these events, entorhinal cortex may quietly strengthen links between paired spots or help the animal call up a useful destination when deciding where to go next. In everyday terms, when you pause at one landmark while remembering another, this study suggests that your brain’s internal map may be actively connecting those places in the background, even when you are not moving.
Citation: Aery Jones, E.A., Low, I.I.C., Cho, F.S. et al. Entorhinal cortex represents task-relevant remote locations independently of CA1. Nat Neurosci 29, 1181–1190 (2026). https://doi.org/10.1038/s41593-026-02232-0
Keywords: spatial navigation, entorhinal cortex, memory replay, mouse maze, hippocampus