Clear Sky Science · en
Tunable hydrogel-based micropillar arrays for myelination studies
Why tiny pillars may help fix damaged nerves
Diseases like multiple sclerosis damage the insulating coat around nerve fibers, slowing brain signals and causing problems with movement, vision and thinking. The body has cells that can rebuild this insulation, but scientists still struggle to predict which drugs will truly help them do the job. This paper describes a clever lab-made landscape of tiny, soft pillars that imitate nerve fibers, giving researchers a much more realistic way to watch and measure how brain cells rebuild insulation—and to test future treatments safely and efficiently.
Building a fake forest of nerve-like posts
The team created a platform made of a water-rich gel, a bit like very firm Jell‑O, dotted with thousands of upright microscopic pillars. These pillars stand in for nerve fibers in the brain. By using standard chip-making techniques, the researchers could precisely tune how wide each pillar was, how far apart pillars sat from one another and how stiff or soft they were. The range spans from brain‑like softness to much stiffer conditions, similar to other body tissues, while keeping the shapes at sizes that match real nerve fibers. This grid of uniform pillars sits in a small well, so only modest numbers of cells and little liquid are needed for experiments.
Convincing brain cells to wrap the pillars
Next, the researchers placed oligodendrocytes—specialized brain cells that normally wrap nerve fibers in myelin—onto these pillar arrays. Over two weeks, they watched the cells grow, change and eventually send out long processes that curled around the pillars. With advanced microscopes, including 3D confocal imaging and electron microscopy, they saw multiple compact layers of insulation forming around many of the gel posts, very similar to natural myelin in the brain. More than half the pillars carried multilayered wraps, and the thickness of the insulating layer closely matched the number of turns the cell had made, confirming that a simple fluorescent stain could reliably stand in for more laborious ultrastructural checks.
How shape, spacing and softness steer insulation
Because the platform is so tunable, the team could systematically probe which physical features matter most. They varied pillar width and spacing to echo the diversity of nerve fiber sizes in real brain tissue. Thicker pillars were wrapped more frequently and more completely, mirroring the way larger nerve fibers are preferentially insulated in living brains. When pillars were very thin but packed too closely, each cell had more potential targets than it could handle, and wrapping efficiency dropped. The ratio of pillar size to coating thickness—the so‑called g‑ratio used by neuroscientists—fell into the range seen in healthy central nervous system tissue, suggesting the artificial system captures key aspects of natural design.
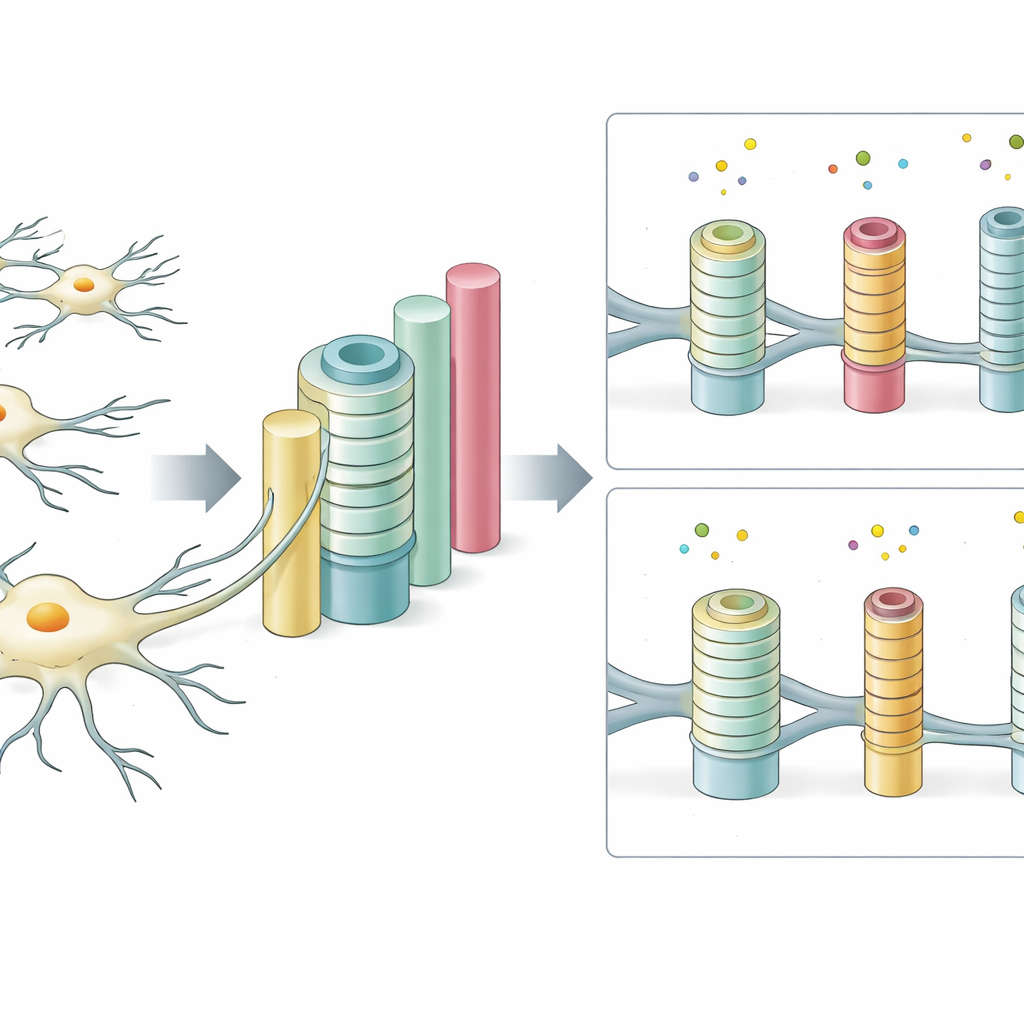
The feel and surface of the landscape change cell behavior
