Clear Sky Science · en
A small-molecule inverse agonist of PPARγ for advanced solid tumors: a phase 1 trial
Why a New Approach to Bladder Cancer Matters
Bladder cancer that has spread through the body is notoriously hard to treat. Even with modern immunotherapies and targeted drugs, many patients eventually see their disease worsen. This article describes the first human trial of a pill called FX-909, designed to switch off a key growth program inside many bladder cancer cells. For people following cancer research, it offers a glimpse of a new way to treat tumors by going after the internal "identity" of cancer cells rather than just trying to kill them outright. 
A Hidden Switch Inside Tumor Cells
Many advanced bladder cancers belong to a group called luminal tumors, which share a pattern of gene activity with certain breast cancers. At the heart of this pattern is a protein called PPARγ, a kind of master switch that helps cells behave like normal bladder lining. When this switch is cranked up too high, cancer cells can become dependent on it for survival, much like some breast cancers depend on estrogen signals or prostate cancers on male hormones. Genetic studies of thousands of tumors have shown that luminal bladder cancers frequently carry changes that overactivate this switch, suggesting that turning it off could be a powerful way to slow or shrink these cancers.
Designing a Drug to Flip the Switch Off
Earlier attempts to block PPARγ did not work well, in part because they acted like simple roadblocks, preventing natural signals from binding but failing to truly silence the pathway. FX-909 was built differently. It is a small molecule taken by mouth that latches onto PPARγ in a way that locks it into a tightly repressed state. This altered shape attracts natural "brakes" inside the cell and keeps "accelerators" from attaching, switching off PPARγ-driven gene activity even when strong tumor-promoting signals are present. Experiments in cancer cells and in mice carrying human tumors suggested that this deep shutdown could cause durable tumor shrinkage, especially in cancers that rely heavily on PPARγ. 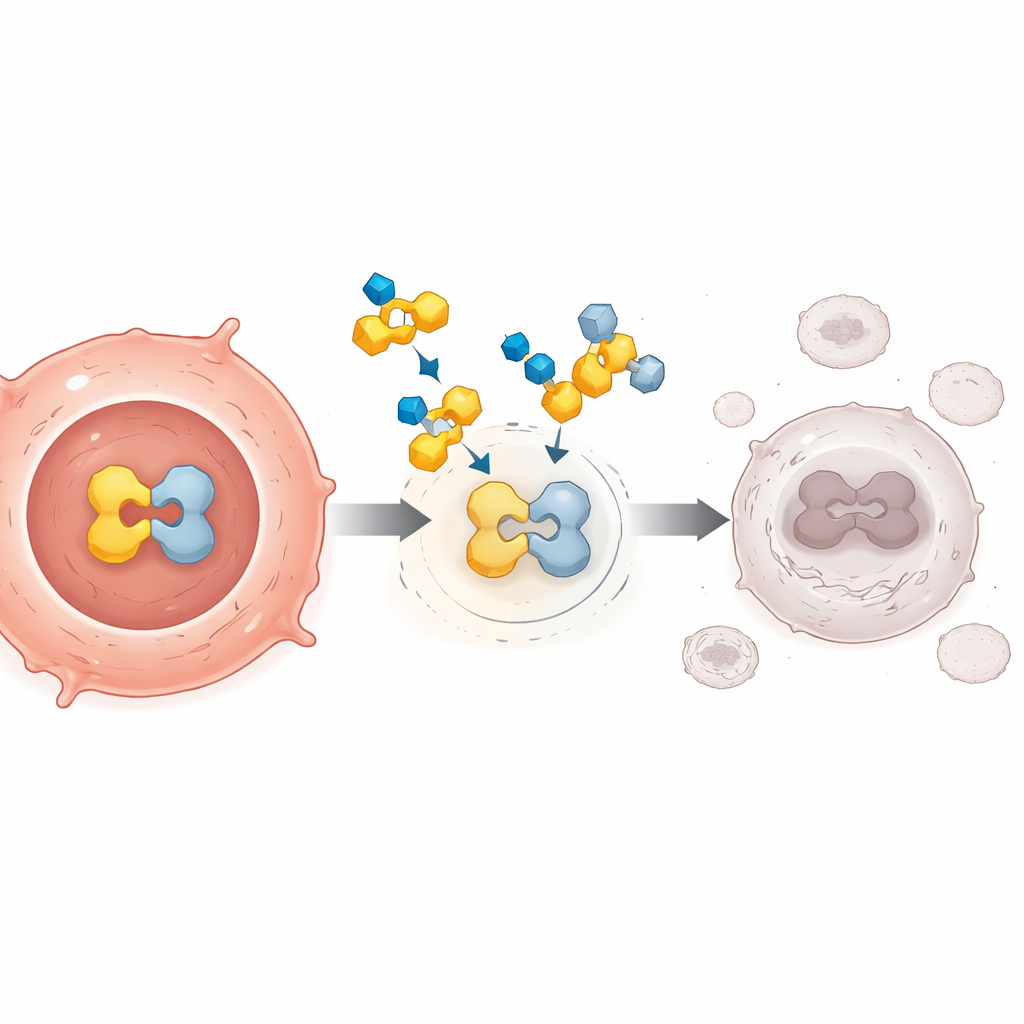
Testing Safety and Early Signs of Benefit
The phase 1A study enrolled 56 people with advanced solid tumors, including 46 with advanced urothelial (bladder and urinary tract) cancer who had already received several lines of treatment. Volunteers took daily doses of FX-909 ranging from 30 to 100 milligrams in repeating 28-day cycles. The main goals were to gauge safety, find dose ranges suitable for further testing and look for any hints that the drug was affecting tumors. Overall, FX-909 had a manageable safety profile. Nearly all patients had some side effects, but serious problems such as low red blood cells, low platelets, fatigue and high blood sugar were generally reversible and became more frequent only at the higher doses. No deaths were attributed to the drug. Based on the overall balance of tolerability, drug levels in the blood and biological activity, daily doses of 30 and 50 milligrams were chosen for further study.
