Clear Sky Science · en
Impact of PSA- versus STN-DBS on effective connectivity in Parkinson’s disease – a 3.0T resting-state fMRI study
Why this brain surgery study matters
For people with Parkinson’s disease, electrical stimulation deep inside the brain can dramatically steady shaking hands and ease stiffness. Surgeons already have a well‑tested target called the subthalamic nucleus (STN), but a nearby region—the posterior subthalamic area (PSA)—has emerged as a promising alternative, especially for patients whose main problem is tremor. This study uses advanced brain scanning to watch how each of these two stimulation sites reshapes communication inside the brain, with an eye toward more personalized treatment choices in the future.
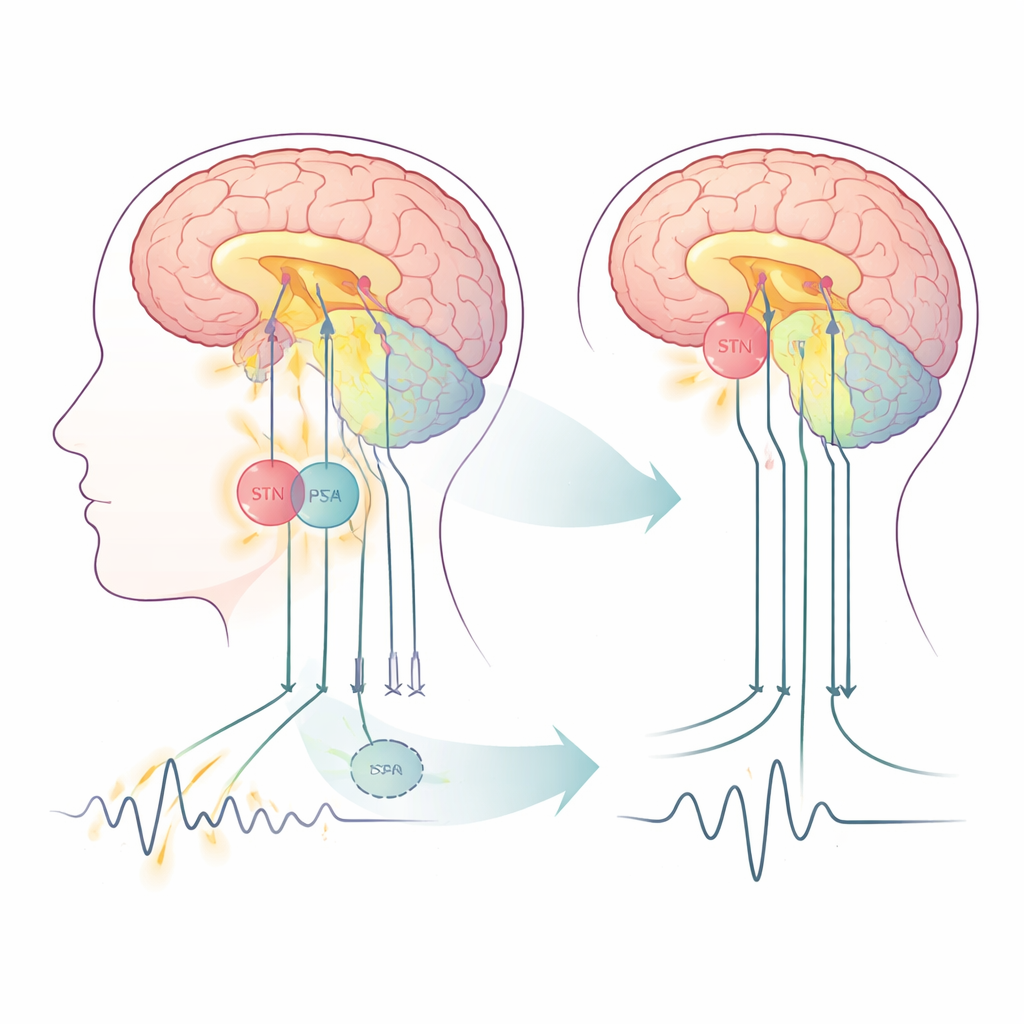
Two nearby switches in the movement network
Parkinson’s disease disrupts a set of brain circuits that control movement, involving the outer brain (cortex), deeper structures called the basal ganglia, and a “fine‑tuning” region at the back of the brain, the cerebellum. Deep brain stimulation (DBS) delivers rapid electrical pulses through implanted electrodes and is already an established treatment when medications are no longer enough. Traditionally, the STN has been the main target. The PSA, a fiber‑rich zone just behind and above the STN, is attracting interest because it connects strongly to tremor‑related pathways. In this study, fifteen people with tremor‑dominant Parkinson’s received a single pair of electrodes placed so that different contacts could stimulate either STN or PSA along the same track, allowing a rare, head‑to‑head comparison inside the same brains.
