Clear Sky Science · en
Understanding low-pressure CO2 insertion chemistry in epoxide–CO2 copolymerization catalysis
Turning Waste Gas into Everyday Materials
Carbon dioxide (CO2) is usually seen as a waste gas warming our planet, but chemists are learning how to turn it into useful plastics. The catch is that most current processes need CO2 to be squeezed to high pressures, which costs money and energy. This paper explores how to make CO2-based plastics efficiently at much lower pressures, pointing the way toward greener manufacturing of materials used in adhesives, batteries and flexible plastics.
Why Pressure Matters in Green Chemistry
To recycle CO2 into plastics, chemists use catalysts—special molecules that help link CO2 with small building blocks called epoxides to form long polycarbonate chains. Industrial plants often run these reactions at high CO2 pressures to get good speeds and product quality, but compressing gas is energy-hungry and expensive. The authors use process simulations to show that increasing CO2 pressure from modest to high levels can boost energy use for a key plastic, poly(propene carbonate), by more than 200 percent. That makes low-pressure operation crucial if CO2-based plastics are to be both climate-friendly and commercially attractive.
Probing the Hidden Step in the Reaction
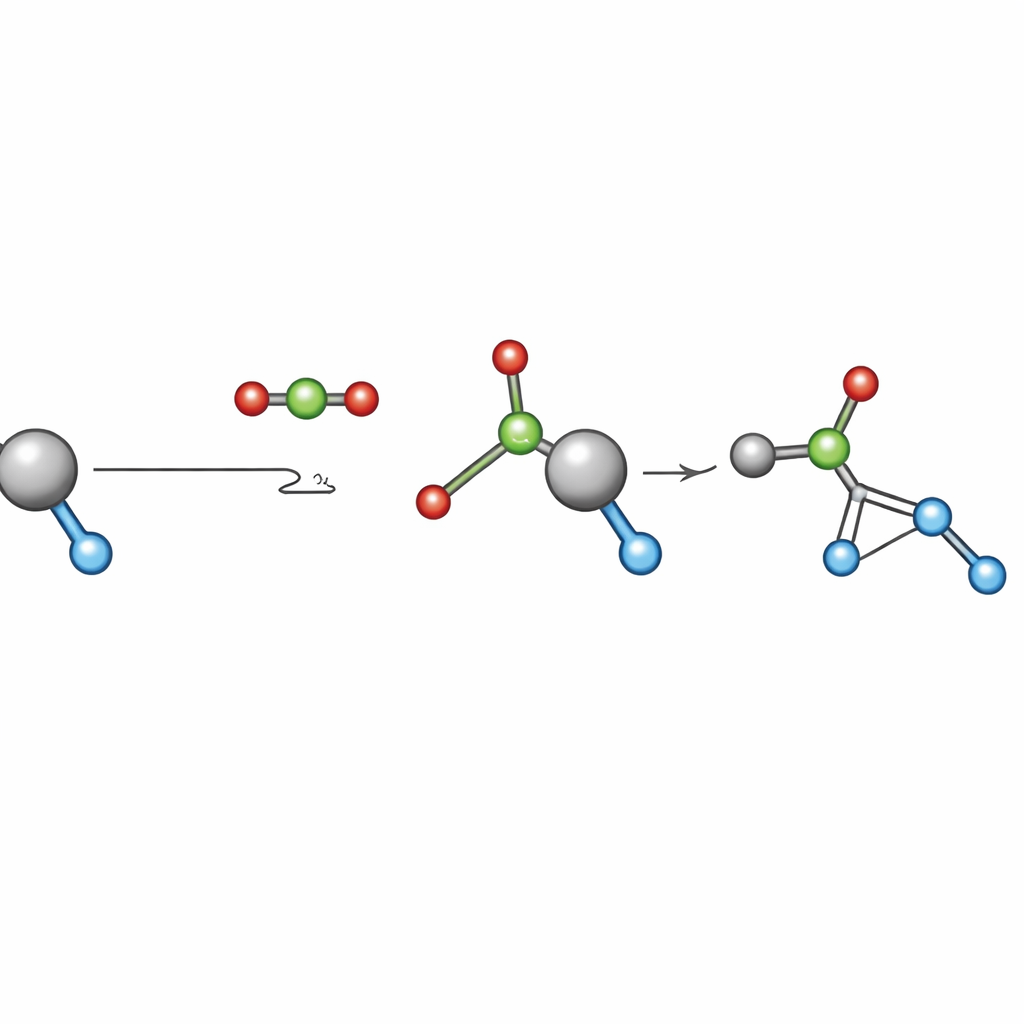
A critical but poorly understood step in many CO2-based processes is CO2 "insertion": CO2 reacts with a metal–oxygen bond inside the catalyst to form a new metal–carbonate species. This step is often assumed to be fast and unimportant for the overall rate, so it has rarely been examined under real production conditions. The team chose a widely studied reaction—ring-opening copolymerization of CO2 with epoxides such as propene oxide and cyclohexene oxide—to study this insertion step. They selected five of the best-known cobalt-based catalysts that already work at relatively low pressures and modest temperatures, then ran systematic tests between 2 and 30 bar of CO2 while carefully tracking how fast polymer formed.
Discovering a Pressure-Dependent Balance
Across all five catalysts, the reaction rates showed two clear regimes. At low CO2 pressure, the rate increased steadily with pressure: more CO2 pushed the balance inside the catalyst from a metal–alkoxide form toward a metal–carbonate form that actually carries out the key bond-forming step. Above a specific "threshold" pressure for each catalyst, adding more CO2 no longer helped—the rate leveled off because nearly all catalyst molecules were already in the active carbonate form. From these measurements, the authors extracted two practical numbers for each catalyst: an equilibrium constant, which measures how strongly CO2 is inserted into the catalyst, and a threshold pressure, the minimum CO2 pressure needed to reach maximum speed.
From Fundamental Numbers to Design Rules
When the researchers compared catalysts, a simple pattern emerged. Catalysts with larger CO2 insertion equilibrium constants ran faster and reached their maximum speeds at lower pressures. Poorer performers had weaker insertion and needed higher CO2 pressures to do their best. These correlations held not only for different cobalt complexes but also when switching the epoxide building block. The team showed that by measuring the reaction rate at just one moderate pressure (5 bar), they could predict both the equilibrium constant and the threshold pressure for that catalyst–monomer pair. They confirmed these predictions experimentally with additional catalysts, including a mixed-metal system, and found that one standout catalyst could already operate efficiently below 5 bar for certain monomers.
Guiding Future CO2 Recycling Technologies
For a non-specialist, the key outcome is that the authors have turned a complicated microscopic step—CO2 slipping into a metal–oxygen bond—into two simple, measurable numbers that tell engineers how to run a process with minimal energy. By linking catalyst structure to CO2 insertion strength and required operating pressure, the work offers a roadmap for designing next-generation catalysts that work quickly, cleanly and at low pressure. This approach could accelerate the development of scalable CO2-to-plastics technologies, helping transform a major greenhouse gas into useful products with far smaller energy and climate costs.
Citation: Thorogood, R., Eisenhardt, K.H.S., Smith, M.L. et al. Understanding low-pressure CO2 insertion chemistry in epoxide–CO2 copolymerization catalysis. Nat. Chem. 18, 931–938 (2026). https://doi.org/10.1038/s41557-026-02098-6
Keywords: carbon dioxide utilization, low-pressure catalysis, polycarbonate plastics, epoxide copolymerization, green chemistry