Who Seems Most Likely to Benefit?
Among patients with advanced bladder cancer whose tumors could be measured on scans, seven experienced confirmed shrinkage of their tumors, including one complete disappearance, and many others had prolonged disease stability. Importantly, these responses appeared at all dose levels tested rather than only at the highest doses. To understand who benefited most, the researchers analyzed tumor samples for markers of the luminal program and for the amount of PPARγ protein present in cancer cell nuclei. They developed a simple staining test that scores what fraction of tumor cells show PPARγ in the nucleus. Tumors where at least 60% of cells stained positive were labeled "PPARγ-high." In this subgroup, most patients showed some tumor shrinkage, and five had confirmed responses, some lasting many months.
Tracking Tumors Through the Blood
The team also explored a blood-based "liquid biopsy" that tracks fragments of tumor DNA circulating in the bloodstream. In 29 patients with advanced bladder cancer who had blood samples before and during treatment, more than half showed marked drops in tumor-related DNA after starting FX-909. These molecular responses lined up well with scan results: patients whose blood tumor markers fell were much more likely to have visible tumor shrinkage. Notably, most of these molecular responders also had PPARγ-high tumors, reinforcing the idea that this biomarker can help select patients most likely to benefit from the drug.
What This Could Mean for Future Care
To a lay reader, the main message is that FX-909 represents a new kind of targeted therapy that goes after a core identity program inside many bladder cancers. In this early trial, the drug could be given safely at daily doses of 30 or 50 milligrams, clearly switched off its intended target in patients and showed promising signs of tumor control in people whose cancers had already resisted multiple treatments. The simple tumor staining test for PPARγ offers a practical way to find those patients. Larger, ongoing studies will determine how well FX-909 works compared with existing options and whether this strategy can be expanded to other cancers that share the same overactive growth switch.
Citation: Galsky, M.D., Mantia, C., Bowden, M. et al. A small-molecule inverse agonist of PPARγ for advanced solid tumors: a phase 1 trial. Nat Med 32, 1249–1256 (2026). https://doi.org/10.1038/s41591-026-04263-3
Keywords: urothelial carcinoma, PPAR gamma, targeted therapy, FX-909, phase 1 clinical trial