Clear Sky Science · en
Real-world clinical utility of tumor whole-genome sequencing in solid cancers
Why this matters for people with cancer
For many people with advanced cancer, the hardest part of treatment is knowing whether a given drug is actually likely to help. This study looks at a powerful test called whole-genome sequencing, which reads nearly all of the DNA changes in a person’s tumor in one go. The researchers asked a simple but crucial question: when this test is used in everyday hospital care, does it really change what happens to patients?
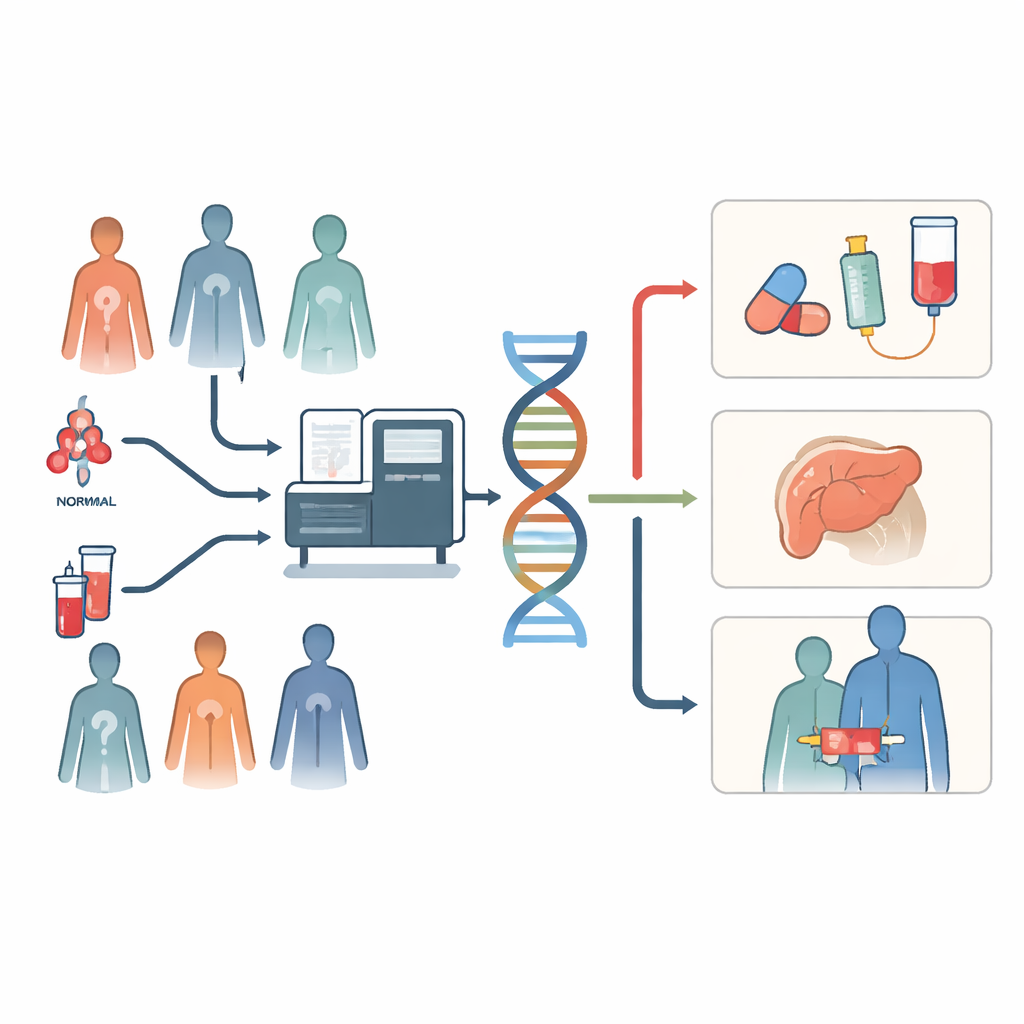
A single test instead of many
Traditional cancer testing often works like a slow relay race. Doctors order one DNA test, wait for the result, then order another, each looking for a different marker that might match a specific drug. This stepwise approach can burn through precious tumor tissue and time. Whole-genome sequencing takes a different tack: from a fresh-frozen tumor sample and a blood sample, it captures almost all DNA changes in a single run. That includes common drug targets, complex rearrangements, signs that a tumor might respond to immunotherapy, and inherited risk variants that could matter for family members. In the Netherlands, one cancer center made this broad test part of routine care for patients with hard-to-treat solid tumors and followed what happened next.
How well the test worked in real life
The team reviewed 1,052 requests for whole-genome sequencing between early 2021 and late 2022. After excluding samples without enough tumor cells or DNA, they obtained successful reports for 793 tumor samples from 723 patients. Most samples came from lung cancers, cancers of unknown primary origin, and soft tissue sarcomas. Turnaround was fast enough for clinical decisions: typically six working days from sample arrival at the sequencing lab to a report back in the hospital. Using the DNA readout, specialists cataloged which tumors had features that could point to existing or experimental treatments and logged those decisions in a molecular tumor board.
Finding more options than standard panels
Across all patients, 73% had at least one “actionable” DNA signal that could guide therapy selection or avoidance. About a quarter had markers linked to drugs already reimbursed as standard care, while nearly two-thirds had markers that could make them eligible for clinical trials or early-access programs. When the researchers digitally mimicked what would have been seen with smaller targeted gene panels, they found that even a large 523-gene test would have missed important opportunities in roughly one in ten patients, particularly complex gene fusions and a pattern called homologous recombination deficiency that points to sensitivity to certain drugs. Whole-genome sequencing also uncovered clinically relevant inherited variants in about 6.5% of patients, half of which had not been picked up by previous genetic testing.
