Clear Sky Science · en
Modeling epithelial deformation and cell rearrangement in response to external forces during Zebrafish epiboly
How an Early Embryo Grows a Protective Skin
Before a zebrafish embryo looks anything like a fish, it must perform a remarkable feat: a thin sheet of cells spreads over and seals in a giant yolk. This movement, called epiboly, is one of the first big shape changes in many fish embryos. Understanding how this fragile layer can stretch so far, stay intact, and move in a coordinated way helps scientists grasp how embryos build bodies—and why tissues sometimes fail, tear, or heal badly in disease and injury.

A Cell Sheet That Must Stretch Without Tearing
At the start of epiboly, the early zebrafish is mostly one huge yolk cell capped by thousands of much smaller cells. The outermost of these is a very thin “skin-like” sheet called the enveloping layer (EVL). During epiboly, the EVL has to spread from a small cap at the top of the yolk to wrap all the way around it, more than doubling its area. It must do this while staying sealed to the outside world and firmly attached to its neighbors, despite being pulled by forces generated deep in the yolk. Experiments have shown that the EVL’s edge starts out ragged and then straightens into a smooth ring as it moves, but the mechanical rules that make this possible have been unclear.
Building a Virtual Embryo
To probe these rules, the authors built a computer model of the EVL using an “agent-based” strategy. Instead of simulating every detail of cell shapes, they represented each EVL cell as a point—its center of mass—on the surface of a sphere standing in for the yolk. These points are linked by virtual springs that mimic both the cells’ resistance to being squashed and the stickiness of cell–cell junctions. The model pulls only on the cells at the exposed edge of the sheet, echoing how real embryos transmit force from the yolk through junctions at the margin. As the simulated time advances, the springs stretch or compress and the points move, allowing the layer to slide over the spherical yolk.
Letting Cells Swap Neighbors Safely
When cell–cell links in the model were fixed, the EVL behaved like a rubber band: it stretched but became jagged and recoiled strongly when the pulling stopped. Real EVL does something subtler—it remodels internally and does not snap back. To capture this, the researchers allowed links between neighboring cells to be randomly broken and re-formed with nearby cells, representing a coarse version of how epithelia exchange neighbors. They added a simple “energy” rule that favors local arrangements where each cell has roughly hexagon-like packing. This kept the layer from developing holes while still permitting extensive rearrangement. With this remodeling in place, the virtual EVL could spread around the yolk, thin out and shear, and then remain stable when the pull was removed. The tissue’s behavior was neither purely elastic nor purely plastic, but “viscoelastoplastic”: it deformed, relaxed, and partly locked into its new shape.
Keeping the Moving Edge in Step
One puzzle in living embryos is that all parts of the EVL edge reach the bottom of the yolk at nearly the same time. In the first version of the model, however, tiny random differences were amplified: one region of the edge would run ahead, forming a protrusion, while others lagged behind. Simply tightening the ring of edge cells did not fix this. The team then added a simple form of feedback: edge cells that were further from the vegetal pole were pulled slightly harder than those already closer. This negative feedback synchronized the motion of the edge without changing the overall expansion rate. Interestingly, both versions of the model—whether or not this feedback was present—spontaneously straightened the initially ragged edge as cells continually exchanged neighbors, suggesting that rapid local rearrangements and rising tension naturally smooth the boundary.