Beyond geometry, the “feel” of the environment also strongly influenced how well cells formed insulation. On very soft pillars that mimicked squishy brain tissue, some wrapping occurred, but it was reduced for certain pillar sizes. As pillars became firmer, wrapping generally increased, especially on larger posts. The researchers also changed the molecules decorating the pillar surface. Coating with laminin, a natural component of the brain’s support mesh, boosted wrapping, while fibronectin altered how many pillars each cell could fully surround. When they combined changes in softness and surface chemistry, they saw that both factors jointly controlled how many pillars were fully insulated, underlining how sensitive these cells are to subtle physical and chemical cues.
Revealing drug effects—and false promises
The team then turned the system into a test bed for potential medicines. They applied several compounds previously proposed to enhance myelin repair, along with one that is known to interfere with the process. The platform picked up clear increases in wrapping for candidate “pro‑myelinating” drugs and dose‑dependent decreases for the inhibitor. Crucially, the apparent strength of some drugs depended on how stiff the pillars were. On stiffer posts, drugs looked more potent; on softer, brain‑like ones, their benefits were smaller. This suggests that older, overly rigid lab models may have exaggerated the promise of certain compounds, helping explain why some failed in clinical trials despite encouraging early data.
Bringing human biology into the picture
To make the system more relevant to patients, the researchers showed it works not only with rodent cells but also with human cells derived from fetal tissue and human stem cells. These human oligodendrocytes extended long, intricate processes and wrapped the gel pillars, and in the case of stem‑cell‑derived cells even formed compact, multilayered insulation. That means the platform can be paired with modern stem cell technologies, including patient‑specific lines, to model human diseases that affect myelin and to search for tailored treatments.
What this means for future treatments
In simple terms, this work provides a realistic miniature playground where insulation‑forming brain cells can interact with lifelike “fake nerves.” Because the size, spacing, softness and surface of the pillars can all be controlled, scientists can dissect how each feature shapes myelin repair and how potential drugs truly behave in a brain‑like setting. By reducing misleading results from overly rigid or flat lab systems and by working well with human cells, this tunable pillar platform could speed the discovery of more reliable therapies for multiple sclerosis and related disorders, bringing the prospect of restoring damaged nerve insulation closer to reality.
Citation: Lasli, S., Vinel, C., Agrawal, A. et al. Tunable hydrogel-based micropillar arrays for myelination studies. Nat Methods 23, 854–864 (2026). https://doi.org/10.1038/s41592-026-03048-3
Keywords: myelination, hydrogel micropillars, oligodendrocytes, mechanobiology, multiple sclerosis