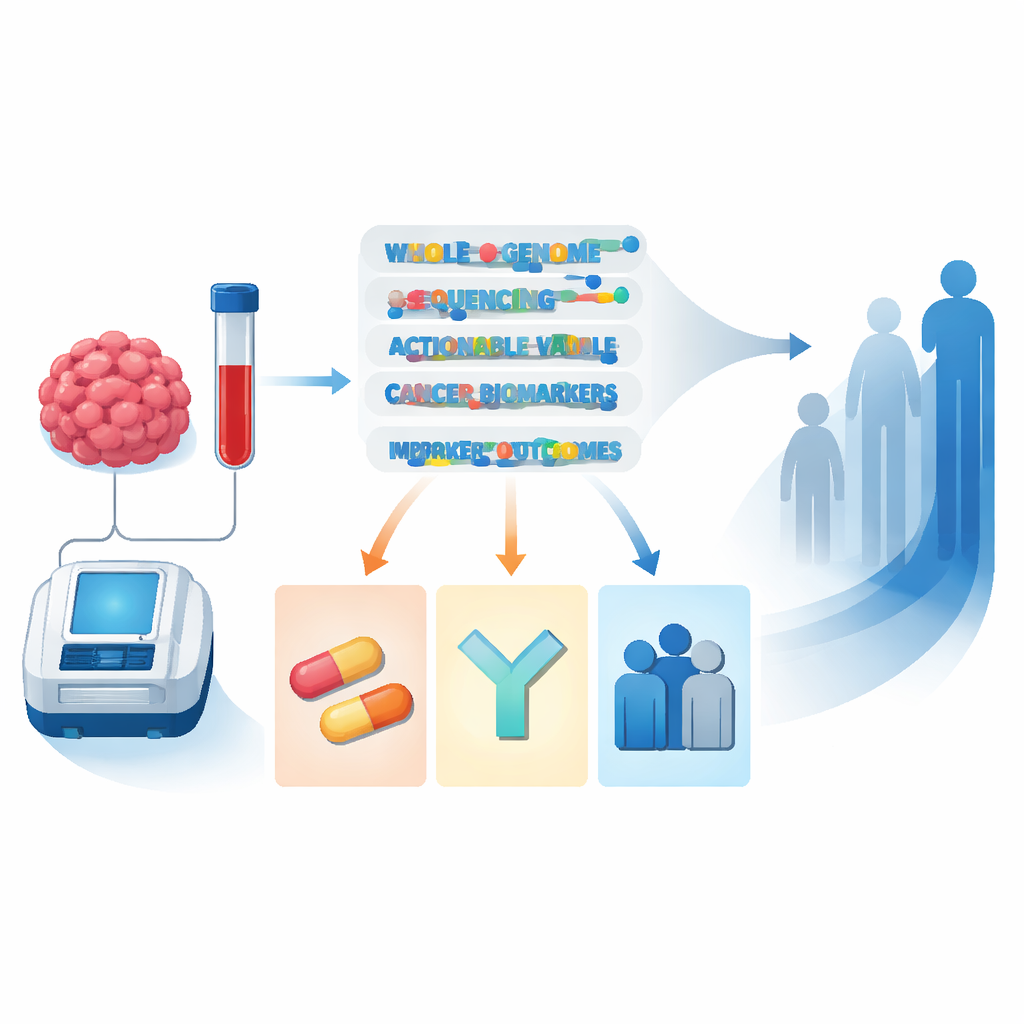
Solving mystery cancers and guiding treatment
A striking benefit emerged for people with cancers of unknown primary origin, where doctors can see metastases but cannot find where the cancer started. In 63% of these cases, the genome test helped pin down a likely tissue of origin or refine the diagnosis, often allowing doctors to switch from vague “unknown primary” regimens to more tailored, tumor-type-specific treatment. Overall, 41% of all tested patients experienced a concrete clinical consequence of the whole-genome report: starting a biomarker-guided drug, entering a trial, receiving a revised or clarified diagnosis, or being referred for genetic counseling. For some patients with established diagnoses, the test even led to reclassification or finer subtyping of their cancer, with direct treatment changes.
What it meant for survival
Because this was not a randomized trial, the researchers could not prove cause and effect, but they could compare groups. Among patients whose tumors carried at least one actionable marker, those who actually received biomarker-informed therapy after whole-genome sequencing lived a median of 405 days after the report, versus 309 days for similar patients who did not receive such treatment—about a 31% gain. The benefit was most pronounced when the genome test was done before any systemic therapy: in this group, survival for patients receiving guided treatment had not yet reached a median at four years, clearly longer than for those treated without genomic guidance or not treated systemically at all. In contrast, when sequencing was delayed until after one or more treatment lines, the survival edge largely disappeared.
What this means going forward
For patients and clinicians, this study shows that reading the entire tumor genome in routine care is not just technically feasible; it frequently alters diagnoses, opens doors to tailored treatments, and is linked to longer survival when used early. Whole-genome sequencing also builds a rich data resource that can help refine cancer care for future patients. The authors argue that as costs fall and data systems improve, this broad DNA test should become a cornerstone of precision oncology rather than a last-resort option.
Citation: van Putten, J., Snaebjornsson, P., Bosch, L.J.W. et al. Real-world clinical utility of tumor whole-genome sequencing in solid cancers. Nat Med 32, 1286–1295 (2026). https://doi.org/10.1038/s41591-026-04280-2
Keywords: whole-genome sequencing, precision oncology, actionable biomarkers, cancer of unknown primary, biomarker-guided therapy