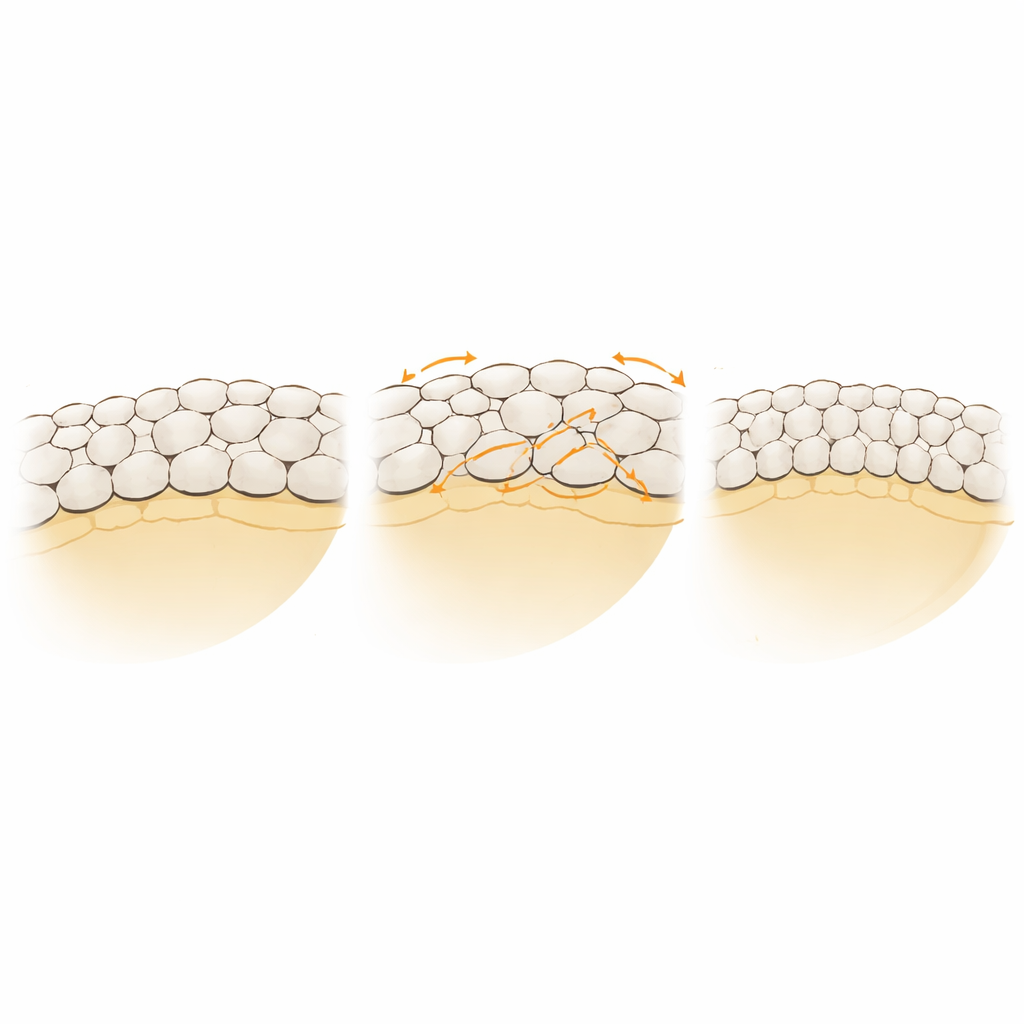
What This Reveals About Living Tissues
By distilling a complex embryonic movement into points and springs, the study pinpoints two key ingredients for realistic epiboly: a way for cells to reorganize their connections without breaking the sheet, and a feedback mechanism that balances pulling forces around the margin. Together these allow a delicate epithelial layer to spread far, change shape dramatically, and maintain its integrity. The work suggests that real embryos may use similar strategies—local junction remodeling that can “unjam” tissue where needed, and force regulation that keeps an advancing front in step. Such principles are likely to matter not just in fish development, but wherever tissues must expand, close gaps, or heal without falling apart.
Citation: Minsuk, S.B., Sego, T.J., Umulis, D.M. et al. Modeling epithelial deformation and cell rearrangement in response to external forces during Zebrafish epiboly. npj Syst Biol Appl 12, 63 (2026). https://doi.org/10.1038/s41540-026-00708-0
Keywords: zebrafish development, epiboly, epithelial mechanics, computational modeling, cell rearrangement