Watching resting brain traffic at high resolution
The researchers scanned each participant three times with a powerful 3‑Tesla MRI scanner: before surgery, with stimulation off, and after surgery while stimulating either the PSA or the STN. All scans were done at rest, without any movement task, to capture the brain’s ongoing activity patterns. Using a technique called resting‑state functional MRI, they measured slow fluctuations in blood oxygen that reflect underlying nerve cell activity. They then applied a modeling approach that estimates not just whether regions are linked, but how strongly one region drives another—known as “effective connectivity.” Two main loops were modeled: a cortex–basal ganglia loop involved in starting and stopping movement, and a cortex–cerebellum–thalamus loop thought to maintain and shape tremor.
Shared calming effects on overactive links
Both PSA and STN stimulation led to large and clinically meaningful improvements in movement scores, including tremor, slowness, stiffness, and posture, with no clear winner in overall symptom relief in this small group. In the brain networks, the two targets showed several common effects. Stimulation at either site dampened a direct pathway from the main movement area of the cortex down to the STN (“hyperdirect” input), and it increased the STN’s tendency to rein in its own activity. Both targets also reduced signals flowing from the cortex to the cerebellum. Importantly, the amount by which this cortex‑to‑STN pathway was quieted, and the strengthening of the STN’s self‑control, tracked with how much better patients moved: stronger changes in these connections went hand‑in‑hand with bigger gains in speed and overall motor scores.
Different emphasis on tremor‑related circuits
Despite these shared patterns, the two targets were not identical in how they shaped the network. PSA stimulation produced a stronger reduction of the cortex‑to‑cerebellum connection than STN stimulation did, and it boosted communication from the striatum (another basal ganglia structure) to the STN. Across all scan conditions, people with worse tremor tended to show stronger two‑way coupling involving the cerebellum, while overall and bradykinesia (slowness) scores were tied to the strength of cortex‑to‑STN connections. These relationships support a “two‑circuit” view of Parkinson’s tremor: basal ganglia regions act as a switch that triggers tremor, while the cerebellum‑centered loop acts like a dimmer that controls its intensity. The findings suggest that PSA stimulation may be especially effective at turning down this tremor “dimmer,” even if, within the limits of this small study, that extra physiological effect did not yet translate into clearly better tremor scores than STN stimulation alone.
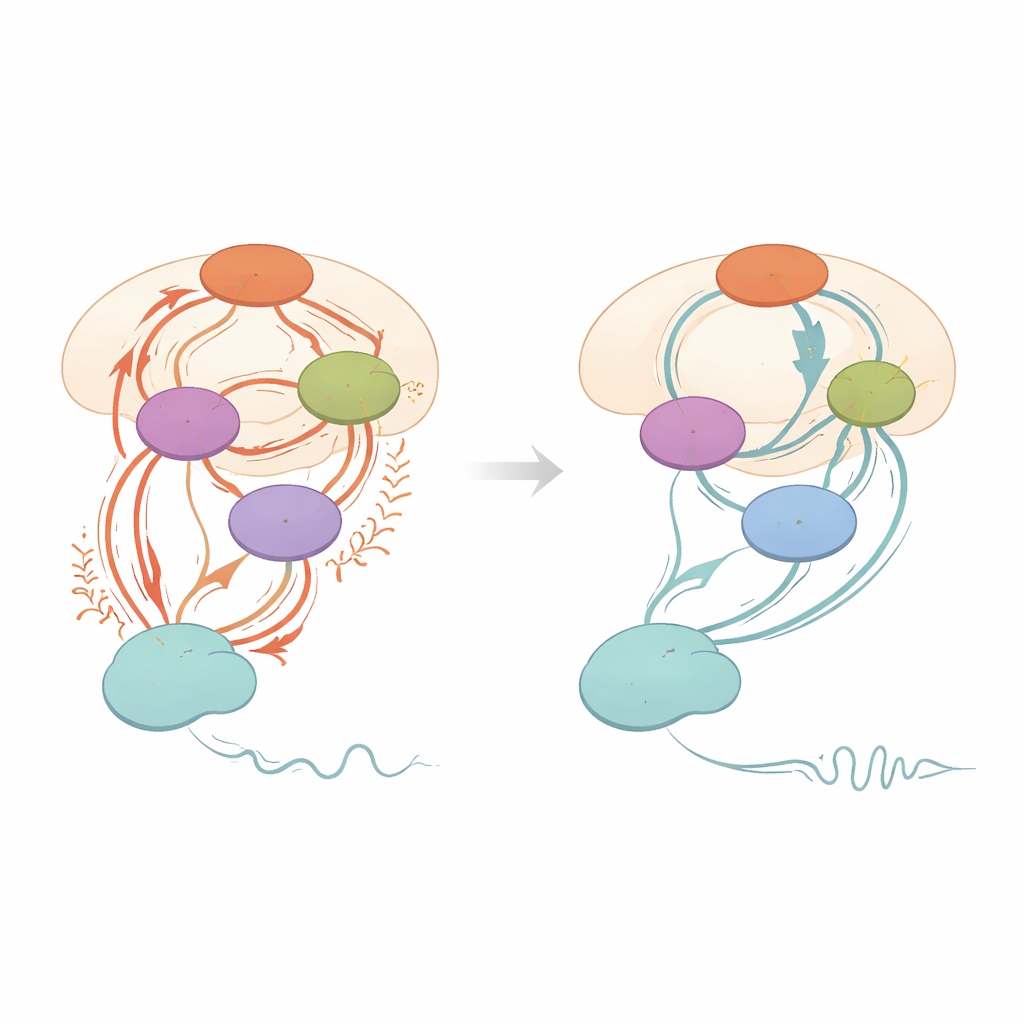
Toward tailored brain stimulation for Parkinson’s
In plain terms, this work shows that stimulating either of two closely spaced brain regions can calm abnormal traffic in key movement circuits and improve symptoms, but each target nudges the network in a slightly different way. STN stimulation strongly affects pathways linked to overall movement control, while PSA stimulation more powerfully dampens cerebellar routes tied to tremor maintenance. By placing modern, steerable electrodes along the border between these areas, clinicians may be able to “blend” stimulation—sending current more toward STN for rigidity and slowness, or more toward PSA for stubborn tremor. Although the study is small and focused on short‑term effects, it offers a mechanistic roadmap for turning DBS from a one‑size‑fits‑all procedure into a more precise, circuit‑guided therapy for people living with tremor‑dominant Parkinson’s disease.
Citation: Lin, Z., Zeng, Z., Duan, C. et al. Impact of PSA- versus STN-DBS on effective connectivity in Parkinson’s disease – a 3.0T resting-state fMRI study. npj Parkinsons Dis. 12, 92 (2026). https://doi.org/10.1038/s41531-026-01305-y
Keywords: Parkinson’s disease, deep brain stimulation, subthalamic nucleus, posterior subthalamic area, brain connectivity